Using millers anaesthesiology book discuss in detail about Spinal Anaesthesia and Epidural Anaesthesia
Spinal and Epidural Anaesthesia
KEY POINTS (from Miller's)
- The distal termination of the spinal cord varies from L3 in infants to the lower border of L1 in adults.
- The speed of neural blockade depends on the size, surface area, and degree of myelination of the nerve fibers.
- Blockade of the peripheral (T1–L2) and cardiac (T1–T4) sympathetic fibers causes the decrease in arterial blood pressure seen with neuraxial techniques.
- CSF volume and local anesthetic baricity are the most important determinants of spread (block height) in spinal anesthesia.
- Smaller-gauge needles decrease the incidence of post-dural puncture headache (PDPH).
- Serious neurologic complications are rare but risk increases in the elderly and those with preexisting spinal pathology.
- LMWH and potent platelet inhibitors increase risk of epidural hematoma.
- Epidural blood patches are >90% effective in relieving PDPH.
PART I — SPINAL ANAESTHESIA
1. Historical Context
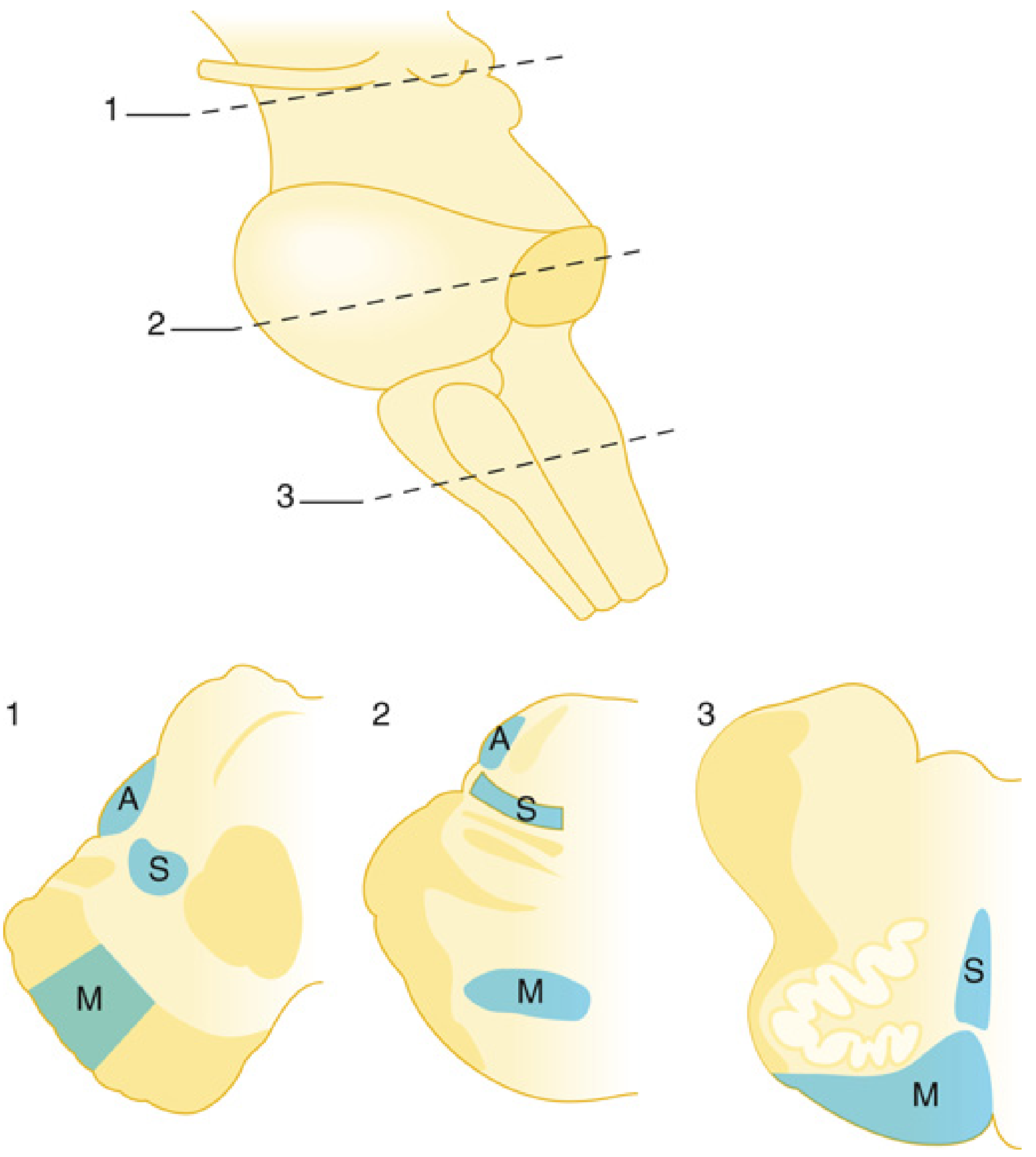
2. Relevant Anatomy
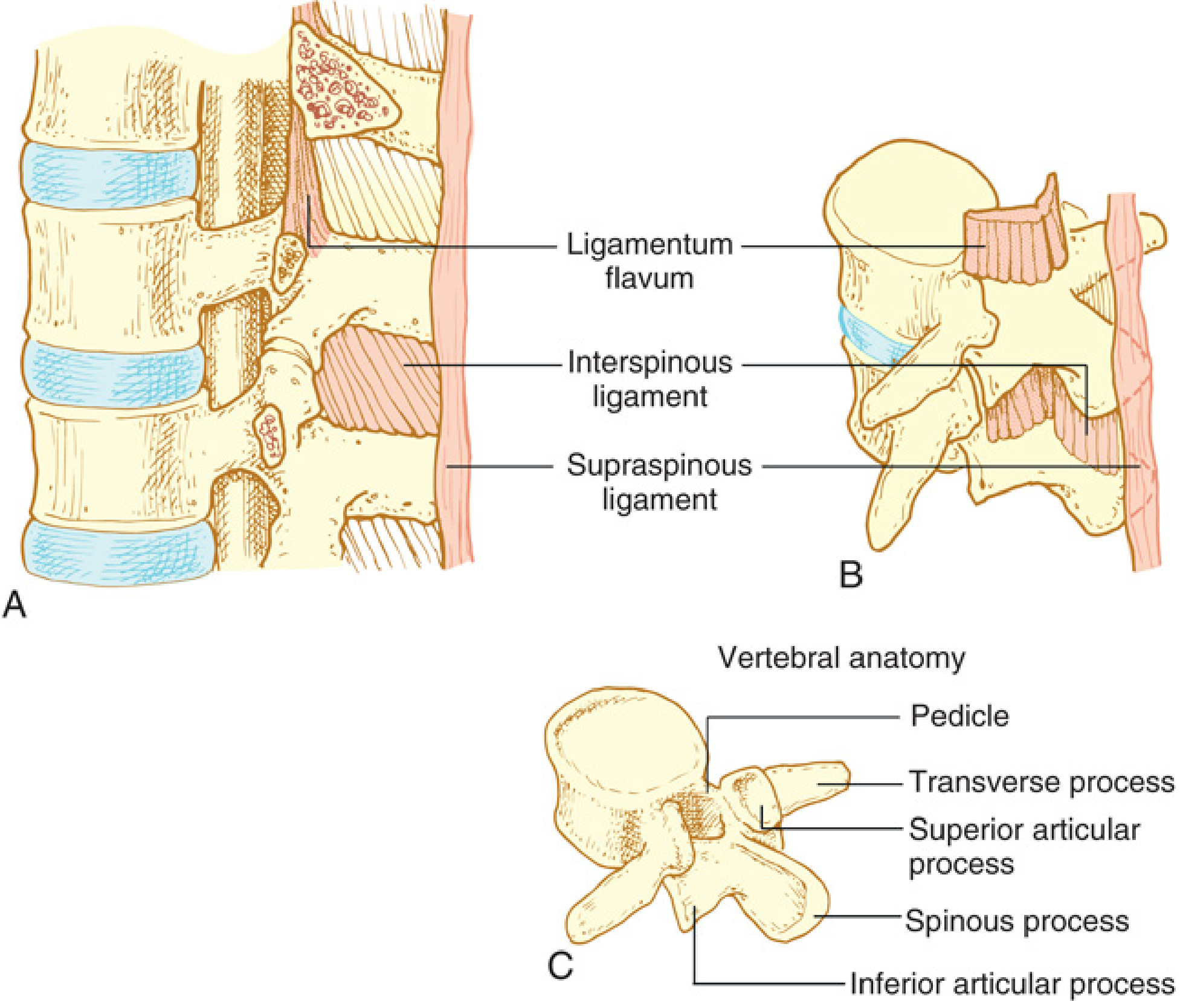
- Skin and subcutaneous fat
- Supraspinous ligament — connects spinous processes C7–L4
- Interspinous ligament
- Ligamentum flavum — two fused ligamenta flava with variable midline fusion; extends from foramen magnum to sacral hiatus
- Epidural space
- Dura mater
- Subdural space (potential)
- Arachnoid mater
- Subarachnoid space (containing CSF)
3. Mechanism of Action & Drug Uptake
| Fiber | Size | Myelination | Function Blocked | Order |
|---|---|---|---|---|
| B fibers | 1–3 μm | Minimally | Preganglionic sympathetic | 1st (most sensitive) |
| C fibers | 0.3–1 μm | Unmyelinated | Cold sensation | 2nd |
| A-delta | 1–4 μm | Myelinated | Pinprick | 3rd |
| A-beta | 5–12 μm | Myelinated | Touch | 4th |
| A-alpha | 12–20 μm | Myelinated | Motor | Last (most resistant) |
4. Physiologic Effects
Cardiovascular
- Sympathectomy from T1–L2 blockade decreases systemic vascular resistance and cardiac output.
