Explain 1) bronchiectasis 2) pleural effusion 3) consolidation clinically and theoretically in detail with their investigations and management
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Searching Images
lung consolidation chest X-ray pneumonia lobar
I now have comprehensive material from the textbooks. Let me compile the full detailed answer.
1. Bronchiectasis
Definition & Pathophysiology
Bronchiectasis is a chronic condition characterized by local, irreversible dilatation of bronchi and bronchioles, usually associated with persistent inflammation. It is an acquired disorder where abnormal bronchial dilatation results from three main pathological mechanisms:
- Bronchial obstruction — mucus plugging with downstream collapse and secondary infection
- Bronchial wall damage — cytokines and enzymes from inflammatory cells, combined with bacterial toxins (from H. influenzae, S. aureus, Pseudomonas aeruginosa) create a vicious cycle of increasing wall damage, mucus retention, and bacterial proliferation
- Parenchymal fibrosis — traction bronchiectasis, where maturation and retraction of adjacent fibrous tissue mechanically dilates the airway
The central concept is a "vicious circle": recurrent infection → airway inflammation → impaired mucociliary clearance → more infection → progressive structural destruction.
(Murray & Nadel's Textbook of Respiratory Medicine; Grainger & Allison's Diagnostic Radiology)
Morphological Classification (Increasing Severity)
| Type | Description |
|---|---|
| Cylindrical | Uniform, parallel bronchial dilatation ("tramline" appearance) |
| Varicose | Non-uniform, serpiginous/beaded dilatation |
| Cystic (Saccular) | Progressive dilated sacs, often with air-fluid levels |
Causes / Etiology
| Category | Examples |
|---|---|
| Post-infective | Necrotising pneumonia (delayed treatment), TB, pertussis, measles |
| Genetic/Congenital | Cystic fibrosis (most common in young patients), Primary Ciliary Dyskinesia (Kartagener syndrome), α1-antitrypsin deficiency |
| Immunodeficiency | Hypogammaglobulinaemia, HIV, CVID |
| Allergic | Allergic Bronchopulmonary Aspergillosis (ABPA) |
| Obstructive | Endobronchial tumour, foreign body |
| Inflammatory/Autoimmune | Rheumatoid arthritis, IBD |
| Non-TB Mycobacteria | M. avium-intracellulare complex (MAC) — especially in middle-aged women |
| Idiopathic | ~50% of cases |
Clinical Features
Symptoms:
- Chronic productive cough — cardinal symptom; typically ≥30 mL/day of mucoid or mucopurulent sputum; sometimes in large volumes ("mouthfuls")
- Dyspnoea — variable, worsens with exacerbations
- Haemoptysis — can be mild to life-threatening (massive haemoptysis from bronchial artery erosion)
- Fever and weight loss — with infective exacerbations
