A 23 year old male complaints of low libido, no morning erection, lack of muscle mass, while has patchy beard and adequate pubic hair growth, had had gynacomastia during teenage years, what is the physiology of testosterone in body? When is it released during day? What is this condition and what is the necessary treatment keeping fertility in consideration?
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching PubMed
Klinefelter syndrome testosterone treatment fertility
Finding Sources
Searching PubMed
Klinefelter syndrome management testosterone
Loading Image
Loading Image
I have all the information needed. Here is the comprehensive answer.
Clinical Analysis: A 23-Year-Old Male with Hypogonadal Features
Part 1 — Physiology of Testosterone
Synthesis and Regulation (The HPG Axis)
Testosterone production is governed by the Hypothalamic-Pituitary-Gonadal (HPG) axis:
- The hypothalamus releases GnRH (gonadotropin-releasing hormone) in a pulsatile fashion.
- GnRH stimulates the anterior pituitary to release LH and FSH.
- LH acts on testicular Leydig cells via cAMP/PKA signaling → stimulates cholesterol conversion to testosterone through a steroidogenic enzyme cascade.
- FSH acts on Sertoli cells → promotes androgen-binding protein (ABP), aromatase, inhibin, and growth factors.
A key mnemonic: LH → Leydig cells (testosterone); FSH → Sertoli cells (spermatogenesis).
Sertoli cells aromatize Leydig-derived testosterone to estradiol, which feeds back negatively on Leydig cells. Inhibin from Sertoli cells specifically suppresses FSH. Testosterone itself feeds back to suppress GnRH and LH.
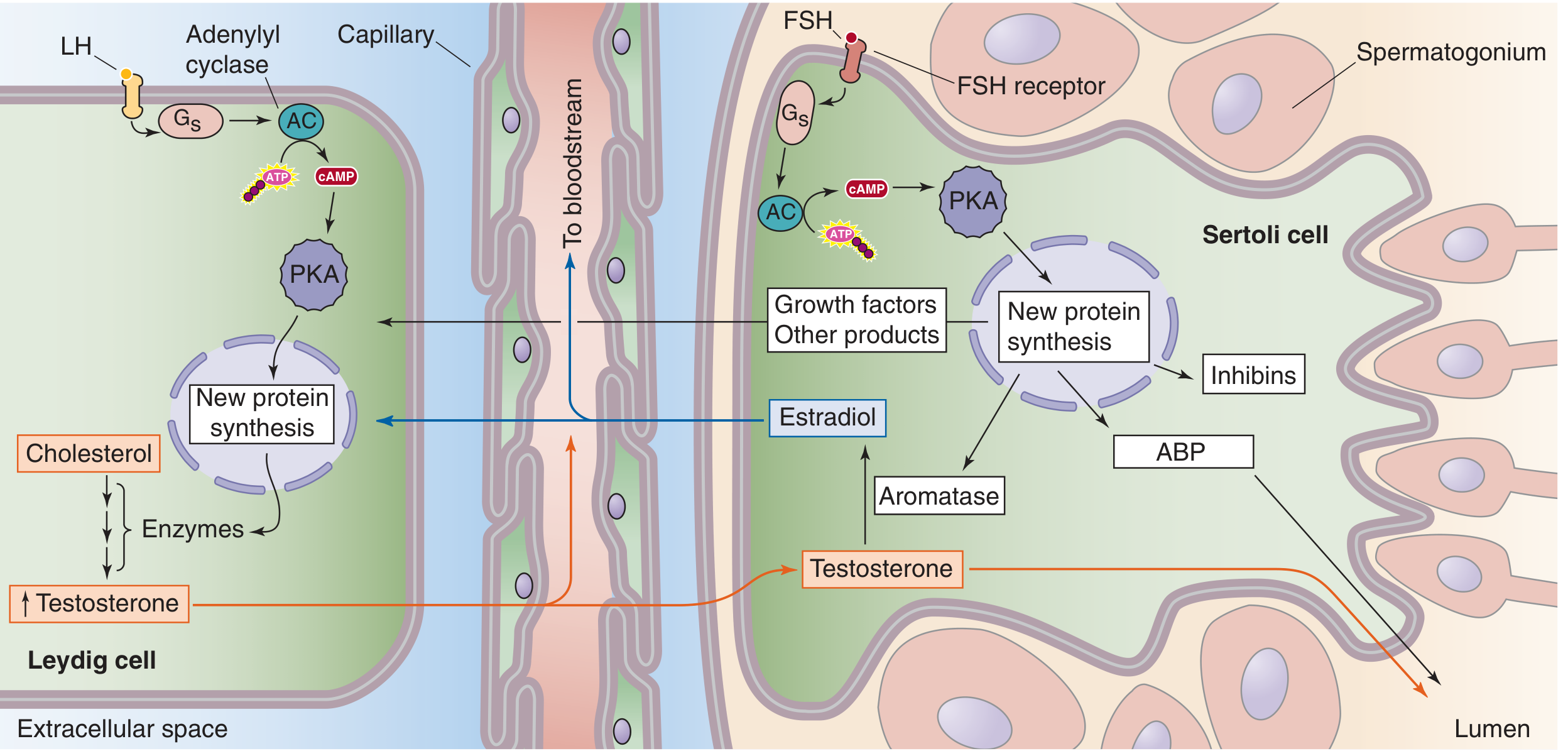
Fig. 54-4 — Leydig and Sertoli cell physiology. Medical Physiology (Boron & Boulpaep), p. 1598
Actions of Testosterone in the Body
| Category | Effect |
|---|---|
| Androgenic | Growth of penis, scrotum, prostate; facial/body/axillary hair; male pattern hair distribution; deepening of voice; sebum production (acne) |
| Anabolic | Skeletal muscle mass and strength; erythropoiesis; bone density and epiphyseal closure |
