Atrial fibrillation
Reading File
Finding Sources
Searching PubMed
"atrial fibrillation"[MeSH Terms] AND management
Searching Images
atrial fibrillation ECG irregular rhythm
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information to deliver a thorough, well-structured answer. Let me compile it:
Atrial Fibrillation
Definition & Epidemiology
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia in clinical practice, afflicting over 2 million Americans. Prevalence rises sharply with age — occurring in ~12% of patients ≥75 years and ~18% ≥85 years. A threefold increase in prevalence is anticipated over the next 20 years as the population ages. AF is almost always a recurrent disorder, with the possible exception of AF arising from hyperthyroidism or cardiac surgery.
ECG: The Classic 12-Lead
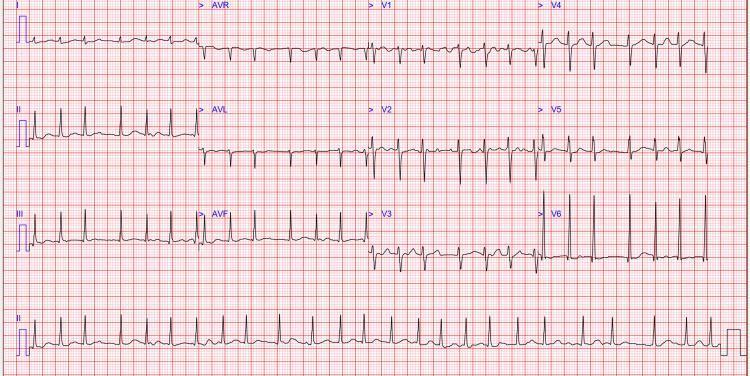
ECG hallmarks:
- Absent P waves — replaced by fine, low-amplitude fibrillatory (f) waves, best seen in V1
- Irregularly irregular ventricular rate — the defining feature
- Narrow QRS complexes (unless aberrant conduction or pre-excitation)
- Coarse AF shows residual atrial activity in V1 that can mimic flutter waves; irregularity of the QRS confirms AF
AF can paradoxically show a regular ventricular rate if concomitant complete heart block or ventricular tachycardia is present. At least 2 minutes of sustained arrhythmia consistent with AF is conventionally required for diagnosis.
Classification
| Type | Definition |
|---|---|
| Paroxysmal | Terminates spontaneously (usually within 7 days) |
| Persistent | Lasts ≥7 days or requires cardioversion to terminate |
| Long-standing persistent | Continuous AF >12 months |
| Permanent | Accepted by patient and physician; rhythm control no longer pursued |
| Silent/Subclinical | Detected on implanted device/wearable; ≥2 min required for diagnosis |
Pathophysiology
AF requires a trigger — most commonly atrial premature depolarizations originating from the pulmonary veins — acting on a susceptible atrial substrate. Key mechanisms:
- Electrical remodeling: prolonged AF shortens atrial refractory periods and promotes further perpetuation ("AF begets AF")
- Structural remodeling: atrial fibrosis and dilation create re-entrant circuits
- Loss of atrial kick: normally contributes ~15% of ventricular filling in healthy individuals, but up to 40% in patients with stiff, noncompliant ventricles (e.g., aortic stenosis, hypertrophic or restrictive cardiomyopathy, chronic hypertension) — explaining why these patients decompensate acutely with AF onset
- Tachycardia-induced cardiomyopathy: ventricular rates >120 bpm sustained for weeks causes biventricular dysfunction
Risk Factors & Associated Conditions
- Aging (most potent independent risk factor)
- Hypertension, coronary artery disease, heart failure, valvular disease
- Diabetes mellitus, obesity, obstructive sleep apnea
- Hyperthyroidism, COPD
- "Holiday heart": AF triggered by acute alcohol binge
- Vagally-induced AF: after vigorous exercise or a large meal
- ~20% of patients have no associated comorbidity ("lone AF")
Clinical Manifestations
| Symptom | Notes |
|---|---|
| Palpitations | Most common at onset; often dissipate with recurrent AF |
| Dyspnea, fatigue | From reduced cardiac output |
| Chest pain | Less common |
| Syncope | Usually a conversion pause when AF terminates |
| Acute pulmonary edema | In patients with stiff LV losing atrial kick |
| Asymptomatic | Not uncommon, especially with chronic AF at controlled rates |
Complications
1. Thromboembolism / Stroke
- Nonvalvular AF carries a fivefold increase in stroke risk
- Thrombus forms in the left atrial appendage (LAA) and embolizes
- Risk is independent of AF pattern (paroxysmal = persistent risk)
- Even subclinical/silent AF carries a 2.5× increased risk for ischemic stroke or systemic embolism
- ~12% of embolic events are extracranial (70% to extremities, remainder to mesenteric circulation)
- AF is also associated with 1.4× higher risk of cognitive impairment and dementia
2. Tachycardia-Induced Cardiomyopathy
- Ventricular rates >120 bpm for weeks → biventricular dysfunction
- Often reversible with rate/rhythm control
Stroke Risk Stratification: CHA₂DS₂-VASc Score
| Risk Factor | Points |
|---|---|
| C — Congestive heart failure | 1 |
| H — Hypertension | 1 |
| A₂ — Age ≥75 years | 2 |
| D — Diabetes mellitus | 1 |
| S₂ — Stroke/TIA (prior) | 2 |
| V — Vascular disease (CAD, PAD, aortic plaque) | 1 |
| A — Age 65–74 years | 1 |
| Sc — Sex category (female) | 1 |
Recommendations:
- ≥2 points: Anticoagulate (DOAC preferred over warfarin)
- 1 point: Anticoagulation or no therapy — shared decision with patient
- 0 points: No therapy required
All patients ≥75 years automatically score ≥2 and are candidates for anticoagulation regardless of AF pattern.