- Postnasal drip / rhinosinusitis — co-exists in many patients
- "Dry bronchiectasis" — may have minimal or no sputum, especially in upper lobe disease (e.g., post-TB)
Signs:
- Persistent coarse crackles (crepitations) on auscultation — typically basal, but can be widespread
- Wheeze (in approximately 75%)
- Clubbing — in severe/longstanding disease
- Cyanosis in advanced disease
- Features of cor pulmonale in end-stage disease
Pathogens on sputum culture: Haemophilus influenzae (most common), Staphylococcus aureus, Pseudomonas aeruginosa (associated with severe disease), Moraxella catarrhalis, M. avium-intracellulare
Investigations
Imaging
Chest X-ray (CXR):
- Increased bronchial wall thickening visible as "tramlines" (parallel line opacities)
- When seen end-on, bronchiectatic airways appear as ring opacities
- Cystic bronchiectasis — multiple thin-walled ring shadows, some with air-fluid levels
- Lower lobe predominance in post-infective cases
- Overinflation in generalised disease (e.g., cystic fibrosis)
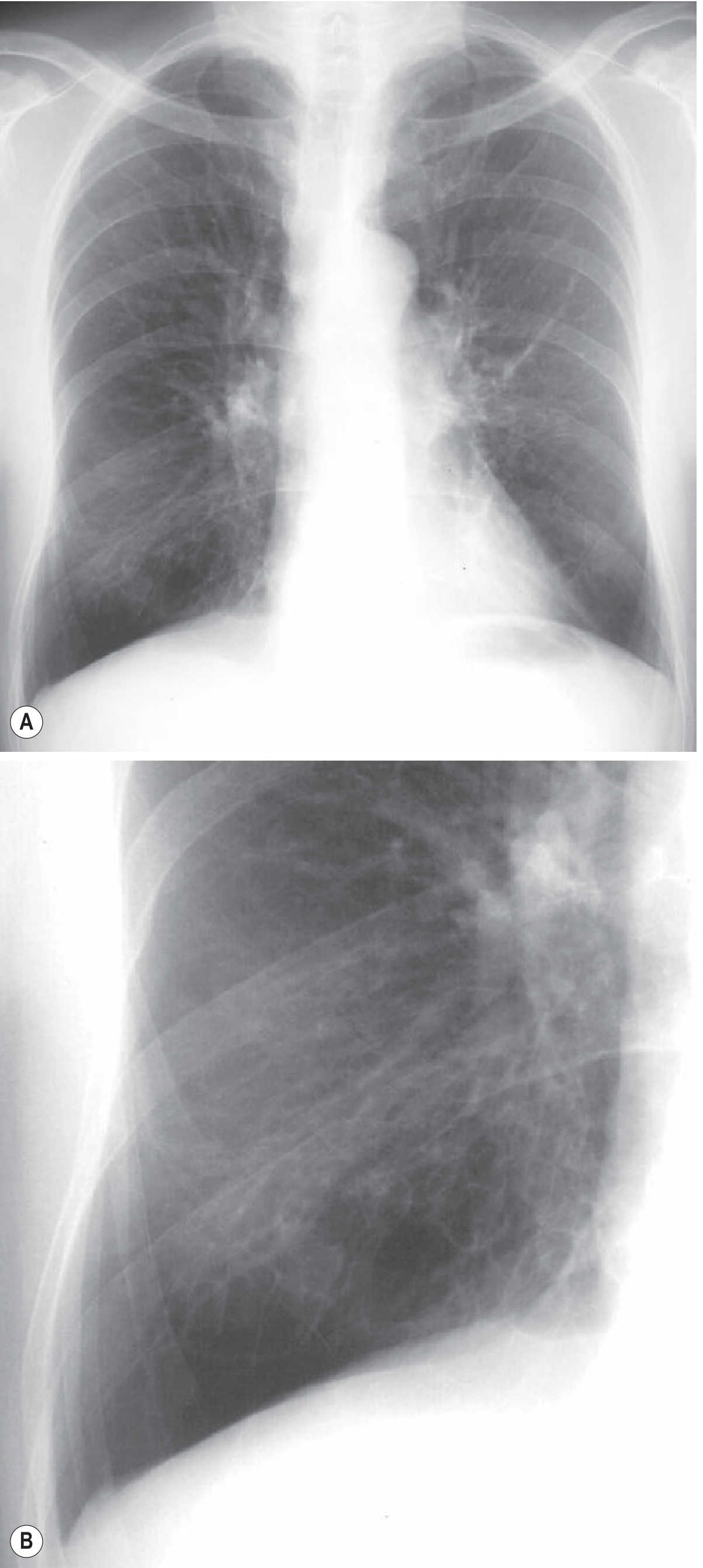
HRCT Chest (Investigation of Choice):
- Signet ring sign — bronchial internal diameter greater than that of the adjacent pulmonary artery (the artery is the "stone")
- Lack of tapering of bronchial lumina — cardinal sign
- Visualisation of bronchi within 1 cm of the costal pleura (normally not visible)
- Mucus plugging — lobulated "glove-finger," V- or Y-shaped densities
- Varicose type: beaded bronchial contour
- Cystic type: string of cysts or cluster of cysts; air-fluid levels in dependent portions
- Associated bronchiolitis, ground-glass change, tree-in-bud pattern
Laboratory
| Test | Purpose |
|---|---|
| Full blood count | Leukocytosis in infection; eosinophilia suggests ABPA |
| Immunoglobulins (IgA, IgG, IgM) — quantitative | Screen for hypogammaglobulinaemia |
| Sputum Gram stain and culture (bacteria, AFB, fungi) | Identify pathogens; guide antibiotics |
| Serum IgE, RAST/precipitins to Aspergillus spp. | Screen for ABPA |
| Sweat chloride / CFTR gene mutation | Cystic fibrosis screen |
| Nasal ciliary biopsy / exhaled NO | Primary ciliary dyskinesia |
| Spirometry + bronchodilator response | Obstructive pattern common; FEV₁/FVC ↓ |
| ABG | Assess hypoxaemia in advanced disease |
Management
Non-Pharmacological
- Chest physiotherapy / airway clearance techniques — essential; active cycle of breathing techniques (ACBT), postural drainage, flutter devices, high-frequency chest wall oscillation
- Pulmonary rehabilitation — exercise capacity improvement
- Smoking cessation
- Vaccinations — influenza, pneumococcal, pertussis
Pharmacological
| Indication | Treatment |
|---|---|