- In healthy normovolemic patients, SVR decreases only 15–18% even with near-total sympathectomy.
- In elderly cardiac patients, SVR may fall ~25%; cardiac output falls ~10%.
- Heart rate decreases due to: (a) blockade of T1–T4 cardiac accelerator fibers, (b) reduced venous return activating intrinsic atrial chronotropic stretch receptors → increased vagal tone.
- Bezold–Jarisch reflex: profound bradycardia/circulatory collapse, especially in hypovolemia when a small left ventricular end-systolic volume triggers mechanoreceptor-mediated bradycardia.
- Risk factors for exaggerated bradycardia: baseline HR <60 bpm, age <37 years, male sex, non-emergency status, β-blocker use.
- Coronary blood flow decreases parallel to MAP; myocardial oxygen extraction is unchanged. High thoracic block may improve ischemia by reducing afterload.
Respiratory
- Intercostal muscles affected first at thoracic levels, then accessory muscles (lower cervical), then diaphragm (C3–C5), then brainstem centers (total spinal).
- Clinical concern is mainly from motor block of intercostal muscles causing impaired cough, not from reduced tidal volume (diaphragm usually preserved).
CNS / Cerebral
- Spinal anesthesia may modestly reduce cerebral blood flow in proportion to blood pressure reduction.
- In the very elderly, cerebral autoregulation may be impaired.
Other Organ Systems
- Hepatic: blood flow falls proportionate to BP reduction; hepatic arteriovenous oxygen difference unchanged.
- Renal: renal blood flow decreases with BP but autoregulation is partially maintained.
- GI: unopposed parasympathetic activity causes bowel contraction (small, contracted bowel — useful for surgical exposure).
5. Factors Affecting Block Height in Spinal Anaesthesia
Drug Factors
| Factor | Effect |
|---|---|
| Baricity | Most important. Hyperbaric → dependent spread; hypobaric → non-dependent spread; isobaric → gravity-independent |
| Dose | Primary determinant of block height and duration |
| Volume/concentration | Less important than dose; may increase spread modestly |
| Temperature | Minor effect on baricity |
Patient Factors
- CSF volume — the single most important non-manipulable factor; decreased CSF volume (obesity, pregnancy, ascites) leads to higher, more unpredictable blocks.
- Body habitus: height influences spread (particularly extremes); weight less important.
- Pregnancy: engorgement of epidural veins reduces CSF volume → higher blocks with less drug.
- Vertebral column curvature: thoracic kyphosis (T5–T8) is the most dependent region in supine patients; hyperbaric solutions pool here.
Procedure Factors
- Level of injection: more cephalad injection → higher block (particularly with isobaric).
- Patient position: lateral decubitus allows preferential block of dependent side with hyperbaric solutions.
- Needle orifice direction: hypobaric solutions may spread more cephalad with cephalad-directed orifice.
- Injection rate and barbotage have NOT consistently been shown to affect block height.
6. Spinal Technique — The Four P's
Preparation
- Informed consent, IV access, monitors (SpO₂, NIBP, ECG).
- Resuscitation equipment must be immediately available.
- Choose local anesthetic matched to surgical duration (see Table 41.4).
- Use preprepared sterile packs; sterility is paramount.
Position
- Lateral decubitus (left or right): most common; allows hyperbaric solutions to block the operative (dependent) side preferentially.
- Sitting: used for saddle block (perineal/anal surgery). Dose of 1–2 mg of long-acting hyperbaric agent achieves saddle block.
- Thoracic kyphosis should be maximized ("curled" position) to open interspinous spaces.
