What is Gullain barre syndrome
Reading File
Finding Sources
Searching PubMed
"Guillain-Barre syndrome"[MeSH Terms]
Reading File
Reading File
Searching Images
Guillain Barre syndrome nerve pathology demyelination
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information to provide a thorough answer. Here is a full overview of Guillain-Barré Syndrome.
Guillain-Barré Syndrome (GBS)
Definition
Guillain-Barré syndrome is a rapidly progressive, acute immune-mediated polyneuropathy affecting the peripheral nervous system — most notably the motor axons and nerve roots — resulting in ascending weakness. It is one of the most common life-threatening diseases of the peripheral nervous system, occurring at a rate of 0.81–1.89 cases per 100,000 persons per year worldwide. — Miller's Anesthesia, 10e
Historical Background
First described in 1916 by Guillain, Barré, and Strohl, who identified the cardinal features: motor weakness, areflexia, paresthesias with minor sensory loss, and elevated CSF protein without pleocytosis (the classic albuminocytological dissociation). Before the introduction of positive-pressure ventilation, mortality reached 33%; modern critical care has reduced this to approximately 1–5%. — Bradley and Daroff's Neurology in Clinical Practice
Pathophysiology
About two-thirds of GBS cases are triggered by a preceding infection, which generates microbe-specific T cells and antibodies that cross-react with antigens in the nerve sheath — a process called molecular mimicry.
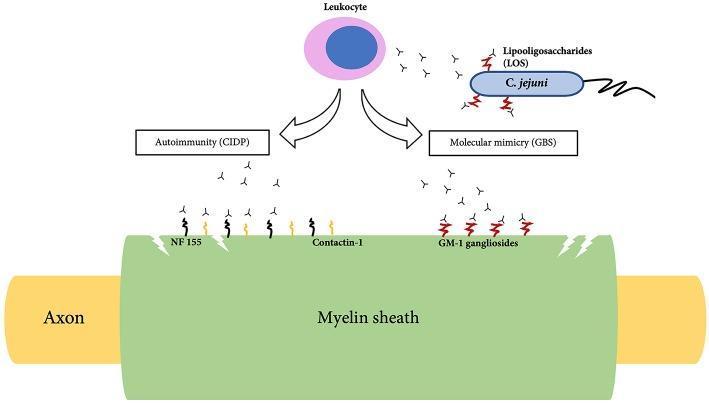
The diagram shows how C. jejuni lipooligosaccharides (LOS) trigger antibodies that cross-react with GM-1 gangliosides on the myelin sheath — the molecular mimicry mechanism of GBS.
- Both T-cell-mediated and antibody-mediated (humoral) responses are involved, with T cells playing the dominant role in the most common subtype.
- Injury is most extensive in the nerve roots and proximal nerve segments, associated with mononuclear cell infiltrates rich in macrophages.
Common triggers:
| Category | Examples |
|---|---|
| Bacterial | Campylobacter jejuni (most frequent) |
| Viral | Cytomegalovirus, Epstein-Barr virus, HIV, Zika virus, Chikungunya, SARS-CoV-2 |
| Non-infectious | Vaccinations, immune checkpoint inhibitors |
Clinical Features & Subtypes
The syndrome is heterogeneous. Subtypes include:
| Subtype | Key Features |
|---|---|
| AIDP (Acute Inflammatory Demyelinating Polyradiculoneuropathy) | Most common in Europe & North America; ascending weakness, demyelinating |
| AMAN (Acute Motor Axonal Neuropathy) | Pure motor; associated with C. jejuni; GM-GD1a antibodies; common in China |
| AMSAN (Acute Motor-Sensory Axonal Neuropathy) | Both motor and sensory; severe; poor recovery |
| Miller-Fisher Syndrome (MFS) | Triad: ophthalmoplegia + ataxia + areflexia; anti-GQ1b antibodies; 6% of cases in the West |
Classic presentation:
- Symmetric ascending muscle weakness starting in the lower limbs
- Areflexia or hyporeflexia
- Mild sensory symptoms/paresthesias
- Bifacial palsies (cranial nerve involvement)
- Autonomic dysfunction in ~20% (fluctuating BP, arrhythmias)
- Absence of fever at onset
Symptoms progress over days to a maximum of 4 weeks, plateau, then gradually improve. In ~5% of patients, maximum deficit occurs within 72 hours.
