APGAR score
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Searching Images
APGAR score newborn assessment table
Loading Image
Here is a comprehensive overview of the APGAR Score:
APGAR Score
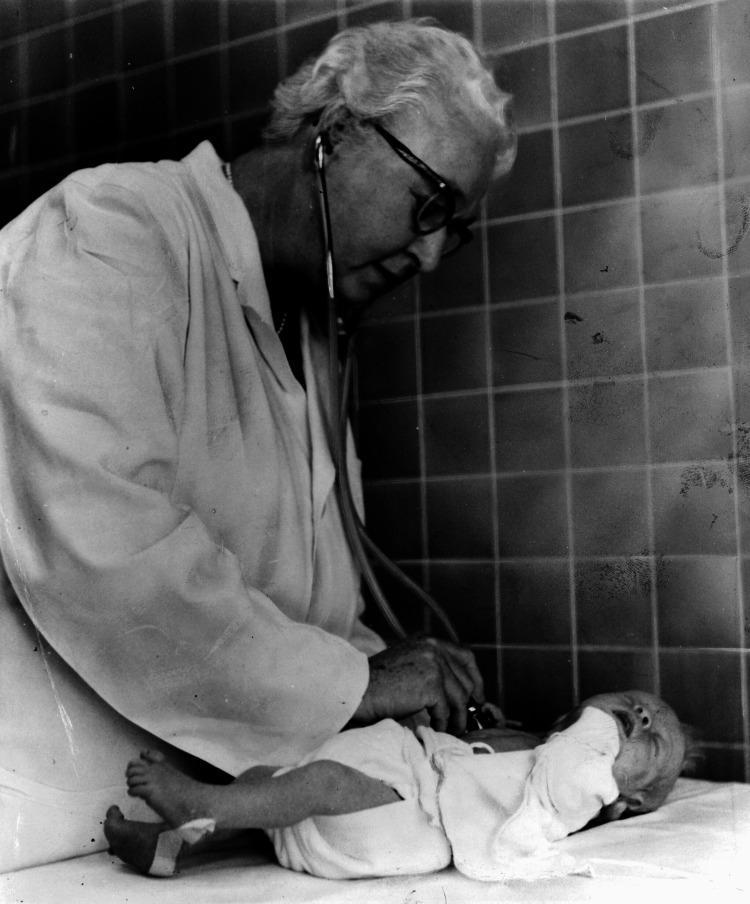
History & Purpose
The Apgar score was devised by Dr. Virginia Apgar in 1952 as a standardized method to assess the physical condition of newborns immediately after birth. It is also used to evaluate an infant's response to resuscitation.
The Mnemonic: A-P-G-A-R
The five criteria can be remembered as:
- A — Appearance (color)
- P — Pulse (heart rate)
- G — Grimace (reflex irritability)
- A — Activity (muscle tone)
- R — Respiration (respiratory effort)
Scoring Table
| Sign | 0 | 1 | 2 |
|---|---|---|---|
| Heart Rate | Absent | < 100 beats/min | ≥ 100 beats/min |
| Respiratory Effort | Absent | Slow, irregular | Strong, crying |
| Muscle Tone | Flaccid/limp | Some flexion | Active motion, good flexion |
| Reflex Irritability (to tactile stimulation) | No response | Grimace | Cough, sneeze, or cry |
| Color | Blue or pale all over | Body pink, extremities blue (acrocyanosis) | Completely pink |
Each criterion is scored 0–2, for a maximum total of 10.
Timing
- Assessed at 1 minute and 5 minutes after birth
- If the 5-minute score is < 7, repeat every 5 minutes up to 20 minutes, continuing resuscitation
Score Interpretation
| Score | Interpretation |
|---|---|
| 7–10 | Normal / reassuring (vigorous neonate) |
| 4–6 | Abnormal; moderate depression |
| 0–3 | Severe depression; poor outcome indicator |
- 8–10: Neonate is vigorous; may need only gentle stimulation
- 4–7 (1-min): Mildly asphyxiated; usually responds to stimulation and supplemental O₂
- 0–3: Severely depressed; requires active resuscitation
Clinical Significance
- The 1-minute score correlates with neonatal survival
- The 5-minute score has a limited (but real) relationship to neurological outcome
- A persistently low score after 5 minutes despite CPR is associated with increased morbidity and mortality
Important Limitations
- Resuscitation must not be delayed to calculate the Apgar score
- A low score (0–3) cannot diagnose perinatal asphyxia in isolation, nor confirm a hypoxic event occurred in utero
- Score can be affected by:
- Gestational age (preterm infants score lower)
- Maternal medications
- Neurological and cardiorespiratory conditions
- Infection, birth trauma
- Ongoing resuscitation efforts
- Cannot be used independently to diagnose perinatal asphyxia (Miller's Anesthesia, 10e)
Expanded Apgar Score
The modern expanded version includes a section to document resuscitative measures being performed at the time of scoring (e.g., supplemental O₂, PPV, intubation, chest compressions, medications), providing context for why a score may be low.
