Multiple sclerosis in detail
Reading File
Finding Sources
Searching PubMed
"multiple sclerosis"[MeSH Terms] AND treatment
Searching the Web
multiple sclerosis 2025 updated guidelines diagnosis treatment
Finding Sources
Searching Images
multiple sclerosis MRI lesions white matter plaques
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
I now have comprehensive content covering epidemiology, genetics, pathology, immunopathogenesis, and treatment. I have enough to compose a thorough, well-structured answer. Let me now compile the full response.
Multiple Sclerosis — A Comprehensive Overview
Bradley and Daroff's Neurology in Clinical Practice; Textbook of Family Medicine 9e
1. Definition & Overview
Multiple sclerosis (MS) is a chronic, immune-mediated demyelinating disease of the central nervous system (CNS) characterized by neurological deficits disseminated in time and space. It is the leading non-traumatic cause of neurological disability in young adults in North America and Europe. The disease is heterogeneous in its clinical course, pathology, and response to treatment.
2. Epidemiology
| Parameter | Data |
|---|---|
| Prevalence | ~265 per 100,000 (Ontario, 2013); rising globally |
| Sex ratio | Female:Male = 2:1 (RRMS); closer to 1:1 in PPMS |
| Age of onset | RRMS: early 20s; PPMS: mid-30s |
| Ethnicity | Most prevalent in Northern European descent; lower in Arabic and Mediterranean populations |
| Geography | North-to-south gradient in Northern Hemisphere (inverse in Southern Hemisphere) |
| Mortality | Average time to death from MS onset: 24–45 years; SMR 1.3–2.9 vs. general population |
MS mortality has declined 33% in Western countries over recent decades, largely due to effective disease-modifying therapies. ~50% of patients with MS die of disease-related complications (infections being most common).
3. Disease Course / Clinical Subtypes
Four recognized phenotypes:
- Relapsing-Remitting MS (RRMS) — affects ~65% of patients. Discrete neurological attacks with complete or partial recovery. Most convert to SPMS after 15–20 years.
- Secondary Progressive MS (SPMS) — follows RRMS; neurological deficits become fixed and accumulate progressively.
- Primary Progressive MS (PPMS) — ~10–15% of patients; progressive accumulation from onset without distinct relapses.
- Progressive Relapsing MS (PRMS) — progressive course with superimposed acute relapses.
4. Genetics
MS is a complex polygenic disease:
- Monozygotic twin concordance: 27% (vs. 2.3% in dizygotic twins of same sex)
- First-degree relatives: 2–5% risk (vs. <0.1% general population)
- Strongest genetic association: HLA-DR2 (DRB1*1501) haplotype
- GWAS studies identified 110 risk variants at 103 loci outside the MHC, including polymorphisms in IL-2R and IL-7R alleles
- Other HLA associations (DR4, DR6) noted in Arabic/Mediterranean patients
5. Environmental & Lifestyle Risk Factors
| Factor | Association |
|---|---|
| Latitude / sunlight | Diminishing north-to-south prevalence gradient; inverse relation with UV exposure |
| Vitamin D | Low 25-OH-D levels associated with increased risk (OR 0.38 for high quintile) |
| EBV infection | Strong environmental risk factor; possibly via molecular mimicry with myelin antigens |
| Smoking | Increased risk (particularly in women) |
| Childhood obesity | Overweight/obesity at age 18–20 doubles MS risk |
| Migration | Migration to high-risk areas before age 15 increases risk |
Vitamin D appears to have an immunomodulatory role predominantly in women, mediated through functional synergy with 17β-estradiol via estrogen receptor α. However, high-dose vitamin D treatment has not been shown to significantly reduce relapse risk in meta-analyses.
6. Pathology
Lesions (plaques) are sclerotic areas of demyelination found primarily in:
- Periventricular white matter
- Corpus callosum
- Optic nerves
- Brainstem and cerebellum
- Spinal cord
- Cortex (cortical demyelination — increasingly recognized in progressive MS)
Inflammation is perivenular. Lucchinetti et al. classified MS lesions into four histopathological subtypes based on predominant features:
- Cellular infiltration
- Antibody deposition
- Oligodendrocyte apoptosis
- Oligodendrocyte death without apoptosis
A single subtype appears to predominate in each individual patient, suggesting distinct pathogenetic mechanisms across patients.
