Discuss the Pharmacology and drug treatment of arrhythmia
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching PubMed
antiarrhythmic drugs classification management
I now have comprehensive content from multiple authoritative textbooks. Let me compile the full response.
Pharmacology and Drug Treatment of Arrhythmias
1. Overview
Cardiac arrhythmias range from incidental, asymptomatic findings to life-threatening conditions. Antiarrhythmic drugs suppress arrhythmias by modulating ion channel flow or altering autonomic function. They have two primary goals: termination of an ongoing arrhythmia or prevention of arrhythmia recurrence. Critically, these drugs can also cause arrhythmias (proarrhythmia), so therapy must always be preceded by identification of precipitating factors, precise diagnosis, and risk-benefit analysis.
— Goodman & Gilman's The Pharmacological Basis of Therapeutics
2. Mechanisms of Arrhythmogenesis
Arrhythmias arise from three fundamental mechanisms:
- Abnormal automaticity – Enhanced or suppressed spontaneous pacemaker discharge (e.g., accelerated junctional rhythm, sick sinus syndrome)
- Triggered activity – Afterdepolarizations, either:
- Early afterdepolarizations (EADs): occur during Phase 2/3 of the action potential; linked to QT prolongation and torsades de pointes
- Delayed afterdepolarizations (DADs): occur after full repolarization; linked to digitalis toxicity, catecholamine excess
- Reentry – The most common mechanism; requires a unidirectional block and a pathway permitting slow conduction to re-excite previously depolarized tissue (e.g., atrial flutter, AV nodal reentrant tachycardia, WPW syndrome)
3. Vaughan-Williams Classification
The Vaughan-Williams system groups antiarrhythmic drugs by their predominant effects on the cardiac action potential. Note that many drugs have multi-class effects.
| Class | Mechanism | Key Drugs |
|---|---|---|
| IA | Na⁺ channel block (intermediate kinetics); ↑ APD & QT | Quinidine, Procainamide, Disopyramide |
| IB | Na⁺ channel block (fast kinetics); ↓ or no change in APD | Lidocaine, Mexiletine |
| IC | Na⁺ channel block (slow kinetics); no change in APD | Flecainide, Propafenone |
| II | β-adrenergic receptor blockade; ↓ Phase 4 automaticity | Atenolol, Metoprolol, Esmolol |
| III | K⁺ channel block; ↑ APD & QT; ↑ refractoriness | Amiodarone, Sotalol, Dofetilide, Ibutilide, Dronedarone |
| IV | Ca²⁺ channel block (non-DHP); ↓ AV nodal conduction | Verapamil, Diltiazem |
| Other | Multiple / unique mechanisms | Adenosine, Digoxin, Magnesium |
— Lippincott Illustrated Reviews: Pharmacology; Katzung's Basic and Clinical Pharmacology, 16th Ed.
4. Class-by-Class Drug Details
Class I — Sodium Channel Blockers
Na⁺ channel blockers bind to open or inactivated channels, causing state-dependent block. The critical parameter is the time constant of recovery (τ_recovery):
- Short τ: drug dissociates quickly (Class IB — selective for rapidly firing/diseased tissue)
- Intermediate τ: Class IA
- Long τ: Class IC — accumulates at high heart rates, dangerous in ischemic myocardium
Class IA
Quinidine
- Blocks Na⁺ channels + outward K⁺ channels (IKr), prolonging APD and QT
- Vagolytic (can increase AV nodal conduction and paradoxically accelerate ventricular rate in atrial flutter)
- Adverse effects: Cinchonism (tinnitus, visual disturbances, headache), hemolytic anemia, QT prolongation → torsades de pointes, esophagitis