Sources: Textbook of Family Medicine 9e; Morgan and Mikhail's Clinical Anesthesiology 7e; Miller's Anesthesia 10e; Tintinalli's Emergency Medicine; Barash et al. Clinical Anesthesia 9e
HID
HIE in neonates
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Loading Image
Hypoxic-Ischemic Encephalopathy (HIE) in Neonates
Definition
HIE in the term neonate is a neonatal encephalopathy (NE) with a constellation of clinical features and characteristic topography of brain lesions on MRI, resulting from reduced blood flow and oxygen delivery to the brain and excessive accumulation of metabolites — most often mediated by impaired perinatal placental gas exchange.
It applies to neonates ≥ 35 weeks' gestational age.
Pathophysiology
When oxygenated blood supply is limited:
- The fetus initially adapts: increased O₂–hemoglobin binding, preferential shunting to high-risk tissues
- Regions with highest metabolic demand are most vulnerable: Rolandic cortex, thalamus, and basal ganglia
- After the initial insult and reperfusion → transient recovery → then secondary energy failure due to:
- Excitotoxicity
- Apoptosis
- Reactive oxygen species
- Inflammation
This biphasic injury (primary phase → latent phase → secondary phase) is the mechanistic basis for the therapeutic window for cooling.
Etiology / Risk Factors
During pregnancy:
- Gestational diabetes, IUGR, preeclampsia, pregnancy-induced hypertension
During labor & delivery:
- Non-reassuring fetal heart rate tracing
- Placental abruption
- Cord accident (prolapse, nuchal cord)
- Low cord pH / low Apgar scores
- Need for neonatal resuscitation
Diagnosis
ACOG/AAP diagnostic criteria (2014, reaffirmed 2020) require:
- Clinical features of NE (altered consciousness, seizures, abnormal tone/reflexes, respiratory depression) in neonate ≥ 35 weeks
- Neonatal signs consistent with acute peripartum event — at least one of:
- Apgar score < 5 at 5 and 10 minutes
- Umbilical artery pH < 7.0 or base deficit ≥ 12 mmol/L
- Acute brain injury on MRI/MRS consistent with hypoxia-ischemia
- Multisystem organ failure consistent with HIE
- Sentinel hypoxic/ischemic event during labor/delivery (e.g., uterine rupture, abruption, maternal cardiac arrest)
- Developmental outcome: spastic quadriplegia or dyskinetic cerebral palsy
Staging — Sarnat Classification
| Category | Mild (Stage 1) | Moderate (Stage 2) | Severe (Stage 3) |
|---|---|---|---|
| Consciousness | Hyperalert / ↑ | Lethargic / ↓ | Stupor or coma / ↓↓ |
| Spontaneous activity | Normal | ↓ | ↓↓ |
| Tone | Mild distal flexion | ↓ | ↓↓ (flaccid) |
| Muscle stretch reflexes | ↑ | ↓ | ↓↓ |
| Suck reflex | ↓ | ↓ | ↓↓ |
| Moro reflex | ↑ | ↓ | ↓↓ |
| Autonomic system | Sympathetic | Parasympathetic | ↓↓ |