Axonal transection occurs early (even in acute plaques) and underlies irreversible disability. Activated microglia are present in normal-appearing white matter (NAWM) and contribute to progressive axonal damage. B-cell follicles in meninges, cortical demyelination, and epitope spreading within the CNS are hallmarks of progressive disease.
7. Immunopathogenesis
The pathogenesis involves a multi-step immunological cascade:
- Peripheral sensitization: Myelin-specific T cells (predominantly TH1 phenotype) are activated — likely via molecular mimicry with viral antigens (EBV, HHV-6, coronavirus, influenza) or superantigens
- BBB transmigration: T cells upregulate adhesion molecules (VCAM-1/VLA-4) to cross the blood-brain barrier
- CNS reactivation: Perivascular dendritic cells present myelin antigens (MBP, PLP, MOG) to T cells within the CNS
- Cytokine cascade: IL-2, IFN-γ, TNF-α activate macrophages → nitric oxide, lipid peroxidation, complement-induced pore formation → myelin destruction
- TH17 cells also contribute — transfer of TH17 cells in animal models induces more severe disease than TH1 alone
- B-cell involvement: Oligoclonal bands are present in >95% of MS patients; antibodies to myelin epitopes (though no specific autoantigen confirmed — hence MS is called immune-mediated rather than strictly autoimmune)
The TH1/TH2 balance is critical: TH1 promotes inflammation; TH2 (IL-4, IL-10) is protective. Disease progression relies more on microglia, B-cell follicles, and cortical demyelination than on peripheral T-cell infiltration.
8. Clinical Features
Typical presenting syndromes:
- Optic neuritis — painful monocular visual loss, afferent pupillary defect; often the first symptom
- Internuclear ophthalmoplegia (INO) — adduction failure ipsilateral, nystagmus contralateral; due to MLF demyelination
- Transverse myelitis — para/tetraparesis, sensory level, bladder/bowel dysfunction
- Lhermitte's sign — electric shock-like sensation down spine on neck flexion (cervical cord lesion)
- Uhthoff's phenomenon — symptom worsening with heat/exercise (due to heat-sensitive conduction in demyelinated axons)
- Cerebellar features — ataxia, intention tremor, scanning speech (Charcot's triad with nystagmus and intention tremor)
- Fatigue — one of the most disabling symptoms
- Cognitive dysfunction — processing speed, attention, memory
- Bladder dysfunction — urgency, frequency, incontinence (neurogenic bladder)
- Depression and psychiatric symptoms
9. Diagnosis — McDonald Criteria (2017, updated 2024)
Diagnosis requires demonstration of dissemination in space (DIS) and dissemination in time (DIT), plus exclusion of alternative diagnoses.
MRI criteria for DIS — lesions in ≥2 of 4 CNS regions:
- Periventricular
- Juxtacortical/cortical
- Infratentorial
- Spinal cord
MRI criteria for DIT:
- New T2 or gadolinium-enhancing lesion on follow-up MRI
- Simultaneous presence of both gadolinium-enhancing and non-enhancing lesions
Updated 2024 McDonald criteria incorporate novel biomarkers:
- Central vein sign (CVS) — veins running through MS lesion center (distinguishes MS from other white matter disease)
- Kappa free light chain (KFLC) in CSF as a substitute for oligoclonal bands
CSF findings:
- Oligoclonal bands (IgG) in >95% of patients
- Elevated IgG index
- Mild pleocytosis (lymphocytes)
- Normal glucose
Evoked potentials (especially visual evoked potentials) detect subclinical demyelination.
