Adrenergic receptors
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the content needed for a comprehensive answer. Here is a complete overview of adrenergic receptors:
Adrenergic Receptors
Adrenergic receptors (adrenoceptors) are cell-surface receptors that respond to the catecholamines norepinephrine (NE) and epinephrine (EPI). All subtypes belong to the heptahelical (7-transmembrane domain) G protein-coupled receptor (GPCR) superfamily.
Classification
There are two major families — α and β — each with important subtypes:
| Receptor | Subtypes | G Protein | Primary 2nd Messenger | Key Locations |
|---|---|---|---|---|
| α₁ | α₁A, α₁B, α₁D | Gq | ↑ IP₃/DAG/Ca²⁺ (via PLC) | Vascular smooth muscle, iris, bladder |
| α₂ | α₂A, α₂B, α₂C | Gi | ↓ cAMP | Presynaptic nerve endings, platelets, pancreas |
| β₁ | — | Gs | ↑ cAMP (via adenylyl cyclase) | Heart (80% of cardiac β-receptors), JG cells (renin) |
| β₂ | — | Gs (+Gi) | ↑ cAMP | Bronchial smooth muscle, vascular SM (skeletal muscle), uterus, liver |
| β₃ | — | Gs | ↑ cAMP | Adipose tissue, bladder detrusor |
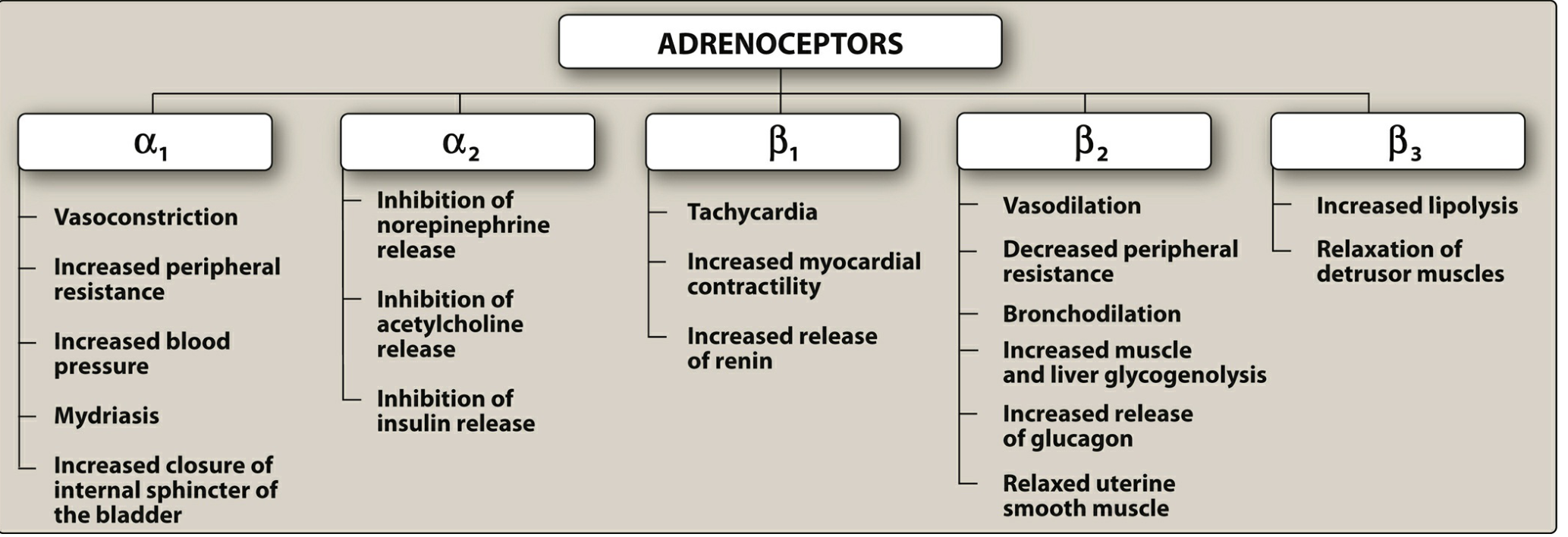
Receptor Effects — Overview Diagram
Agonist Potency (Rank Order)
- α receptors: EPI ≥ NE >> isoproterenol
- β receptors: isoproterenol > EPI > NE
- β₁ receptors: EPI ≈ NE (roughly equal affinity)
- β₂ receptors: EPI >> NE (tissues with β₂ are highly responsive to adrenal-medullary EPI)
Signaling Mechanisms
α₁ Receptors
Couple to Gq → activate phospholipase C-β → generate IP₃ (releases Ca²⁺ from ER) and DAG (activates PKC). The rise in cytosolic Ca²⁺ drives smooth muscle contraction and mediates hypertrophic/proliferative effects in cardiac myocytes. The α₁A subtype dominates vasoconstriction in most vascular beds; α₁D predominates in the aorta.