| Clinical seizures | None | ++ (common) | + (uncommon) |
| EEG background | Normal | ↓ | ↓↓ |
| Poor outcome | ~0% | 20–40% | ~100% |
| Duration | < 24 hr | > 24 hr (variable) | > 5 days |
Neuroimaging
MRI is the modality of choice — two predominant injury patterns:
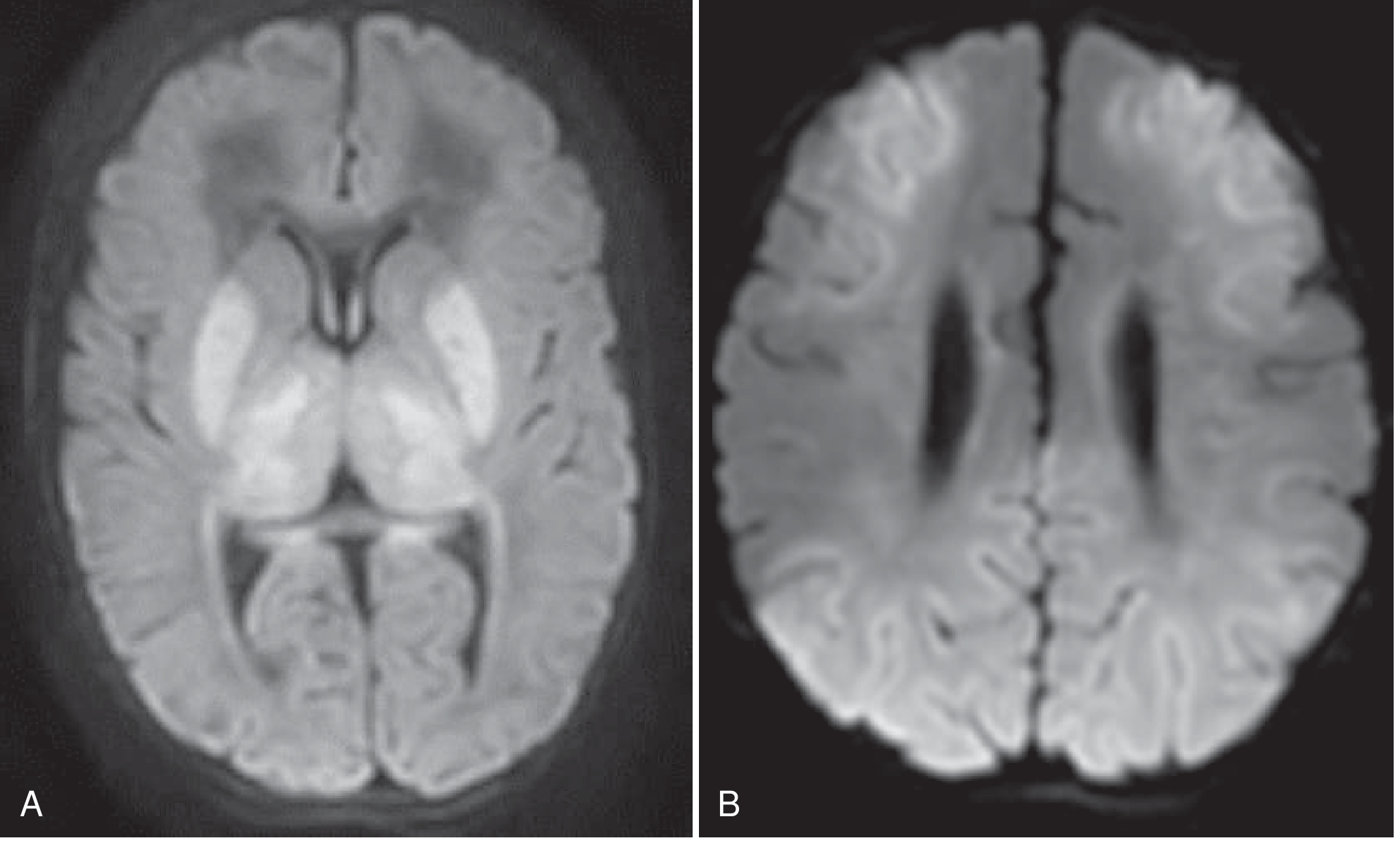
(A) Basal ganglia-thalamus pattern — involves ventrolateral thalami, posterior putamina, perirolandic cortex; typically follows acute sentinel event (e.g., cord prolapse)
(B) Watershed-predominant pattern — anterior-middle and posterior-middle cerebral artery watershed zones; typically follows prolonged partial asphyxia without a clear sentinel event
DWI timing: Restriction peaks within a few days, "pseudonormalizes" by ~1 week. T1/T2 changes begin day 3–4. Optimal MRI timing: day 3–4 (after hypothermia ends).
MR spectroscopy: elevated lactate:N-acetyl aspartate ratio carries prognostic value.
Management
1. Supportive Care
- Resuscitation, airway management
- Correction of metabolic derangements (glucose, electrolytes, acid-base)
- Continuous EEG monitoring + aggressive seizure management
2. Therapeutic Hypothermia (Cooling) ★
The only proven neuroprotective intervention.
Indications (≥ 36 weeks' gestation, within 6 hours of delivery):
- Umbilical cord pH < 7.0 or base excess > 16 mmol/L within 1 hour of delivery, OR
- Moderate to severe encephalopathy on clinical exam
Protocol:
- Target temperature: 33–33.5°C for 72 hours
- Administered as total body cooling or head cooling
- Must be at an experienced comprehensive neonatal center
Efficacy: Meta-analysis of 11 RCTs showed statistically significant reduction in combined mortality or major neurodevelopmental disability at 18 months. NNT = 7.
3. Adjunct / Investigational
- Erythropoietin — under study; may reduce injury
- Melatonin, xenon — investigational neuroprotectants
Prognosis
Despite therapeutic hypothermia, mortality + neurodevelopmental morbidity in moderate-severe HIE remains ~50%.
| Severity | Outcome |
|---|---|
| Mild | Usually no significant neurodevelopmental sequelae |
| Moderate | Unpredictable; resolution by day 5 is a favorable sign |
| Severe | Death or severe impairment: spastic quadriparesis, intellectual disability, cortical visual impairment, epilepsy |
Almost half of neonates with HIE who survive have some neurodevelopmental sequelae.
Sources: Bradley and Daroff's Neurology in Clinical Practice; Creasy & Resnik's Maternal-Fetal Medicine; The Harriet Lane Handbook 23e; Rosen's Emergency Medicine
Congenital torch infections.
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
What can you do? How much free chats do we have?
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.