Spinal Needles
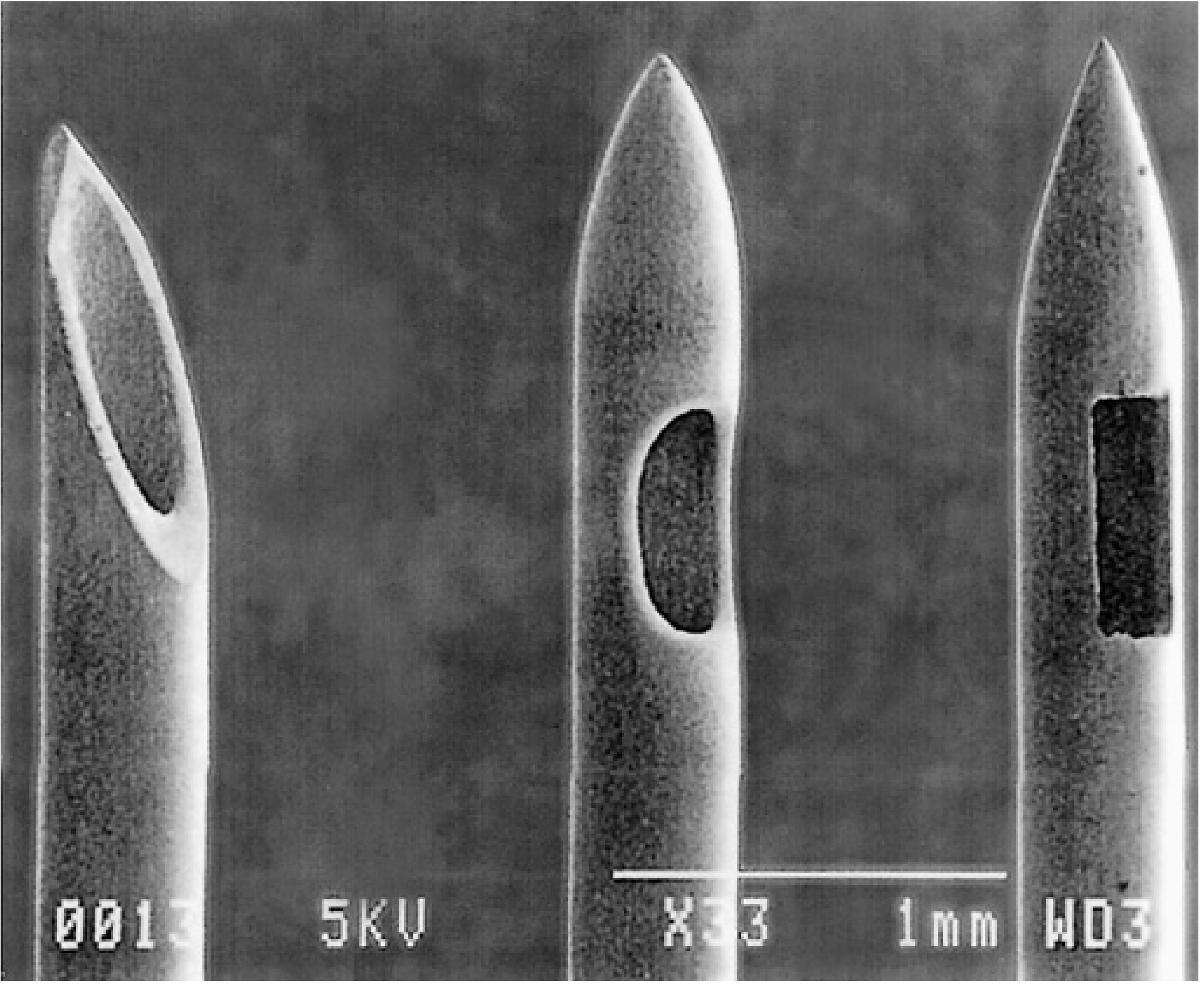
- Cutting tips (Quincke, Pitkin): cut the dura; higher PDPH risk.
- Pencil-point/atraumatic tips (Whitacre, Sprotte, Pencan): separate dural fibres; lower PDPH.
- Needle gauge: PDPH falls from 40% with 22G to <2% with 29G; but 29G has higher procedural failure. 26G atraumatic needles have the lowest combined risk of PDPH and failure.
- For continuous spinal: Tuohy (17G) or Hustead (18G) needles with 20G epidural catheter.
Approaches
- Midline: needle through supraspinous → interspinous ligament → ligamentum flavum → epidural → dura → subarachnoid.
- Paramedian: bypasses interspinous ligament; useful in kyphoscoliosis, calcified ligaments.