Initial Evaluation
Minimum workup:
- ECG (confirm AF, assess rate, identify pre-excitation)
- Labs: CBC, electrolytes, creatinine, TSH (thyroid disease is reversible cause)
- Echocardiogram: assess ventricular function, valvular disease, LA size
- Stress testing only if clinical/ECG evidence suggests ischemia
Treatment
A. Anticoagulation (Stroke Prevention)
DOACs (dabigatran, rivaroxaban, apixaban, edoxaban) are strongly preferred over warfarin for nonvalvular AF:
- No dietary restriction
- No INR monitoring
- Similar or better stroke prevention with similar or less bleeding vs. warfarin in patients ≥75 years
Exceptions requiring warfarin:
- Mechanical prosthetic valves — DOACs are contraindicated
- Mitral stenosis with AF — warfarin required (DOACs not validated)
Pre-cardioversion anticoagulation:
- AF >48 hours or unknown duration: full anticoagulation for ≥3 weeks before and 4 weeks after cardioversion OR TEE-guided approach to exclude LAA thrombus before proceeding
- Risk of embolization is highest in the first 3–4 weeks after cardioversion as atrial mechanical function gradually recovers
LAA occlusion (e.g., WATCHMAN device): non-inferior alternative to anticoagulation in patients at very high bleeding risk.
B. Rate Control vs. Rhythm Control
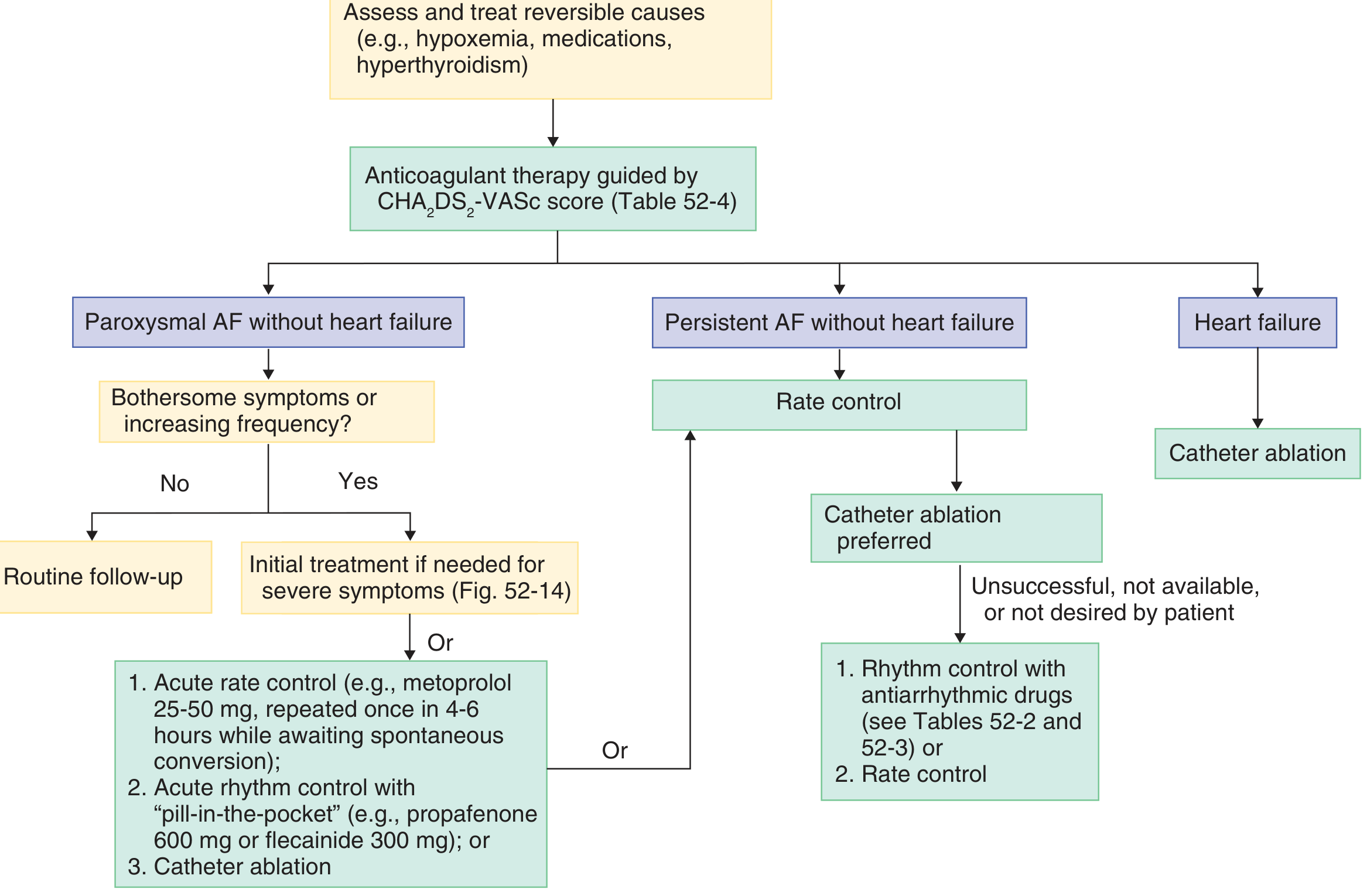
Rate Control
First-line for most patients with persistent AF, especially if minimally symptomatic.
