Pda
Checking Date & Time
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Searching Images
patent ductus arteriosus anatomy diagram
I have comprehensive information. Let me compile a thorough answer on PDA.
Patent Ductus Arteriosus (PDA)
Anatomy & Embryology
The ductus arteriosus is derived from the sixth aortic arch and connects the main/left pulmonary artery to the upper descending thoracic aorta, just distal to the left subclavian artery. In fetal life, ductal flow accounts for ~60% of combined ventricular output, directed from pulmonary artery → aorta (bypassing the fluid-filled lungs). At birth it measures 2–8 mm long and 4–12 mm in diameter.
Mediators of ductal tone:
| Dilators (fetal) | Constrictors (postnatal) |
|---|---|
| PGE₂, PGI₂ (from placenta & lungs) | ↑ PaO₂ (main stimulus) |
| Hypoxia | Histamine, catecholamines, bradykinin, acetylcholine |
After birth, rising arterial oxygen tension is the primary trigger for smooth muscle contraction → functional closure within 10–15 hours. Anatomic obliteration by fibrosis produces the ligamentum arteriosum over the first few months of life.
Epidemiology
- Incidence: ~1 in 2000 births (isolated PDA); accounts for ~7% of all congenital heart lesions
- Female:Male = 2:1
- Markedly increased with prematurity: up to 75% of infants at 28–30 weeks gestation
- Associated with: high-altitude birth, congenital rubella, hypoxia, Down syndrome, other chromosomal abnormalities
Pathophysiology
A PDA creates a left-to-right shunt (aorta → pulmonary artery), whose magnitude depends on:
- Size and length of the ductus
- Ratio of pulmonary to systemic vascular resistance (PVR:SVR)
Hemodynamic consequences:
- ↑ Pulmonary blood flow → left atrial & left ventricular volume overload, LA/LV dilation
- ↓ Aortic diastolic pressure → wide pulse pressure, bounding pulses, risk of myocardial ischemia and underperfusion of systemic organs (especially gut → NEC in neonates)
- Progressive pulmonary hypertension → eventual Eisenmenger syndrome (right-to-left shunt, cyanosis) in ~5% of untreated cases
Clinical Features
Symptoms by Size
| PDA Size | Symptoms |
|---|---|
| Small | Usually asymptomatic; risk of infective endarteritis (~0.45%/year) |
| Moderate | Dyspnea, palpitations, exercise intolerance by 2nd–3rd decade |
| Large/unrestrictive | Heart failure, pulmonary hypertension, Eisenmenger syndrome |
Classic Signs
- "Machinery" murmur — continuous systolic and diastolic murmur at 1st–2nd left intercostal space below left clavicle; diastolic component shortens as pulmonary pressure rises; disappears in Eisenmenger physiology
- Widened pulse pressure, bounding/collapsing pulses (Corrigan's pulse)