| Infective exacerbations | High-dose antibiotics guided by sputum culture; amoxicillin-clavulanate, or ciprofloxacin if Pseudomonas suspected; IV therapy for severe exacerbations |
| Chronic colonisation with Pseudomonas | Long-term inhaled tobramycin or colistin |
| Mucociliary clearance | Inhaled hypertonic saline; nebulised dornase alfa (in CF); mannitol powder |
| Bronchospasm | Inhaled β₂-agonists (also aid mucociliary clearance) |
| Reduce exacerbations | Long-term macrolide therapy (azithromycin 250 mg 3×/week) — reduces exacerbation frequency and improves lung function |
| ABPA | Systemic corticosteroids ± itraconazole |
| Haemoptysis | Bronchial artery embolisation; surgery for massive haemoptysis |
Surgical
- Surgical resection — only curative option for localised disease; indicated when medical management fails and disease is anatomically confined
- Lung transplantation — for end-stage bilateral disease (especially CF)
2. Pleural Effusion
Definition & Pathophysiology
A pleural effusion is an abnormal accumulation of fluid in the pleural space (normally contains only 10–20 mL). Fluid accumulates when production exceeds absorption, due to alterations in:
- Hydrostatic pressure ↑ (e.g., heart failure) → transudate
- Oncotic pressure ↓ (e.g., hypoalbuminaemia, cirrhosis) → transudate
- Capillary permeability ↑ (e.g., inflammation, infection, malignancy) → exudate
- Lymphatic obstruction (e.g., malignancy) → exudate
- Direct passage from other cavities (e.g., hepatic hydrothorax via diaphragmatic defects; ascites via peritoneal-pleural communication)
(Fishman's Pulmonary Diseases and Disorders)
Aetiology — Most Common Causes (USA Annual Incidence)
| Cause | Incidence |
|---|---|
| Congestive heart failure | 500,000 |
| Infectious (parapneumonic) | 300,000 |
| Malignancy | 200,000 |
| Pulmonary embolism | 150,000 |
| Viral illness | 100,000 |
| Cirrhosis (hepatic hydrothorax) | 50,000 |
| Post-cardiac surgery | 50,000 |
Laterality clues (Grainger & Allison):
- Right-sided → heart failure, ascites, liver abscess
- Left-sided → pancreatitis, pericarditis, oesophageal rupture, aortic dissection
- Bilateral → usually transudates (heart failure, hypoalbuminaemia); bilateral exudates: malignancy, lymphoma, SLE, RA, pulmonary embolism
- Massive effusion → most commonly malignancy, but also empyema, TB, cirrhosis
Clinical Features
Symptoms:
- Dyspnoea — proportional to fluid volume and rate of accumulation
- Pleuritic chest pain — sharp, worse on inspiration, suggests inflammatory/exudative aetiology; pleurisy may precede effusion
- Dry, non-productive cough — from pleural irritation
- Systemic features — fever (infection/malignancy), weight loss (malignancy/TB), ankle oedema (cardiac)
Signs (classical on affected side):