α₂ Receptors
Couple to Gi → inhibit adenylyl cyclase → ↓ cAMP. Also activate Gi-gated K⁺ channels (hyperpolarization) and inhibit voltage-gated Ca²⁺ channels. Physiologically, α₂A and α₂C receptors on presynaptic sympathetic nerve endings act as autoreceptors — released NE "circles back" to inhibit further NE release (negative feedback). They also inhibit ACh release from parasympathetic neurons (heteroreceptors), suppress sympathetic outflow from the CNS (→ antihypertensive action of clonidine), and inhibit insulin secretion from pancreatic β-cells.
β₁, β₂, β₃ Receptors
All three couple to Gs → activate adenylyl cyclase → ↑ cAMP → activate PKA → phosphorylate downstream targets (e.g., L-type Ca²⁺ channels, phospholamban, troponin I). β₂ receptors additionally couple to Gi in cardiac myocytes, producing a bifurcated response. β₂ receptors are largely confined to caveolae in cardiomyocytes, creating compartmentalized signaling.
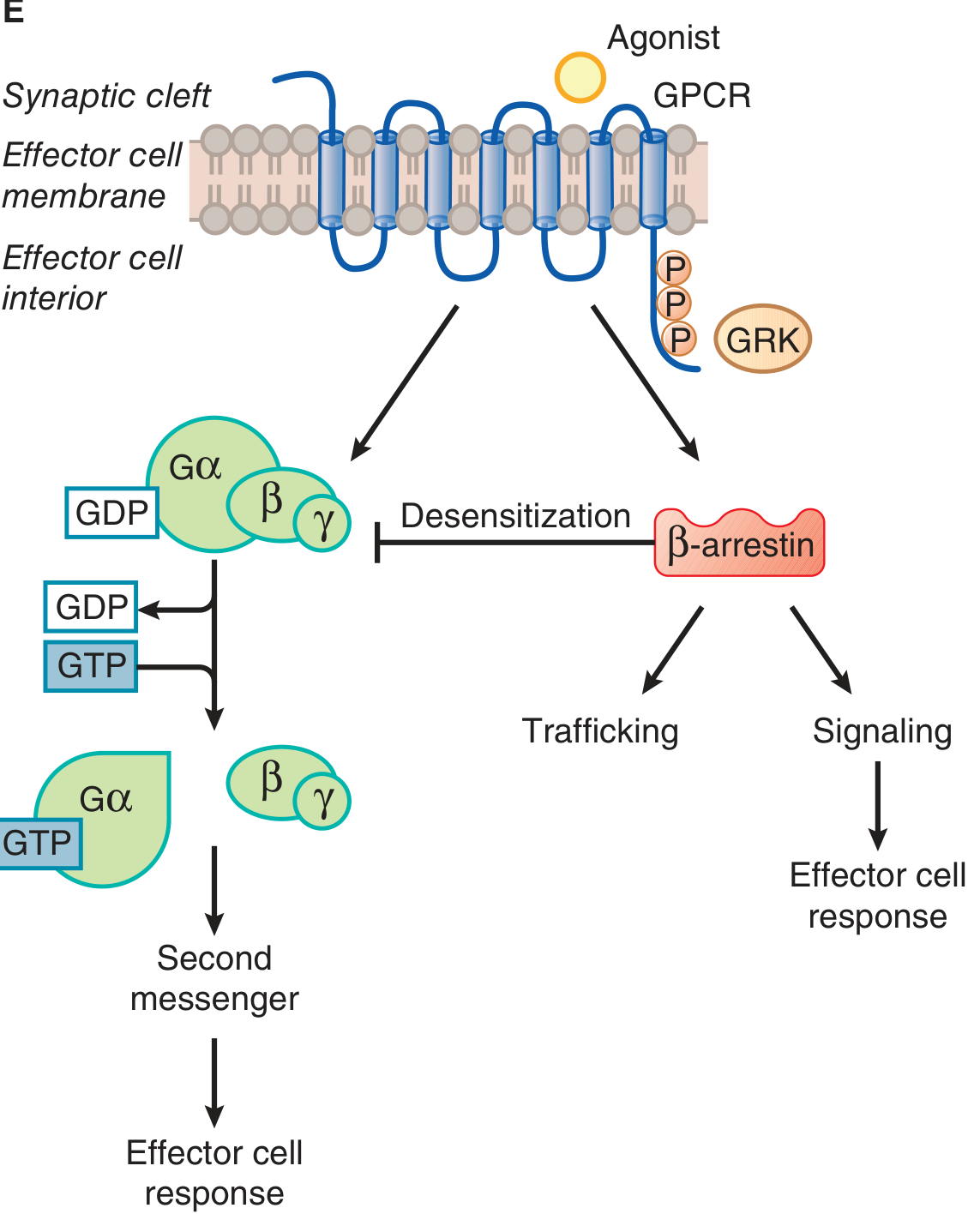
Signal Transduction Diagram
Physiological Effects by Receptor
α₁
- Vasculature: constriction of vascular smooth muscle (skin, viscera) → ↑ peripheral resistance → ↑ BP
- Eye: pupil dilation (mydriasis) via radial muscle contraction
- Bladder: contraction of internal urethral sphincter (urinary retention)
- Heart: mild positive inotropy; promotes cardiac hypertrophy (chronic)
α₂
- Presynaptic: inhibit NE release (autoreceptors); inhibit ACh release (heteroreceptors)
- CNS: ↓ sympathetic outflow → ↓ BP (basis for clonidine, dexmedetomidine)
- Pancreas: inhibit insulin secretion
- Platelets: promote aggregation
- Fat: inhibit lipolysis
β₁
- Heart: ↑ HR (chronotropy), ↑ contractility (inotropy), ↑ conduction velocity (dromotropy), ↑ lusitropy
- JGA (kidney): ↑ renin release → ↑ angiotensin II → ↑ aldosterone
β₂
- Bronchi: bronchodilation (basis for salbutamol/albuterol)
- Vasculature (skeletal muscle): vasodilation → ↓ peripheral resistance
- Uterus: relaxation (tocolysis)
- Liver/skeletal muscle: glycogenolysis → ↑ blood glucose
- Pancreas: ↑ glucagon release
β₃
- Adipose tissue: lipolysis
- Bladder: relaxation of detrusor muscle (basis for mirabegron in overactive bladder)
Desensitization and Downregulation