| CNS | Libido, sexual motivation, morning/nocturnal erections, mood, cognition |
| Metabolic | Insulin sensitivity, fat distribution (prevents central adiposity), lipid metabolism |
| Reproductive | High intratesticular concentration required for spermatogenesis |
In peripheral tissues, testosterone is converted to:
- DHT (dihydrotestosterone) by 5α-reductase — mediates prostate, beard, scalp, external genitalia effects
- Estradiol by aromatase — mediates bone health, sexual function, and feedback
When is Testosterone Released? Diurnal Rhythm
Testosterone secretion in young men follows a strong diurnal (circadian) rhythm:
"Because of the strong diurnal rhythm in testosterone secretion in young men (highest in the morning), testosterone, LH, and FSH should be determined from morning blood samples (before 10 AM)." — Goldman-Cecil Medicine, p. 2539
- Peak: Early morning (approximately 6–8 AM), shortly after waking, driven by the overnight LH pulse surge
- Trough: Evening/night hours
- Normal range: 270–1,100 ng/dL
- This rhythm is robust in young men but blunts with age
- Clinical implication: Blood sampling for testosterone must always be done before 10 AM, or the result will be falsely low
Part 2 — The Diagnosis: Klinefelter Syndrome (47,XXY)
The clinical constellation precisely maps to Klinefelter syndrome (KS):
| Patient's Feature | Explanation in KS |
|---|---|
| Low libido, no morning erections | Low testosterone → reduced CNS androgenic drive |
| Lack of muscle mass | Reduced anabolic testosterone effect |
| Patchy beard (reduced facial hair) | Low DHT/testosterone → inadequate androgen-driven facial hair growth |
| Adequate pubic hair | Pubic/axillary hair is adrenal androgen (DHEA)-dependent, not solely testicular — preserved |
| Gynecomastia in teenage years | Elevated estradiol-to-testosterone ratio during puberty → breast tissue proliferation |
What is Klinefelter Syndrome?
KS is the most common sex chromosome aneuploidy and the most frequent genetic cause of male hypogonadism, with an incidence of ~1 in 660 live male births.