- Inspection: reduced chest expansion, tracheal deviation away (large effusion)
- Palpation: reduced tactile vocal fremitus (TVF)
- Percussion: stony dullness — pathognomonic; dullness extends to the level of the effusion; above the effusion there may be an area of increased resonance (Skodaic resonance)
- Auscultation: absent/reduced breath sounds; bronchial breathing at the upper level of the effusion (compressed lung); reduced vocal resonance; pleural friction rub if early/resolving
- Mediastinal shift: away from a large effusion (towards if associated with collapse)
Investigations
Imaging
Chest X-ray (PA erect):
- Small effusion (200–500 mL): blunting of the posterior then lateral costophrenic angle
- Moderate: homogeneous opacification of lower chest, obliteration of hemidiaphragm, concave (meniscus) upper border — higher laterally than medially
- Large (>1000 mL): reaches level of anterior 4th rib
- Massive: dense hemithoracic opacification with contralateral mediastinal shift
- Absence of shift with a large effusion → suspect ipsilateral collapse, or malignant "frozen mediastinum" (mesothelioma, carcinoma)
- Subpulmonary effusion: apparent "high hemidiaphragm" peaking laterally
- Supine CXR (ICU): hazy opacity over lower hemithorax — easily missed
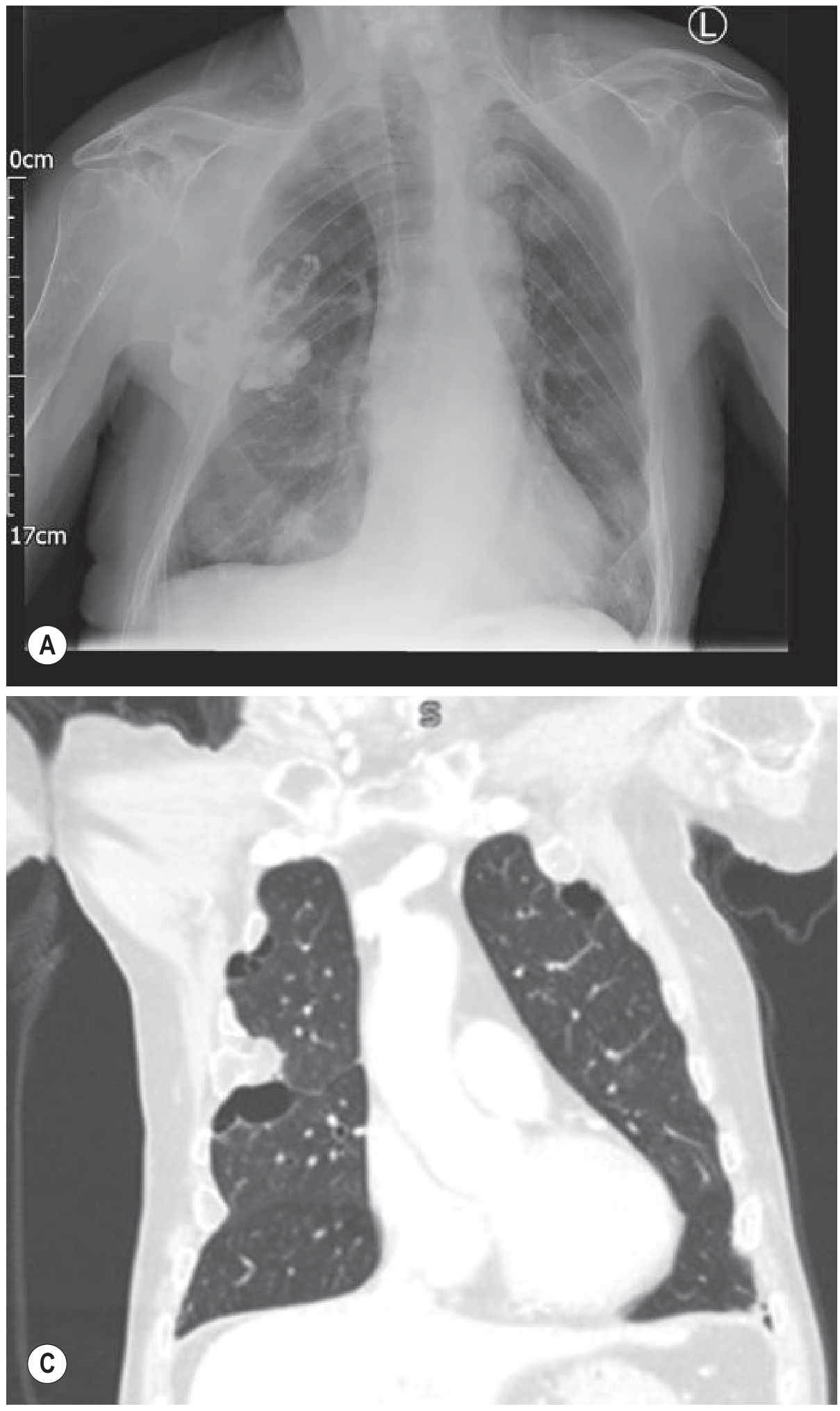
Thoracic Ultrasound (preferred next step after CXR):
- Detects effusions as small as 5–50 mL
- Transudate: typically anechoic
- Exudate: may be complex septated, complex non-septated, or homogenously echogenic
- Pleural thickening >7 mm or pleural nodule → suggests malignancy
- Guides safe thoracentesis — significantly improves safety
CT Chest:
- Characterises the effusion and underlying lung pathology
- Identifies pleural thickening, nodules, malignant features
- Differentiates effusion from consolidation/collapse in complex cases
Thoracentesis & Pleural Fluid Analysis
Indicated when effusion ≥1–2 cm on ultrasound and no clear aetiology (e.g., bilateral effusions in obvious CHF can be treated with diuresis first).
The key step: Transudate vs. Exudate — Light's Criteria (1972)
An effusion is an EXUDATE if ANY one criterion is met:
| Criterion | Cut-off |
|---|---|
| Pleural fluid protein / serum protein | > 0.5 |
| Pleural fluid LDH / serum LDH | > 0.6 |
| Pleural fluid LDH | > 2/3 upper limit of normal serum LDH |
Sensitivity ~98% for exudates. However ~25% of transudates are misclassified as exudates (especially with diuresis in CHF). In such cases, the albumin gradient (serum albumin − pleural fluid albumin >12 g/L) reclassifies these back to transudates.
Additional pleural fluid tests:
| Test | Result | Suggested Condition |
|---|---|---|
| pH | <7.2 | Empyema, malignancy, oesophageal rupture |