- Taylor approach: modified paramedian at L5–S1 (largest interspace).
Confirmation of Placement
- Free flow of CSF confirms subarachnoid placement.
- Aspiration before and after injection; injection of only preservative-free solutions.
7. Local Anaesthetic Pharmacology for Spinal Use
| Drug | Class | Duration | Typical Dose |
|---|---|---|---|
| Procaine | Ester, short | 45–60 min | 50–200 mg |
| Chloroprocaine | Ester, short | 30–60 min | 30–60 mg |
| Lidocaine* | Amide, intermediate | 60–90 min | 25–100 mg |
| Mepivacaine | Amide, intermediate | 90–150 min | 40–80 mg |
| Bupivacaine | Amide, long | 120–240 min | 5–20 mg |
| Levobupivacaine | Amide, long | 120–240 min | 5–20 mg |
| Ropivacaine | Amide, long | 120–210 min | 8–20 mg |
| Tetracaine | Ester, long | 120–240 min | 6–20 mg |
8. Spinal Additives
- Effect is through dorsal horn opioid receptor activation + cerebral receptor activation via CSF transport + systemic vascular uptake.
- Lipophilic opioids (fentanyl, sufentanil): rapid onset, short duration, limited CSF spread, less late respiratory depression. Intrathecal:IV potency ratio = 10–20:1.
- Hydrophilic opioids (morphine, diamorphine): slow onset, prolonged analgesia (up to 24h), wide CSF spread, risk of delayed respiratory depression. Intrathecal:IV potency ratio = 200–300:1.
- Preservative-free intrathecal morphine: 100 mcg for cesarean delivery; up to 300–1000 mcg for major abdominal/thoracic surgery.
- Side effects of neuraxial opioids: nausea/vomiting, pruritus, urinary retention, respiratory depression.
- Epinephrine: prolongs block duration; vasoconstriction reduces vascular uptake.
- Clonidine (α₂-agonist): prolongs sensory and motor block via potassium channel opening/membrane hyperpolarization.
- Neostigmine: provides analgesia via spinal muscarinic receptors; side effects include nausea.
9. Block Monitoring
- Cold sensation (ethyl chloride/alcohol/ice) — most cephalad level, approximates sympathetic block (C-fibers)
- Pinprick (Neuropen) — Aδ fibers, 1–2 segments below cold level
- Touch — Aβ fibers, 1–2 segments below pinprick
- 0: Full flexion of knees and feet
- 1: Unable to raise extended leg; full flexion of knees and feet
- 2: Unable to raise extended leg, flex knee; able to move feet
- 3: Complete block of lower limb
10. Special Techniques
Continuous Spinal Anaesthesia
- Allows incremental dosing → predictable titration + better hemodynamic stability (useful in severe aortic stenosis, complex cardiac disease in obstetrics).
- Catheter threaded 2–3 cm into subarachnoid space; never withdraw catheter back into needle.
- Spinal microcatheters associated with cauda equina syndrome due to lumbosacral pooling.
Unilateral / Selective Spinal Anaesthesia
- Uses hyperbaric solutions + prolonged lateral positioning to produce a unilateral block.
- 4–5 mg hyperbaric bupivacaine adequate for knee arthroscopy; 8 mg for inguinal herniorrhaphy.
- Goal: minimize drug dose, hasten recovery, reduce hemodynamic effects.
PART II — EPIDURAL ANAESTHESIA
1. Principles & Comparison with Spinal
| Feature | Spinal | Epidural |
|---|---|---|
| Drug volume | Small (1–4 mL) | Large (10–20+ mL) |
| Systemic absorption | Negligible | Significant; active blood concentrations |
| Onset | Rapid (2–5 min) | Slower (10–20 min) |
| Block predictability | High | Moderate (more variable) |
| Catheter technique | Less common | Standard practice |
| Duration flexibility | Limited (single shot) | Unlimited with catheter |
"Spinal anesthesia requires a small mass of drug almost devoid of systemic pharmacologic effects to produce rapid, profound and reproducible sensory analgesia. In contrast, epidural anesthesia progresses more slowly after a large mass of local anesthetic that produces pharmacologically active systemic blood concentrations." — Miller's, p. 6014
2. Epidural Space Anatomy
- Posterior boundary: ligamentum flavum, laminae
- Anterior boundary: posterior longitudinal ligament covering vertebral bodies
- Skin-to-epidural space depth: typically 4–6 cm; varies significantly.
- Epidural veins are valveless and communicate with pelvic veins — distended in pregnancy/raised abdominal pressure → increased risk of intravascular injection.