Procainamide
- Analogue of procaine; blocks open Na⁺ channels (intermediate τ); also blocks K⁺ channels
- Active metabolite: N-acetyl procainamide (NAPA) — prolongs APD but lacks Na⁺ channel block
- Short t½ (~3–4 h) requires slow-release formulation; NAPA accumulates in renal failure
- Major adverse effect: Drug-induced lupus syndrome (antinuclear antibodies in nearly all patients with long-term use; 25–50% develop symptoms). Also: bone marrow aplasia (0.2%), hypotension, torsades
- Acetylator phenotype determines lupus risk: slow acetylators develop lupus earlier
Disopyramide
- Similar to procainamide but with significant antimuscarinic effects (urinary retention, dry mouth, constipation, glaucoma precipitation)
- Concentration-dependent protein binding; negative inotrope — can precipitate acute heart failure
- Eliminated via hepatic and renal routes
Class IB
Lidocaine
- Very fast τ_recovery; selectively blocks activated and inactivated channels at rapid rates and depolarized potentials (ischemic tissue)
- Only available IV; extensive hepatic first-pass metabolism
- Dose-dependent CNS toxicity: perioral numbness → tremor → seizures → coma
- Reduce dose in heart failure and liver disease
Mexiletine
- Orally active congener of lidocaine; shortens Phase 3 repolarization
- Used in ventricular arrhythmias and some channelopathies (Long QT syndrome Type 3)
- GI adverse effects (nausea, dyspepsia) are common
Class IC
Flecainide
- Markedly slows Phase 0; very slow dissociation from Na⁺ channels — effect is rate-dependent (worse at fast rates)
- Effective for supraventricular arrhythmias, paroxysmal AF in structurally normal hearts
- Contraindicated post-MI and in ischemic heart disease — the CAST trial showed 2-3× increase in mortality
- Oral; hepatic and renal metabolism; t½ ~20 h
Propafenone
- Similar to flecainide + weak β-blocking activity
- Hepatically metabolized; extensive polymorphism (poor vs. extensive metabolizers via CYP2D6)
- Adverse: bronchospasm (due to β-block), proarrhythmia, rare agranulocytosis
The CAST lesson: Suppressing ventricular ectopy with Na⁺ channel blockers post-MI increases mortality. Treating an asymptomatic arrhythmia marker is not the same as treating the patient. Among all antiarrhythmic drugs, only β-blockers demonstrably reduce mortality in long-term therapy. — Goodman & Gilman's
Class II — β-Adrenergic Receptor Blockers
Mechanism: Block β₁ receptors → decrease Phase 4 slope → reduce SA node automaticity and AV nodal conduction velocity → lengthen PR interval and AV nodal refractory period.
Key agents: Atenolol, Metoprolol, Esmolol (ultra-short-acting IV, t½ ~9 min — ideal for acute rate control)
Clinical uses:
- Rate control in atrial fibrillation/flutter
- Post-MI sudden death prevention
- Suppression of exercise-induced and catecholamine-mediated arrhythmias
- Ventricular arrhythmias in structural heart disease (reduced sudden cardiac death)
Adverse effects: Bradycardia, heart block, acute heart failure exacerbation, bronchospasm, hypotension, fatigue, sexual dysfunction
Class III — Potassium Channel Blockers
Block IKr (and other K⁺ currents) → prolong APD and QT interval → increase effective refractory period → terminate reentrant circuits. The key risk is torsades de pointes (reverse use-dependence: effect greatest at slow rates).