10. MRI Findings
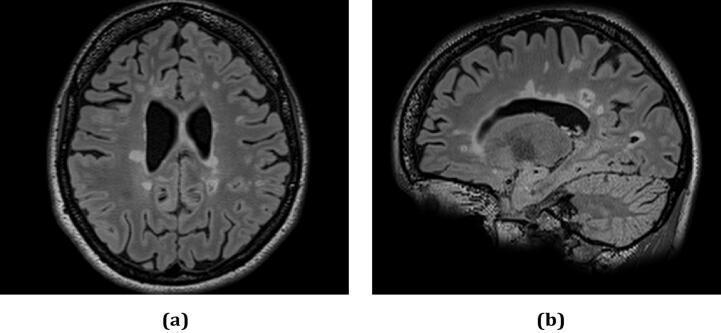
Axial and sagittal FLAIR MRI showing periventricular plaques and "Dawson's fingers" — ovoid hyperintensities perpendicular to the lateral ventricles along medullary veins.
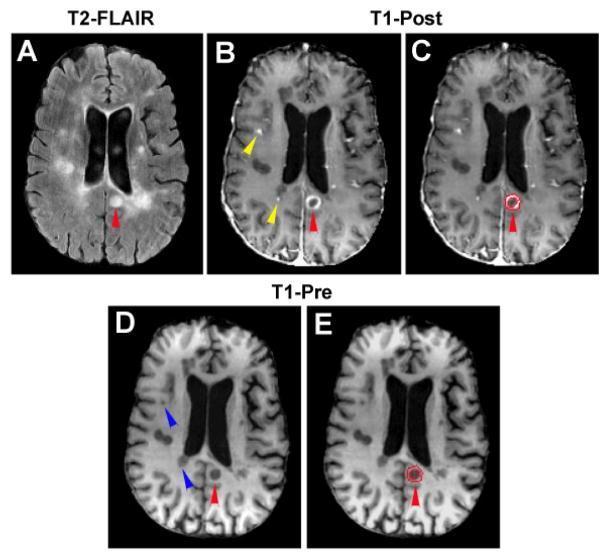
Multi-panel MRI: T2-FLAIR periventricular plaques (A), gadolinium ring-enhancing active lesion (B–C), and T1 hypointense "black holes" indicating axonal loss (D–E).
Key MRI features:
- T2/FLAIR: Hyperintense plaques — periventricular (ovoid, finger-like = "Dawson's fingers"), juxtacortical, infratentorial, spinal cord
- T1 with Gd: Active (enhancing) plaques indicate acute BBB breakdown and inflammation
- T1 hypointensities ("black holes"): Areas of severe axonal loss and irreversible tissue damage
- Spinal cord: Typically short (≤2 vertebral segments), eccentric lesions (cf. NMOSD: longitudinally extensive ≥3 segments)
11. Differential Diagnosis
Important conditions to exclude:
| Condition | Key Distinguishing Features |
|---|---|
| NMOSD (Devic disease) | AQP4-IgG+; longitudinally extensive spinal cord lesions (≥3 segments); area postrema lesions; different treatment |
| MOGAD | MOG-IgG+; often cortical encephalitis, bilateral optic neuritis; more common in children |
| ADEM | Monophasic; post-infectious/vaccination; bilateral, diffuse lesions; no oligoclonal bands (usually) |
| CNS vasculitis | Stroke-like episodes; meningeal enhancement; angiography abnormal |
| Neurosarcoidosis | Systemic sarcoidosis; leptomeningeal enhancement |
| B12 deficiency | Subacute combined degeneration; dorsal column + corticospinal tract |
| Antiphospholipid syndrome | Thrombotic history; anticardiolipin/β2-GP1 antibodies |
12. Treatment
A. Acute Relapse Management
- IV methylprednisolone 1 g/day × 3–5 days — speeds recovery but does not alter long-term disability
- Oral high-dose steroids are equivalent for mild-moderate relapses
- Plasma exchange for steroid-refractory severe relapses
B. Disease-Modifying Therapies (DMTs)
Currently ~20 approved agents, categorized by mechanism and potency:
Platform / Moderate Efficacy
| Drug | Route | Mechanism |
|---|---|---|
| IFN-β-1b (Betaseron, Extavia) | SC | ↑IL-10, ↓IL-12, ↓T-cell adhesion/migration via VCAM-1 modulation |
| IFN-β-1a (Avonex) | IM | Same class |
| IFN-β-1a (Rebif) | SC | Same class |
| Glatiramer acetate (Copaxone) | SC | MBP mimic; TH2 bias induction; neuroprotection |
| Teriflunomide (Aubagio) | Oral | Pyrimidine synthesis inhibitor → ↓T/B-cell proliferation |
| Dimethyl fumarate (Tecfidera) | Oral | Nrf2 pathway activation; anti-inflammatory + neuroprotective |
High Efficacy
| Drug | Route | Mechanism |
|---|---|---|
| Natalizumab (Tysabri) | IV infusion | Anti-VLA-4; blocks T-cell entry across BBB |
| Ocrelizumab (Ocrevus) | IV infusion | Anti-CD20; B-cell depletion; approved for RRMS and PPMS |
| Alemtuzumab (Lemtrada) | IV infusion | Anti-CD52; T and B-cell depletion |
| Cladribine (Mavenclad) | Oral | DNA alkylation; selective lymphocyte depletion |