Diagnostic Criteria
Required:
- Progressive weakness of both legs and arms
- Areflexia or hyporeflexia
Laboratory support:
- CSF: Elevated protein with <10 cells/μL (albuminocytological dissociation)
- Nerve conduction studies (NCS): Slowing of conduction velocities or conduction block
- Antibodies: Anti-GQ1b (MFS), anti-GM1/GD1a (AMAN)
— Bradley and Daroff's Neurology in Clinical Practice, Box 106.10
MRI finding (cauda equina enhancement):
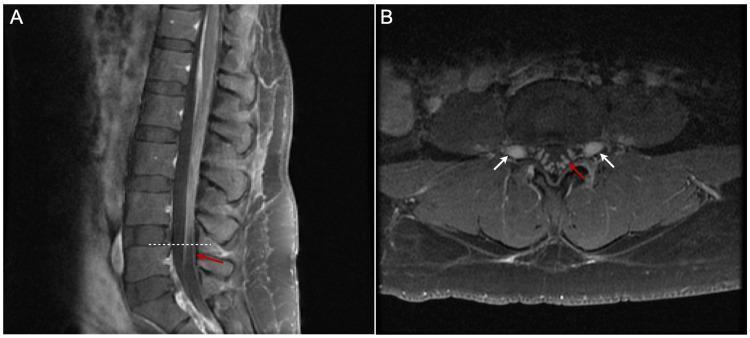
Post-contrast T1 MRI of the lumbar spine showing smooth thickening and linear enhancement of cauda equina nerve roots — a classic radiological sign in GBS reflecting breakdown of the blood-nerve barrier.
Complications
- Respiratory failure in 20–30% of cases — the major reason for ICU admission
- Mortality increases to 14.3% with need for mechanical ventilation
- Dysautonomia in up to 20% (cardiac arrhythmia, labile BP)
- Prolonged recovery (months to years in axonal subtypes)
Respiratory monitoring thresholds:
- Forced vital capacity (FVC) <20 mL/kg → close observation
- FVC <15 mL/kg → probable intubation needed
- Maximal inspiratory pressure (MIP) < −30 cmH₂O → consider intubation
- The EGRIS score (Erasmus GBS Respiratory Insufficiency Score) helps predict need for early ICU admission
Treatment
| Treatment | Mechanism |
|---|---|
| IV Immunoglobulin (IVIg) | Suppresses immune responses (mechanism not fully clear) |
| Plasmapheresis (plasma exchange) | Removes offending antibodies from circulation |
| Supportive care | Ventilatory support, DVT prophylaxis, analgesia, autonomic monitoring |
| Corticosteroids | Not effective — not recommended in GBS |
Both IVIg and plasmapheresis are equivalently effective; they are not combined as there is no added benefit. — Robbins & Kumar Basic Pathology; Miller's Anesthesia, 10e
Prognosis: Most patients who survive the acute phase recover with time, though axonal subtypes (AMSAN) have poorer recovery. Patients with GM1 antibodies also tend to recover less well.
Key Differentiator from CIDP
GBS and Chronic Inflammatory Demyelinating Polyneuropathy (CIDP) share an immune-mediated demyelinating mechanism, but GBS is acute (plateaus by 4 weeks), while CIDP follows a chronic or relapsing-remitting course (progresses ≥2 months). — Robbins & Kumar Basic Pathology
This is a shared conversation. Sign in to Orris to start your own chat.