Amiodarone — the most important drug in this class
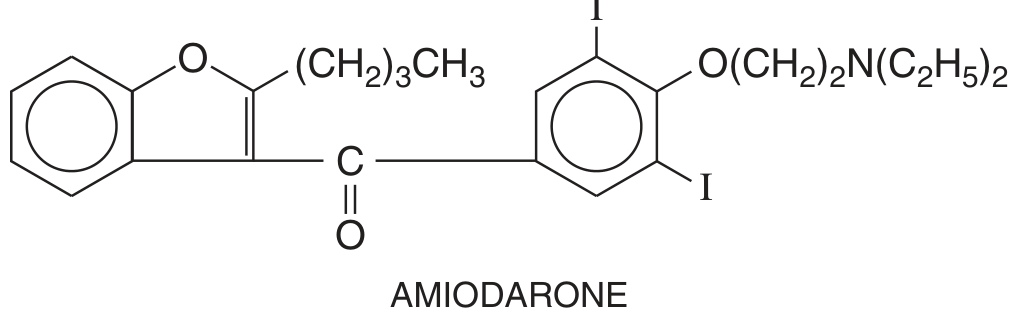
- A structural analogue of thyroid hormone; highly lipophilic; concentrated in fat, liver, and lungs
- Multi-channel blocker: Na⁺ (inactivated channels), K⁺ (IKr, IKs), Ca²⁺, plus non-competitive β-adrenergic block
- Prolongs APD in most tissues; slows conduction; reduces automaticity
- Extremely long t½ (40–55 days) — loading doses required; adverse effects resolve slowly
- First-line IV drug for VT/VF causing cardiac arrest; widely used for AF
- Organ toxicities (necessitate monitoring):
- Pulmonary toxicity — interstitial pneumonitis/fibrosis (5–10%); can be fatal
- Thyroid dysfunction — hyper- or hypothyroidism (iodine-rich; ~37% by weight iodine)
- Hepatotoxicity — elevated transaminases; rarely cirrhosis
- Corneal microdeposits — nearly universal but rarely impair vision
- Photosensitivity/skin discoloration — slate-grey/blue-grey
- Neurological — peripheral neuropathy, tremor, ataxia
Sotalol
- Both Class III (K⁺ block) and Class II (non-selective β-blocker) effects
- Renally eliminated; dose reduction in renal failure is essential
- Risk of torsades proportional to QTc prolongation; women at higher risk
- Used for maintenance of sinus rhythm in AF/flutter and for VT
Dofetilide
- Potent, "pure" IKr blocker — no extracardiac effects
- Effective for AF/flutter cardioversion and maintaining sinus rhythm
- Strict initiation protocol: requires in-hospital monitoring with QTc surveillance
- Renally cleared; contraindicated with severe renal failure and with renal cation transport inhibitors
- Torsades de pointes: 1–3% even in controlled settings
Ibutilide
- IV only; used for acute cardioversion of AF/flutter
- Also enhances slow inward Na⁺ current during plateau phase
- Torsades de pointes occurs in ~4–8%
Dronedarone
- Non-iodinated analogue of amiodarone; fewer organ toxicities but significantly less effective
- Blocks IKr, IKs, IK1, acetylcholine-activated K⁺, peak Na⁺ current, and L-type Ca²⁺ current; stronger antiadrenergic effect than amiodarone
- Inhibits CYP3A4 and P-glycoprotein; significant drug interactions
- Increased mortality in permanent AF (ATHENA trial subset) and in severe heart failure (ANDROMEDA trial)
- GI adverse effects common (diarrhea, nausea, abdominal pain)
Class IV — Calcium Channel Blockers (Non-Dihydropyridine)
Verapamil and Diltiazem are the antiarrhythmic CCBs. Unlike dihydropyridines (nifedipine), they block cardiac Ca²⁺ channels at clinical doses.
Mechanism: Slow AV nodal conduction (↑ PR interval), increase AV nodal refractoriness → terminate AV nodal reentrant circuits; reduce SA node automaticity
Clinical uses:
- Acute termination of AVNRT (PSVT)
- Rate control in AF/flutter
- Verapamil-sensitive VT (fascicular VT — responds to verapamil)
Adverse effects: Bradycardia, AV block, hypotension, constipation (verapamil), negative inotropy (use cautiously in HF)
Warning: Both drugs are contraindicated in WPW with AF — blocking the AV node may accelerate conduction down the accessory pathway → VF
Other Agents
Adenosine
- Activates A₁ receptors → increases IKAch (K⁺ inward) and inhibits I_f → hyperpolarization and AV nodal block
- Drug of choice for acute termination of AVNRT (PSVT) — given as rapid IV bolus (6–12 mg)
- t½ of seconds (cellular uptake and deamination); adverse effects are transient
- Adverse: transient asystole, chest fullness/dyspnea, rarely AF or bronchospasm
- Potentiated by dipyridamole (adenosine uptake inhibitor); antagonized by theophylline and caffeine
- Increased sensitivity in cardiac transplant recipients (denervation hypersensitivity)
Digoxin
- Inhibits Na⁺/K⁺-ATPase → ↑ intracellular Ca²⁺ → positive inotropy
- Antiarrhythmic effect mediated mainly via enhanced vagal tone → ↓ AV nodal conduction