| Glucose | <3.3 mmol/L | Empyema, RA, malignancy, TB |
| Amylase | ↑ | Pancreatitis, oesophageal rupture |
| LDH | Very high | Malignancy, empyema |
| Lymphocytes >85-95% | — | TB, malignancy, lymphoma, sarcoid |
| Neutrophils >10,000/μL | — | Parapneumonic, acute pancreatitis |
| Eosinophils >10% | — | Haemothorax, pneumothorax, parasitic, drug-induced |
| Triglycerides >1.2 mmol/L | — | Chylothorax |
| Haematocrit PF/serum >0.5 | — | Haemothorax |
| Cytology | Malignant cells | Malignancy (~60% sensitivity) |
| Culture + AFB smear | Organisms | Infection, TB |
| ADA (adenosine deaminase) | >40 IU/L | TB pleurisy |
Pleural Biopsy
- Indicated for undiagnosed exudative effusion after thoracentesis
- Closed Abrams needle biopsy: random sampling, lower yield
- CT/ultrasound-guided percutaneous biopsy: targeted, better yield
- Medical thoracoscopy (pleuroscopy): under local anaesthesia/sedation; diagnostic yield >90% for malignancy; can do pleurodesis simultaneously
- VATS: under GA; allows more complex procedures
Management
Transudative Effusions
Treat the underlying cause:
- CHF → diuretics, ACE inhibitors, cardiac optimisation
- Cirrhosis → diuretics (spironolactone ± furosemide), sodium restriction; TIPS for refractory hepatic hydrothorax
- Hypoalbuminaemia → nutritional support, treat cause
- Pulmonary embolism → anticoagulation
Exudative Effusions
Parapneumonic effusion / Empyema:
- Antibiotics (IV for complicated effusion/empyema)
- Uncomplicated (free-flowing, pH >7.2, glucose normal) → antibiotics alone
- Complicated (pH <7.2, glucose <3.3, LDH >1000, or frank pus) → chest drain (intercostal tube drainage)
- Fibrinous/loculated → intrapleural fibrinolytics (alteplase + DNase)
- Non-resolving empyema → VATS decortication
Malignant effusion:
- Therapeutic thoracentesis for symptomatic relief
- Indwelling pleural catheter (IPC) — allows outpatient drainage; first-line for trapped lung or recurrent effusion
- Pleurodesis (talc, via medical thoracoscopy or slurry via drain) — obliterates pleural space; indicated if lung fully expands
- Systemic therapy (chemotherapy/targeted agents) if responsive tumour
Tuberculous effusion:
- Standard anti-TB chemotherapy (2HRZE/4HR)
- Adjunct steroids controversial but may reduce fibrinous organisation
Haemothorax:
- Chest drain; treat cause; if clotted/retained → VATS
3. Consolidation
Definition & Pathophysiology
Pulmonary consolidation is the filling of alveolar airspaces with material (pus, fluid, blood, cells, or protein), resulting in air being displaced from the alveoli, rendering the lung parenchyma solid/airless. The lung tissue itself is intact but the air is replaced, so the lung conducts sound rather than transmitting normal vesicular breath sounds — this is why bronchial breathing is heard over consolidated lung (airway conducting sounds directly through solid parenchyma to the chest wall).