Prolonged catecholamine exposure reduces receptor responsiveness via three mechanisms:
- Uncoupling: receptor is phosphorylated by GRKs (GPCR kinases) on its cytoplasmic tail → β-arrestin binds → blocks G-protein access
- Sequestration: receptor–arrestin complex is internalized into endosomes (receptor is temporarily unavailable)
- Downregulation: prolonged internalization leads to lysosomal degradation or reduced synthesis → net loss of receptors
The β₂ receptor is the most susceptible to desensitization. In heart failure, β₁ receptor desensitization and downregulation are prominent (β₁ density can fall by ~50%), contributing to reduced cardiac reserve. β₁ receptor signaling has greater cardiotoxic potential than β₂, driving the rationale for β-blocker therapy in HF.
Clinically Relevant Drug Examples
| Drug | Action | Receptor(s) |
|---|---|---|
| Phenylephrine | Agonist | α₁ selective |
| Clonidine, dexmedetomidine | Agonist | α₂ selective |
| Prazosin, tamsulosin | Antagonist | α₁ (tamsulosin: α₁A selective) |
| Salbutamol/albuterol | Agonist | β₂ selective |
| Dobutamine | Agonist | β₁ predominant |
| Isoproterenol | Agonist | Non-selective β₁+β₂ |
| Propranolol | Antagonist | Non-selective β |
| Metoprolol, bisoprolol | Antagonist | β₁ selective |
| Carvedilol | Antagonist | β₁+β₂+α₁ |
| Mirabegron | Agonist | β₃ selective |
| Epinephrine | Agonist | α₁, α₂, β₁, β₂ |
| Norepinephrine | Agonist | α₁, α₂, β₁ >> β₂ |
Key Concept: Tissue Receptor Predominance
Most adrenergically innervated tissues express a predominant receptor type that determines the net response to sympathetic stimulation:
- Heart → predominantly β₁
- Bronchi / skeletal muscle vasculature → predominantly β₂
- Skin / splanchnic vasculature → predominantly α₁
- Presynaptic terminals → α₂ (autoreceptors)
- Adipose → β₃
This principle explains why drugs with receptor selectivity can produce targeted effects without activating the entire sympathoadrenal system.
Sources: Goodman & Gilman's The Pharmacological Basis of Therapeutics, 14e | Lippincott Illustrated Reviews: Pharmacology | Braunwald's Heart Disease, 12e
This is a shared conversation. Sign in to Orris to start your own chat.