- Used for rate control in AF (especially with heart failure)
- Narrow therapeutic index; toxicity: nausea, visual disturbances (yellow-green halos), various arrhythmias (DAD-mediated)
- Absolute contraindication in WPW — may accelerate accessory pathway conduction
Magnesium
- Drug of choice for torsades de pointes (regardless of serum Mg²⁺ level)
- Also used for digoxin toxicity arrhythmias and refractory VF
5. Mechanistic Approach to Arrhythmia Treatment
The following table summarizes recommended therapy by arrhythmia type (from Goodman & Gilman's):
| Arrhythmia | Mechanism | Acute Therapy | Chronic Therapy |
|---|---|---|---|
| AF | Disorganized functional reentry | Rate control (AV nodal block) or DC cardioversion | Rate control (β-blocker, CCB, digoxin) or rhythm control (flecainide, amiodarone, dofetilide) |
| Atrial Flutter | Stable reentrant circuit (right atrium) | Same as AF | AV nodal-blocking drugs; ablation preferred |
| AVNRT (PSVT) | Reentry within/near AV node | Adenosine (first-line); vagal maneuvers; verapamil | Ablation (preferred); flecainide; AV nodal blockers |
| WPW-related arrhythmias | Accessory pathway conduction | Procainamide IV (if AF); avoid digoxin/verapamil | Ablation |
| Ventricular tachycardia (stable) | Reentry in diseased myocardium | Lidocaine, procainamide, amiodarone IV | Amiodarone, sotalol, ICD |
| VT/VF (cardiac arrest) | Reentry/multiple mechanisms | Defibrillation; IV amiodarone or lidocaine | ICD ± amiodarone |
| Torsades de pointes | EADs (K⁺ channel block, QT prolongation) | IV Magnesium; temporary pacing | Remove offending drug; correct electrolytes |
6. Principles of Clinical Use
-
Identify and remove precipitating factors — electrolyte imbalances (hypokalemia, hypomagnesemia), ischemia, drug interactions, hypoxia, and thyroid disease are common contributors
-
Establish clear goals — Symptom relief vs. mortality benefit are different endpoints. Rate control vs. rhythm control must be consciously chosen (e.g., in AF)
-
Minimize risks — Proarrhythmia is real. Do not treat asymptomatic arrhythmias unless mortality benefit is established (the CAST lesson). Assess QTc, renal function, and structural heart disease before prescribing
-
Consider structural heart disease — Most Na⁺ channel blockers (Class IC) and some Class III agents are dangerous in ischemic or severely dysfunctional hearts. Only β-blockers and amiodarone have demonstrated safety data in structural heart disease
-
Drug combinations — Amiodarone + a Na⁺ channel blocker (e.g., flecainide) is the most studied combination for refractory ventricular arrhythmias. The HARMONY trial examined ranolazine + low-dose dronedarone for AF burden reduction. However, additive QT prolongation risk must always be anticipated
— Katzung's Basic and Clinical Pharmacology, 16th Ed.; Goodman & Gilman's The Pharmacological Basis of Therapeutics
7. Proarrhythmic Risk Summary
| Risk | Drugs | Mechanism |
|---|---|---|
| Torsades de pointes | Class IA, III (especially dofetilide, sotalol, quinidine); also macrolides, antipsychotics | QT prolongation, EADs |
| Fatal reentrant VT | Class IC (flecainide, encainide post-MI) | Slow conduction in ischemic tissue |
| AV block | Class II, IV; adenosine; amiodarone; digoxin | Excessive AV nodal suppression |
| Accelerated ventricular rate in WPW+AF | Digoxin, verapamil, diltiazem | AV node blocked → accessory pathway conducts faster |
| DAD-triggered arrhythmias | Digitalis | Intracellular Ca²⁺ overload |
Key references: Goodman & Gilman's The Pharmacological Basis of Therapeutics, 14th Ed. • Katzung's Basic and Clinical Pharmacology, 16th Ed. • Lippincott Illustrated Reviews: Pharmacology
This is a shared conversation. Sign in to Orris to start your own chat.