Mechanisms leading to consolidation:
- Infection (pneumonia) — most common; bacteria/viruses/fungi fill alveoli with inflammatory exudate (PMNs, fibrin, red cells)
- Lung malignancy — lepidic adenocarcinoma (BAC) fills alveoli with tumour cells
- Pulmonary oedema — alveolar flooding with oedema fluid
- Haemorrhage — alveolar haemorrhage (vasculitis, trauma, anticoagulation)
- Aspirate/lipoid pneumonia — aspiration of gastric contents or oils
- Inflammatory — organising pneumonia (BOOP/COP), eosinophilic pneumonia, sarcoidosis
- Others — pulmonary alveolar proteinosis, alveolar lipoproteinosis
(Murray & Nadel's Textbook of Respiratory Medicine)
Pathological Stages (Lobar Pneumonia — Classic)
- Congestion (day 1–2): vascular engorgement, serous exudate
- Red hepatisation (day 3–4): massive PMN + RBC + fibrin exudate; lung firm, red, airless ("liver-like")
- Grey hepatisation (day 5–7): RBCs lyse, fibrin persists, WBCs continue; grey, firmer
- Resolution (day 8+): enzymatic digestion of exudate, macrophage clearance; lung re-aerates
Clinical Features
Symptoms:
- Productive cough — purulent sputum; "rusty" or blood-tinged sputum in pneumococcal pneumonia
- Fever — high fever (38.5–40°C), often with rigors ("shaking chill" in pneumococcal)
- Dyspnoea — proportional to extent of consolidation
- Pleuritic chest pain — localised, worse on breathing and coughing
- Myalgia, malaise, anorexia — systemic features
- In atypical pneumonia (Mycoplasma): dry cough, gradual onset, extrapulmonary features (rash, haemolytic anaemia, cold agglutinins)
- Elderly patients may present atypically — confusion, falls, without classical features
Signs (over the consolidated area):
- Inspection: reduced chest expansion on affected side
- Palpation: increased tactile vocal fremitus (TVF) — solid lung conducts sound better
- Percussion: dull (but less stony than effusion; percussion note is dull, not stony dull)
- Auscultation:
- Bronchial breathing — harsh, high-pitched; expiration longer than inspiration; pathognomonic of consolidation
- Coarse crackles (crepitations) — from alveolar opening during inspiration
- Increased vocal resonance — aegophony (bleating quality), whispering pectoriloquy (whispered speech clearly heard)
- Pleural friction rub if pleurisy is present
Key distinction:
| Sign | Consolidation | Pleural Effusion |
|---|---|---|
| TVF/VR | Increased | Decreased |
| Percussion | Dull | Stony dull |
| Breath sounds | Bronchial | Absent/reduced |
| Aegophony | Present | Present (at upper border) |
Investigations