- Hyperdynamic precordium, tachycardia, tachypnea
- Differential cyanosis and clubbing (toes > fingers) in Eisenmenger — because deoxygenated pulmonary artery blood shunts into the descending aorta distal to the subclavian arteries
Diagnosis
| Investigation | Findings |
|---|---|
| CXR | ↑ Pulmonary vascularity, cardiomegaly, dilated ascending aorta & pulmonary artery; calcification at PDA site in older patients |
| ECG | LV strain, left atrial enlargement, ± RV hypertrophy |
| Echocardiography | Directly visualizes PDA; Doppler color flow confirms shunt; estimates shunt size — primary diagnostic tool |
| Cardiac catheterization | Reserved when pulmonary hypertension suspected; assesses PVR before closure |
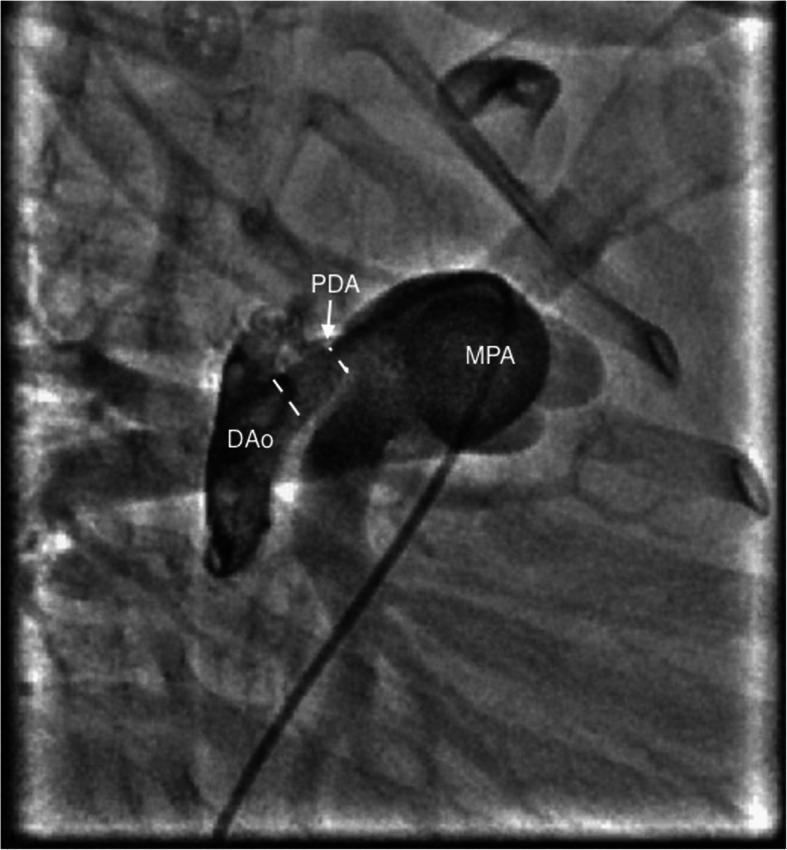
Treatment
The presence of a persistent PDA is an indication for closure due to increased mortality and endocarditis risk. Closure is generally contraindicated once Eisenmenger syndrome is established.
1. Pharmacologic (Premature Infants)
- Indomethacin or Ibuprofen (COX inhibitors → ↓ PGE₂) — first-line in preterm neonates
- Contraindicated with necrotizing enterocolitis, renal insufficiency, thrombocytopenia, or bleeding
2. Transcatheter Closure (Preferred in most children & adults)
- Gianturco coils — for small ducts (<3 mm)
- Amplatzer duct occluder — for moderate/large ducts
- Rashkind double-umbrella device
- Complications: thromboembolism, incomplete occlusion, vascular injury, endocarditis, device embolization
- May not be feasible in very small premature infants (inadequate vascular access)
3. Surgical Closure
- Open approach: muscle-sparing posterolateral thoracotomy, 3rd–4th intercostal space; ligation with surgical clip or suture
- Must protect the recurrent laryngeal nerve (wraps around PDA)
- Division + oversewing for short, broad ducts
- Video-assisted thoracoscopic (VATS) clip occlusion — also used
- Median sternotomy reserved for patients with concurrent cardiac lesions
Prognosis without treatment: estimated 30% mortality from isolated PDA; mortality rises to 3–4%/year by the 4th decade; two-thirds die by age 60.
PDA as a Lifesaver in Duct-Dependent Lesions
In cyanotic congenital heart disease (e.g., pulmonary atresia, critical aortic stenosis, hypoplastic left heart), ductal patency is essential to maintain pulmonary or systemic perfusion. Prostaglandin E₁ (alprostadil) is administered IV to keep the ductus open until definitive palliation or repair.
Sources: Schwartz's Principles of Surgery, 11th ed. — Goldman-Cecil Medicine — Robbins & Kumar Basic Pathology — The Developing Human (Clinically Oriented Embryology)
This is a shared conversation. Sign in to Orris to start your own chat.