- Classic karyotype: 47,XXY (90% of cases)
- Mosaic form: 46,XY/47,XXY (milder phenotype)
- Results from non-disjunction during meiosis (maternal and paternal equally)
Pathophysiology
The extra X chromosome leads to:
- Seminiferous tubule hyalinization → progressive atrophy and azoospermia
- Leydig cell dysfunction despite their prominent appearance → ↓ testosterone
- Compensatory ↑ LH and FSH (especially FSH — consistently elevated)
- ↑ Estradiol (by an incompletely understood mechanism) → elevated estrogen:androgen ratio → gynecomastia
- The ratio of estrogens to testosterone determines the degree of feminization in individual cases
Clinical Features
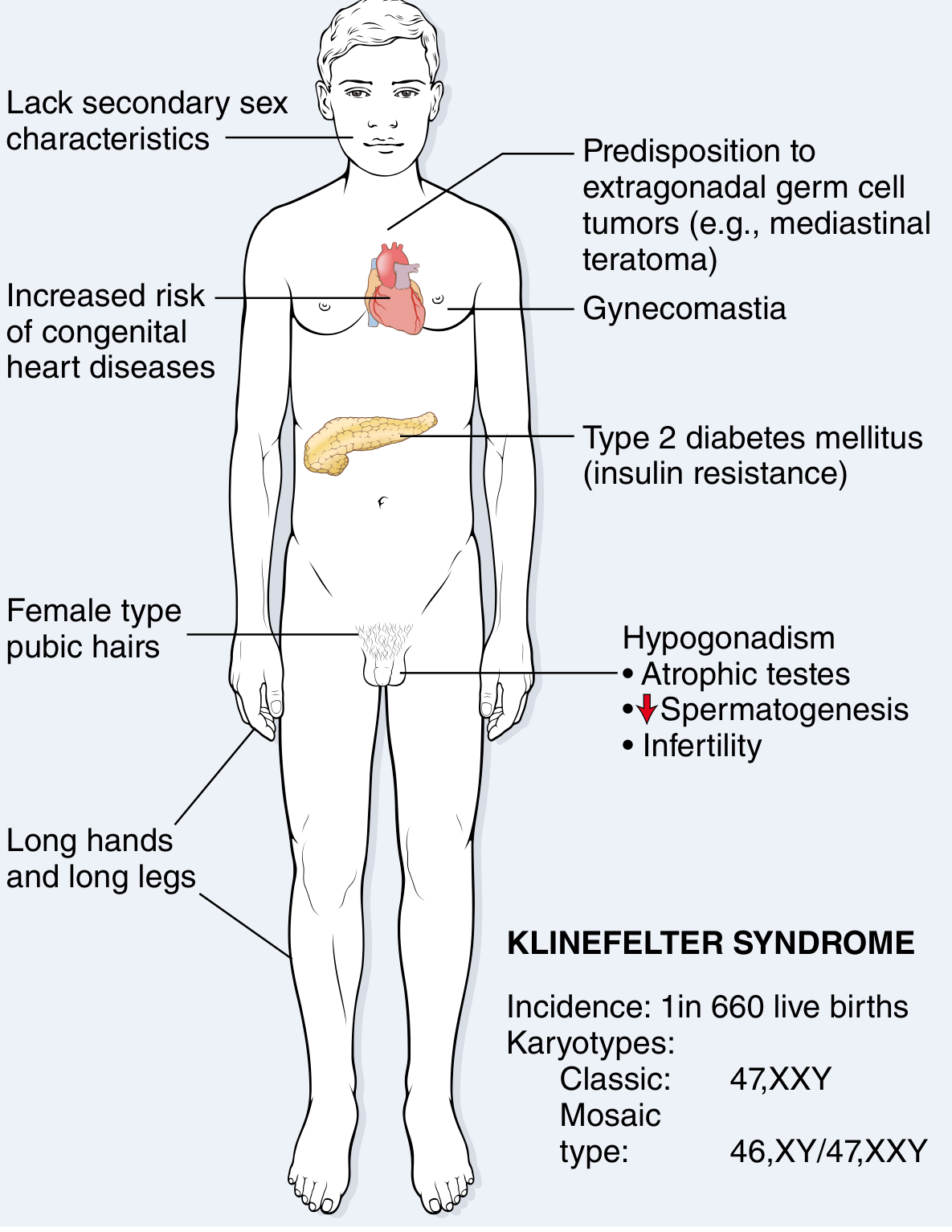
Fig. 5.25 — Clinical features and karyotype of Klinefelter syndrome. Robbins & Kumar Pathologic Basis of Disease, p. 167
- Eunuchoid body habitus: long legs, span > height
- Small atrophic testes (most consistent finding)
- Reduced facial/body hair, sparse beard
- Gynecomastia (variable, often present in teens)
- Cognitive: average to slightly below average; modest verbal skill deficits
- Comorbidities: type 2 diabetes, metabolic syndrome, mitral valve prolapse (~50%), osteoporosis, 20–30× increased risk of mediastinal germ cell tumors, SLE, breast cancer
Investigations
| Test | Expected Finding in KS |
|---|---|
| Morning serum testosterone | Low |
| LH | Elevated (primary hypogonadism pattern) |
| FSH | Consistently and markedly elevated |
| Estradiol | Elevated |
| Karyotype (confirmatory) | 47,XXY |
| Semen analysis | Azoospermia (most cases) |
Part 3 — Treatment (with Fertility in Mind)
⚠️ Critical Distinction: Testosterone Replacement vs. Fertility-Preserving Treatment
Exogenous testosterone replacement (TRT) suppresses the HPG axis, further reducing intratesticular testosterone and eliminating any residual spermatogenesis. In a 23-year-old who may wish to father children, TRT must not be started without addressing fertility first or offered concurrently with sperm banking.