| Drug Class | Agents | Notes |
|---|---|---|
| β-blockers | Metoprolol, atenolol, carvedilol | First-line; preferred with CAD or HFrEF |
| Non-DHP CCBs | Diltiazem, verapamil | Avoid in HFrEF (negative inotropy) |
| Digoxin | 0.25 mg IV/PO; reduce with renal impairment | Third-line; useful in HF with low output; narrow therapeutic window |
| Amiodarone | IV/PO | Reserved for refractory cases; multiple toxicities with long-term use |
Target heart rate: generally <110 bpm at rest (lenient control); <80 bpm if symptoms persist.
Rhythm Control
Preferred when:
- Symptomatic despite rate control
- Tachycardia-induced cardiomyopathy
- Persistent AF with bothersome symptoms
- Heart failure (catheter ablation is preferred)
Cardioversion:
- ~50% of acute-onset AF reverts spontaneously within 48–96 hours → "wait-and-see" approach is reasonable
- Electrical cardioversion (DC cardioversion): preferred for hemodynamic instability (hypotension, acute ischemia, worsening HF)
- Pharmacologic cardioversion: flecainide, propafenone ("pill-in-the-pocket" for paroxysmal AF); ibutilide, amiodarone for in-hospital use
Antiarrhythmic drugs (AADs) for maintaining sinus rhythm:
| Drug | Use case | Key caution |
|---|---|---|
| Flecainide / Propafenone | No structural heart disease | Contraindicated with CAD or HF |
| Sotalol | Moderate structural disease | QTc monitoring; renal dosing |
| Dofetilide | With HF or post-MI | In-hospital initiation; QTc monitoring |
| Amiodarone | Any structural heart disease | Most effective; multi-organ toxicity limits long-term use |
| Dronedarone | No permanent AF, no severe HF | Less effective than amiodarone; hepatotoxicity risk |
Catheter ablation (pulmonary vein isolation):
- Preferred for persistent AF (especially with HF) or paroxysmal AF refractory to AADs
- Superior to antiarrhythmic drugs alone for rhythm control
- Targets ectopic foci and re-entrant circuits around pulmonary vein ostia
Special Populations
Valvular AF (mitral stenosis): Embolic risk is particularly high — even higher than other forms of AF. Warfarin (not DOACs) is required. OAC is recommended regardless of CHA₂DS₂-VASc score and even for transient episodes detected on monitoring.
AF + CAD / post-stenting: Dual therapy (DOAC + P2Y12 inhibitor, without aspirin) is preferred over triple therapy to reduce bleeding. Dabigatran or rivaroxaban + clopidogrel are validated options.
AF + Heart failure: Catheter ablation is the preferred rhythm control strategy. Rate control with β-blockers is first-line; digoxin useful if low output state.
Elderly (≥75 years): All have CHA₂DS₂-VASc ≥2. DOACs preferred; dose adjustment based on age, weight, and renal function. Warfarin maintenance dose typically 2–5 mg/day.
Recent Evidence (2024–2025)
A 2024 meta-analysis in JACC Clinical Electrophysiology (PMID 38727662) comparing rhythm vs. rate control strategies across RCTs found updated comparative effectiveness data favoring early rhythm control in selected patients. A 2025 meta-analysis in JACC (PMID 39918465) confirmed that anticoagulation monotherapy (DOAC alone, without antiplatelet agents) is safest for most AF patients with stable coronary disease, reinforcing the move away from triple therapy.
Sources: Goldman-Cecil Medicine, 7th ed. | Braunwald's Heart Disease, 11th ed. | Washington Manual of Medical Therapeutics | JACC Clinical Electrophysiology 2024 | JACC 2025
This is a shared conversation. Sign in to Orris to start your own chat.