| Ofatumumab (Kesimpta) | SC | Anti-CD20; self-administered |
Natalizumab mechanism detail:
VLA-4 (α4β1 integrin) on T cells binds VCAM-1 on activated brain endothelium; natalizumab (anti-VLA-4) prevents this interaction, dramatically reducing CNS lymphocyte trafficking. Risk: PML (progressive multifocal leukoencephalopathy) from JC virus reactivation, especially with JCV antibody index >1.5 and prior immunosuppressant use.
C. Progressive MS
- Ocrelizumab (first approved for PPMS — only DMT with proven benefit in primary progressive disease)
- Siponimod (Mayzent) — approved for active SPMS
- Ozanimod — S1P receptor modulator; approved for RRMS; under study in SPMS
- Cochrane 2024 network meta-analysis (PMID: 39254048) found ocrelizumab and cladribine had best evidence for reducing disability progression in progressive MS
D. Symptom Management
| Symptom | Treatment |
|---|---|
| Spasticity | Baclofen (oral/intrathecal), tizanidine, cannabinoids |
| Fatigue | Amantadine, modafinil, lifestyle modification |
| Bladder | Oxybutynin, solifenacin, intermittent self-catheterization |
| Neuropathic pain | Gabapentin, pregabalin, amitriptyline |
| Depression | SSRIs, psychotherapy |
| Tremor | Clonazepam, propranolol; DBS for refractory cases |
| Cognitive rehab | Virtual reality-based rehabilitation (Cochrane 2025, PMID: 39775922) |
13. Prognosis & Disability
- EDSS (Expanded Disability Status Scale) 0–10 is the standard outcome measure
- ~50% of RRMS patients need a walking aid within 15 years of onset
- Poor prognostic factors: male sex, progressive onset, motor/cerebellar symptoms at onset, high MRI lesion burden, black holes, age >40 at onset
- Favorable factors: sensory onset, complete recovery after first relapse, low MRI burden, female sex, younger onset
14. Special Situations
Pregnancy: MS relapse rate decreases during the third trimester but rebounds sharply in the first 3 months postpartum. Most DMTs are contraindicated in pregnancy (natalizumab may be used cautiously).
Tumefactive MS: Large (>2 cm) lesions with mass effect and edema, mimicking brain tumors; often requires biopsy for diagnosis.
MS misdiagnosis is common (~20% of patients initially diagnosed with MS may have an alternative diagnosis — NMOSD, MOGAD, migraine with white matter changes, small vessel disease).
15. Emerging Concepts (2025)
- 2024 McDonald Criteria updates: Central vein sign and KFLC now incorporated; improved early diagnosis
- CMSC 2025 guidelines updated best practices in DMT selection including escalation vs. high-efficacy first strategies
- Growing interest in neuroprotective and remyelination strategies (anti-LINGO-1, opicinumab)
- Gut microbiome alterations linked to MS susceptibility and relapse
- Bruton's tyrosine kinase (BTK) inhibitors (e.g., tolebrutinib) in late-stage trials for both relapsing and progressive MS, targeting CNS-resident microglia
This is a shared conversation. Sign in to Orris to start your own chat.