- The epidural space is largest at L2 (~5 mm posterior depth) and smallest in the thoracic region.
3. Factors Affecting Epidural Block Height
Drug Factors
- Volume and total mass are most important; as a general principle, 1–2 mL per spinal segment to be blocked.
- Additives (bicarbonate, epinephrine, opioids) influence onset/quality/duration but NOT spread.
Patient Factors
- Age: stronger correlation with thoracic epidurals; elderly may require 40% less volume. Reasons: less leakage through foramina, increased compliance of epidural space, increased nerve sensitivity.
- Height: only extremes affect spread.
- Weight: not well correlated with spread.
- Pregnancy: less drug required (partly due to epidural vein engorgement reducing epidural volume, but also occurs in early pregnancy).
- CPAP: increases height of thoracic epidural block.
Procedure Factors
- Level of injection: most important procedural factor.
- Upper cervical → mostly caudal spread
- Mid-thoracic → equal cephalad and caudal spread
- Low thoracic/lumbar → primarily cephalad spread
- Patient position: lateral decubitus → preferential spread to dependent side; head-down tilt increases cephalad spread in obstetrics.
- Needle bevel direction and injection speed do not significantly affect spread.
4. Epidural Pharmacology
Local Anaesthetics
| Duration | Agents |
|---|---|
| Short-acting | Chloroprocaine (30–60 min), Lidocaine |
| Intermediate-acting | Mepivacaine (2–3h), Prilocaine |
| Long-acting | Bupivacaine, Levobupivacaine, Ropivacaine (2–4h+) |
Epidural Opioids
- Extended-release epidural morphine (DepoDur): single epidural injection provides up to 48h of analgesia; 10–15 mg for lower abdominal surgery.
- Standard morphine: reliable, long-acting analgesia; risk of delayed respiratory depression (6–18h post-injection) requiring monitoring.
- Fentanyl: lipophilic; rapid onset, primarily systemic rather than spinal mechanism; useful in combination infusions.
Epidural Additives
- Epinephrine: reduces vascular absorption, prolongs block duration, serves as marker for intravascular injection (15 mcg = test dose).
- Clonidine (α₂-agonist): prolongs sensory > motor block via potassium channel opening; reduces epidural LA and opioid requirements; side effects: hypotension, bradycardia, sedation. Cardiovascular effects greatest with thoracic epidural clonidine.
- Dexmedetomidine: reduces intraoperative requirements, prolongs sensory and motor block.
- Bicarbonate/Carbonation: raises solution pH → more non-ionized drug → faster onset; carbonated solutions used for emergency cesarean delivery to rapidly establish block.
- Neostigmine: provides labor analgesia without respiratory depression or motor block.
5. Epidural Technique
Equipment
- Tuohy needle (most common): 16–18G; 15–30° curved blunt "Huber" tip designed to reduce accidental dural puncture and guide catheter cephalad; shaft marked in 1-cm intervals.
- Epidural catheter: flexible plastic, 3–5 cm advanced into epidural space.
- Catheter should be advanced no more than 5–6 cm into the epidural space to reduce risk of intravascular placement.
Identification of the Epidural Space
| Method | Description |
|---|---|
| Loss of Resistance (LOR) to air | Resistance disappears as needle exits ligamentum flavum; risk of pneumocephalus, patchy block |
| LOR to saline | Preferred method; syringe with continuous gentle pressure; safer, more reliable |
| Hanging drop technique | Drop of saline on needle hub is drawn in by negative pressure in epidural space; less reliable |
- Identifies optimal needle insertion site, predicts skin-to-dura distance, reduces failure rate.
- Real-time guidance is more challenging in adults due to ossification; easier in pediatric patients.
- Thoracic ultrasonography is harder (T5–T8 have narrowest interspinous windows).
Test Dose
- 3 mL of 1.5% lidocaine with 15 mcg epinephrine:
- Intravascular placement: HR increases >20 bpm within 60 seconds (epinephrine marker).
- Intrathecal placement: rapid dense motor and sensory block within 3–5 minutes.
- Controversy in obstetrics: epinephrine may theoretically reduce uterine blood flow; false-positive responses in active labor.
- Incremental dosing (5 mL fractions) and aspiration before each dose remain essential — no fail-safe test exists.