A. Fertility Preservation — FIRST PRIORITY in Young Men
Testicular Sperm Extraction (microTESE/TESE):
- Despite azoospermia on semen analysis, ~50–70% of KS men have focal residual spermatogenesis within testicular tubules
- Microsurgical TESE (microTESE) can retrieve sperm for use with ICSI (intracytoplasmic sperm injection)
- Should be performed before starting TRT (or after a washout period), as exogenous testosterone suppresses residual spermatogenesis
- Sperm can be cryopreserved for future use
- Success rates are higher in younger patients (tissue less fibrotic)
Pre-TESE hormonal optimization (off-label):
- Clomiphene citrate (SERM): Blocks estrogen feedback at the hypothalamus → ↑ GnRH → ↑ LH/FSH → ↑ endogenous testosterone without axis suppression. Starting dose: 25 mg/day or 50 mg every other day, titrated to testosterone levels. Can improve sperm retrieval rates prior to TESE.
- Aromatase inhibitors (anastrozole 1 mg/day, letrozole): Reduce estradiol → improves testosterone:estradiol ratio → stimulates gonadotropin release. Particularly useful in KS given elevated estradiol.
- hCG (human chorionic gonadotropin): LH analog, stimulates Leydig cells directly. Used 1,500–5,000 IU IM/SC 2–3×/week. Raises testosterone without suppressing FSH (which is needed for spermatogenesis).
B. Androgen Replacement Therapy — After Fertility Goals Are Addressed
Once fertility is no longer an immediate concern (or sperm have been banked), testosterone replacement is the mainstay:
Indications: Symptomatic hypogonadism — low libido, absent morning erections, poor muscle mass, fatigue, low bone density
Formulations:
| Route | Preparation | Notes |
|---|---|---|
| Intramuscular | Testosterone enanthate or cypionate (100–200 mg every 2 weeks) | Most widely used; produces peaks and troughs |
| Transdermal | Gel (1–1.62%) or patches | Mimics diurnal rhythm better; avoid skin-to-skin transfer |
| Injectable long-acting | Testosterone undecanoate (1000 mg IM every 10–14 weeks) | Stable levels, less frequent dosing |
Targets: Morning serum testosterone 400–700 ng/dL; monitor LH, FSH, hematocrit, PSA (prostate), lipid profile, and bone density.
Benefits of TRT in KS:
- Restores libido and sexual function
- Improves muscle mass and bone density (reduces fracture risk)
- Reduces fatigue; improves mood and cognitive performance
- May reduce gynecomastia (especially if started early)
Note on gynecomastia: If prominent and persistent, surgical referral (mastectomy/reduction) may be needed alongside hormonal correction.
Summary Decision Framework for This 23-Year-Old
Age 23 with KS
│
├── Fertility desired now or in future?
│ │
│ YES → Karyotype confirm → microTESE + sperm banking
│ Pre-TESE: clomiphene/anastrozole/hCG to optimize
│ AVOID TRT until sperm retrieved/banked
│
│ NO or done → Start testosterone replacement
│ (IM, gel, or long-acting injection)
│
└── Monitor: testosterone levels, hematocrit, lipids,
bone density, metabolic parameters, mood
Sources
- Robbins & Kumar Pathologic Basis of Disease, p. 167 — Klinefelter syndrome pathology and features
- Goldman-Cecil Medicine, p. 2539 — Laboratory diagnosis, diurnal rhythm, testosterone thresholds
- Medical Physiology (Boron & Boulpaep), p. 1598 — Leydig/Sertoli cell physiology
- Campbell-Walsh Urology, p. 1917 — Gonadotropin therapy and clomiphene for male infertility
- Chen X et al. (2024). Klinefelter syndrome: etiology and clinical considerations in male infertility. Biol Reprod. PMID: 38785325
- Blackburn J et al. (2025). Klinefelter Syndrome: A Review. Clin Endocrinol (Oxf). PMID: 39806878
This is a shared conversation. Sign in to Orris to start your own chat.