Imaging
Chest X-ray:
- Homogeneous (dense) opacification of a lobe, segment, or portion thereof
- Air bronchogram — patent bronchi visible as dark branching lines through the white consolidation; indicates the bronchus is patent while the surrounding alveoli are airless — essentially diagnostic of consolidation
- Lobar pneumonia → consolidation respects lobar anatomy, bordered by fissure
- Segmental or multilobar in Klebsiella, hospital-acquired organisms
- Borders are ill-defined (unlike a mass), except when bounded by a fissure
- Lobar consolidation with expansion (bulging fissure) → Klebsiella pneumoniae
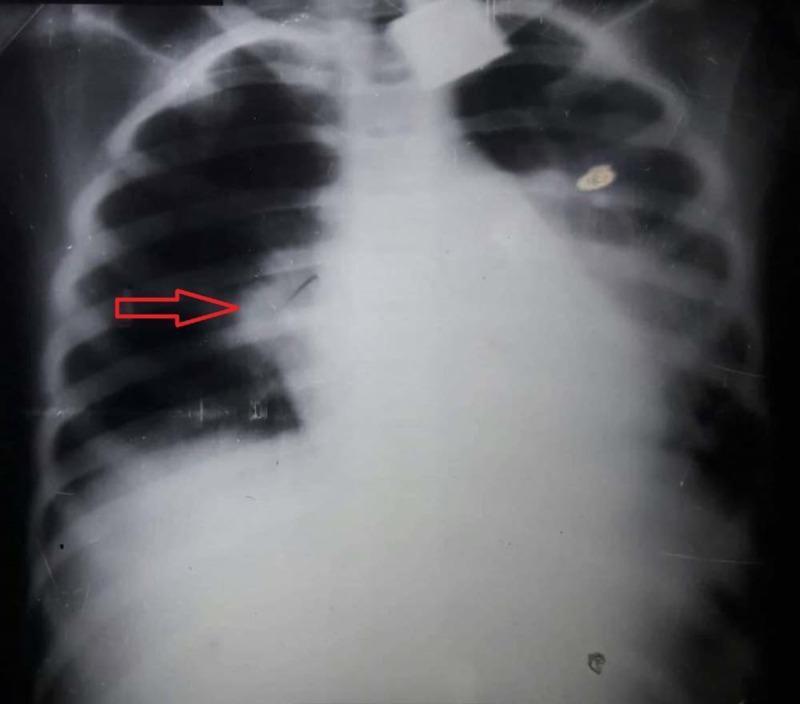
CT Chest:
- More sensitive than CXR; shows extent, multilobar involvement
- Air bronchograms are exquisitely seen
- "CT angiogram sign" — enhancement of branching pulmonary vessels visible through low-attenuation consolidation; seen in lepidic adenocarcinoma and lobar pneumonia
- Identifies complications: cavitation (abscess), empyema, bronchiectasis
- Differentiates consolidation from atelectasis, mass, effusion
Lung Ultrasound:
- Consolidated lung has an echogenic (liver-like) "hepatisation" pattern — solid appearance
- Air bronchograms visible as hyperechoic branching structures that move with respiration (distinguishes from atelectasis where bronchograms are static)
- "Shred sign" / fractal sign at transition zone of consolidation
- B-lines absent in completely consolidated lung
Laboratory
| Test | Relevance |
|---|---|
| FBC | Neutrophilia (bacterial); lymphocytosis (viral); leukopenia (severe infection, viral, Legionella) |
| CRP, ESR, Procalcitonin | Inflammatory markers; procalcitonin guides antibiotic need |
| U&E, LFTs, Glucose | Baseline; liver involvement in atypical organisms |
| Blood cultures × 2 (before antibiotics) | Positive in ~10% CAP; guides targeted therapy |
| Sputum Gram stain + culture | Aerobes, anaerobes; sensitivity varies |
| Urine pneumococcal antigen | Positive in ~60–80% pneumococcal pneumonia; rapid, specific |
| Urine Legionella antigen | Detects serogroup 1 (responsible for ~80% cases); rapid |
| Serology (paired acute/convalescent) | Mycoplasma, Chlamydophila, Legionella, viruses |
| Cold agglutinins | Mycoplasma pneumoniae |
| ABG / SpO₂ | Severity assessment |
| Bronchoscopy + BAL | Immunocompromised patients; non-resolving consolidation; Pneumocystis, CMV, fungi |
Severity Scoring — CAP
CURB-65 Score (each scores 1 point):
- C — Confusion (new)
- U — Urea > 7 mmol/L
- R — Respiratory rate ≥ 30/min
- B — Blood pressure (systolic <90 mmHg or diastolic ≤60 mmHg)
- 65 — Age ≥ 65 years
| Score | Severity | Management |
|---|---|---|
| 0–1 | Low | Outpatient |
| 2 | Intermediate | Hospital |