6. Lumbar, Thoracic, and Cervical Epidural
| Level | Indications | Notes |
|---|---|---|
| Lumbar | Obstetric analgesia, lower limb/pelvic surgery | Most common; spread predominantly cephalad |
| Thoracic (TEA) | Major thoracic, abdominal surgery; postoperative analgesia | Steeply angled spinous processes; higher difficulty; superior analgesia for chest/abdominal pain |
| Cervical | Rare; chronic neck pain, upper limb procedures | Highest risk; requires great expertise |
- Superior postoperative analgesia, particularly for thoracotomy and laparotomy.
- Reduces sympathetic tone to heart → anti-ischemic in coronary disease patients.
- Reduces pulmonary complications in patients with preexisting lung disease undergoing abdominal surgery.
7. Combined Spinal–Epidural (CSE)
- First described in 1937; modified over 40 years.
- Advantages:
- Rapid onset of spinal block (procedure begins early).
- Epidural catheter allows extension as spinal resolves.
- Low-dose spinal with epidural backup → reduced side effects, faster recovery.
- Epidural Volume Extension (EVE): injecting saline through epidural catheter compresses dural sac → increases spinal block height without increasing intrathecal drug dose → comparable sensory block with faster motor recovery.
- Technique: Epidural needle placed first → needle-through-needle subarachnoid injection → catheter placed through epidural needle, OR separate needle insertions at same/different interspaces.
- Particularly useful in: labour analgesia, cesarean delivery in high-risk cardiac patients, prolonged surgeries.
PART III — CONTRAINDICATIONS
Absolute
- Patient refusal
- Localized infection at injection site
- Allergy to planned drugs
- Inability to remain still during needle insertion
- Raised intracranial pressure (theoretical brainstem herniation risk)
Relative (by System)
| System | Condition | Considerations |
|---|---|---|
| Neurologic | Myelopathy/peripheral neuropathy | Double-crush risk; no definitive evidence of harm |
| Neurologic | Spinal stenosis | Increased risk of neurologic complications |
| Neurologic | Multiple sclerosis | Demyelinated fibers more sensitive; use lower dose; epidural preferred |
| Coagulopathy | LMWH, anticoagulants, platelet inhibitors | Follow ASRA guidelines (Table 41.1): time intervals before/after block |
| Cardiovascular | Severe aortic stenosis | Fixed cardiac output; continuous spinal preferred for gradual titration |
| Infection | Systemic bacteremia | Theoretical risk of epidural hematoma/abscess; weigh risks/benefits |
| Neurologic | Previous spinal surgery | Unpredictable LA spread; hardware may block needle access |
PART IV — COMPLICATIONS
Complications Common to Both Techniques
Post-Dural Puncture Headache (PDPH)
- Caused by CSF leakage through dural hole → reduction in CSF pressure → downward traction on pain-sensitive intracranial structures.
- Postural: bilateral frontal/occipital headache, worse upright, better supine; associated nausea, photophobia, tinnitus.
- Risk factors (Box 41.2):
- Younger age (more frequent)
- Female > male
- Larger needle gauge
- Cutting (Quincke) > pencil-point (Whitacre/Sprotte) tip
- Pregnancy
- Multiple puncture attempts
- Bevel orientation perpendicular (rather than parallel) to long axis of neuraxis
- Conservative management: supine position, hydration, caffeine, analgesics, sumatriptan (variable effect).
- Epidural Blood Patch (EBP): definitive treatment. 15–20 mL of autologous blood injected at or below the level of dural puncture. Single EBP has >90% initial improvement rate; persistent resolution in 61–75%. Best performed ≥24h after dural puncture. Repeat EBP in 24–48h if needed.
Transient Neurologic Symptoms (TNS)
- Previously "transient radicular irritation." Bilateral/unilateral buttock/leg pain without neurologic deficit, occurring within 24h of spinal resolution, resolving within 1 week.
- Most associated with intrathecal lidocaine (but reported with all agents).
- Risk with bupivacaine, levobupivacaine, prilocaine, procaine, ropivacaine is RR 0.10–0.23 compared to lidocaine.
- More common in lithotomy position.
- Treatment: NSAIDs (first line); opioids for severe cases.
Hypotension
- Defined as SBP <90 mmHg or >20–30% reduction from baseline.
- Risk factors: block height ≥T5, age ≥40, baseline SBP <120 mmHg, combined spinal-general, puncture at or above L2–L3, phenylephrine in local anesthetic.
- Treatment: IV fluids (co-loading preferred over pre-loading), vasopressors (phenylephrine preferred in obstetrics to maintain uteroplacental blood flow; ephedrine as alternative for bradycardia-associated hypotension).
Cardiac Arrest
- Rate after spinal: 6.4/10,000 vs. 1/10,000 for all other neuraxial and peripheral techniques.