| 3–5 | High (ICU consider) | Urgent hospitalisation |
Management — Pneumonia (Most Common Cause)
Community-Acquired Pneumonia (CAP)
| Setting | Organism | Treatment |
|---|---|---|
| Mild/outpatient | S. pneumoniae, Mycoplasma | Amoxicillin 500 mg–1 g TDS ± clarithromycin (atypical cover) |
| Moderate/hospital | As above + Legionella, Gram-neg | IV co-amoxiclav + clarithromycin; or levofloxacin monotherapy |
| Severe/ICU | S. pneumoniae, Legionella, Gram-neg, S. aureus | IV co-amoxiclav + clarithromycin ± oseltamivir if influenza |
| Aspiration | Anaerobes, Gram-neg | Co-amoxiclav; metronidazole + co-trimoxazole |
| Hospital-acquired | S. aureus (MRSA), Gram-neg incl. Pseudomonas | Piperacillin-tazobactam ± gentamicin; vancomycin/linezolid if MRSA |
Supportive care:
- Supplemental oxygen (target SpO₂ 94–98%; 88–92% in COPD)
- IV fluids for sepsis/dehydration
- Analgesia for pleuritic pain (NSAIDs/paracetamol; avoid opioids)
- VTE prophylaxis
- Physiotherapy for sputum clearance
Duration of antibiotics: typically 5–7 days for uncomplicated CAP (guided by CRP and clinical response); 10–14 days for Legionella, atypicals, or severe disease.
Non-Resolving Consolidation
If not improving at 48–72 hrs, consider:
- Wrong antibiotic / resistant organism → repeat cultures, escalate
- Post-obstructive pneumonia → bronchoscopy
- Malignancy → CT/bronchoscopy
- Organising pneumonia (COP) → responds to corticosteroids
- Pulmonary TB → sputum AFB
When to suspect Malignancy
- Non-resolving consolidation after 6–8 weeks of antibiotics
- Older patient, smoker, no fever
- "Bat-wing" or peribronchovascular distribution → may suggest COP or lepidic adenocarcinoma
- Follow-up CXR at 6 weeks mandatory in patients >50 years after treated pneumonia
Summary Comparison Table
| Feature | Bronchiectasis | Pleural Effusion | Consolidation |
|---|---|---|---|
| Definition | Irreversible bronchial dilatation | Fluid in pleural space | Alveolar airspace filling |
| TVF/VR | Normal/↓ | ↓↓ | ↑↑ |
| Percussion | Normal | Stony dull | Dull |
| Breath sounds | Coarse crackles, wheeze | Absent | Bronchial breathing |
| CXR hallmark | Tramlines, ring shadows | Meniscus sign | Air bronchogram |
| Gold standard imaging | HRCT chest | Thoracic ultrasound | CT chest |
| Key lab | Sputum culture | Pleural fluid Light's criteria | Blood culture, urinary antigens |
| Key treatment | Airway clearance + macrolides + antibiotics | Treat cause / drain | Antibiotics ± supportive care |
Sources: Murray & Nadel's Textbook of Respiratory Medicine (7e); Grainger & Allison's Diagnostic Radiology; Fishman's Pulmonary Diseases and Disorders; Textbook of Family Medicine 9e
This is a shared conversation. Sign in to Orris to start your own chat.