- Mechanism: multifactorial — T1–T4 sympathectomy + reduced venous return → vagally mediated asystole; exacerbated by hypoxia and over-sedation.
- Paradoxically more common in young, healthy, conscious patients.
High/Total Spinal
- Unintentional extension to cervical cord and brainstem.
- Progression: intercostal blockade → accessory muscle paralysis → diaphragm (C3–C5) → brainstem (respiratory arrest + loss of consciousness).
- Risk factors: obesity, short stature, spinal after failed epidural, epidural after dural puncture.
- Management: supportive — reassurance, hemodynamic support, intubation/ventilation until block resolves.
Neurologic Injury
- Serious neurologic complications are rare; risk increases in elderly and those with pre-existing spinal pathology.
- Causes: direct needle/catheter trauma, neurotoxicity of LA or additives, cauda equina syndrome (microcatheters + hyperbaric pooling), arachnoiditis, meningitis, epidural abscess, epidural hematoma.
Complications Unique to Epidural Anaesthesia
Intravascular Injection / Local Anaesthetic Systemic Toxicity (LAST)
- Vascular puncture rate: up to 10% (highest in obstetrics — dilated epidural veins).
- Seizure rate: up to 1%.
- Strategies to reduce risk:
- Lateral position during insertion (vs. sitting)
- Fluid through needle before catheter insertion
- Single-orifice catheter, wire-embedded polyurethane catheter
- Advance catheter <6 cm
- Aspiration before each dose
- Incremental injection (5 mL fractions)
- Epinephrine test dose (15 mcg) → HR rise >20 bpm = intravascular
Subdural Injection
- Incidence <1%; however, the subdural extra-arachnoid space is entered in 66% of autopsy attempts.
- Produces an unexpectedly high, patchy, but incomplete block; also extends intracranially.
- Management: supportive.
Accidental Dural Puncture (ADP)
- Incidence with epidural technique: 0.5–1%.
- If unrecognized → intrathecal injection of epidural dose → total spinal.
- If recognized → can be managed with intrathecal catheter (continuous spinal), or EBP after PDPH develops.
Epidural Hematoma
- Rare but catastrophic; risk significantly elevated by LMWH, antiplatelet agents, and anticoagulants.
- Presents as back pain + neurologic deficit (motor/sensory/sphincter).
- Emergency MRI + surgical decompression within 6–8 hours to prevent permanent paralysis.
- ASRA guidelines (Table 41.1) specify mandatory timing intervals for anticoagulants.
Epidural Abscess
- Risk factors: immunosuppression, long-term catheter, skin contamination.
- Presents with fever, back pain, neurologic deficit.
- Management: emergency MRI, IV antibiotics ± surgical drainage.
PART V — CLINICAL OUTCOMES & BENEFITS
- Reduced thromboembolic complications.
- Reduced intraoperative blood loss.
- Earlier return of gut function after abdominal surgery.
- Reduced pulmonary complications in high-risk patients.
- Potential reduction in cancer recurrence (ongoing research).
- Superior postoperative analgesia vs. systemic opioids → reduced opioid side effects.
Summary Comparison Table
| Feature | Spinal | Epidural |
|---|---|---|
| Drug volume | 1.5–4 mL | 10–25 mL |
| Onset | 2–5 min | 10–20 min |
| Spread determinant | Baricity + CSF volume | Volume/mass of injectate |
| Systemic drug levels | Negligible | Significant |
| Standard needle | 25–27G Whitacre/Sprotte | 16–18G Tuohy |
| Catheter | Occasionally (continuous spinal) | Routinely placed |
| Block predictability | High | Moderate |
| Hemodynamic effect | Often abrupt | Gradual (titratable) |
| PDPH risk | Present (needle dependent) | Only with ADP |
| Unique complication | TNS (lidocaine) | LAST, subdural injection, ADP |
| Test dose needed | No | Yes (epinephrine 15 mcg in 3 mL) |
| Duration flexibility | Fixed (single shot) | Unlimited |
Using millers anaesthesiology book discuss in detail about anaesthetic Management in acute vs chronic spinal cord injury, including classifications, scores, scales, neurosurgery anaesthetic implications, considerations, also you may add management in cord transection
Kindly do it now as I have upgraded
Anaesthetic Management in Spinal Cord Injury
PART I — OVERVIEW & EPIDEMIOLOGY
- 36% — motor vehicle collisions
- 42% — falls
- 4% — firearms
PART II — PATHOPHYSIOLOGY: PRIMARY AND SECONDARY INJURY
Primary Injury
Secondary Injury Cascade
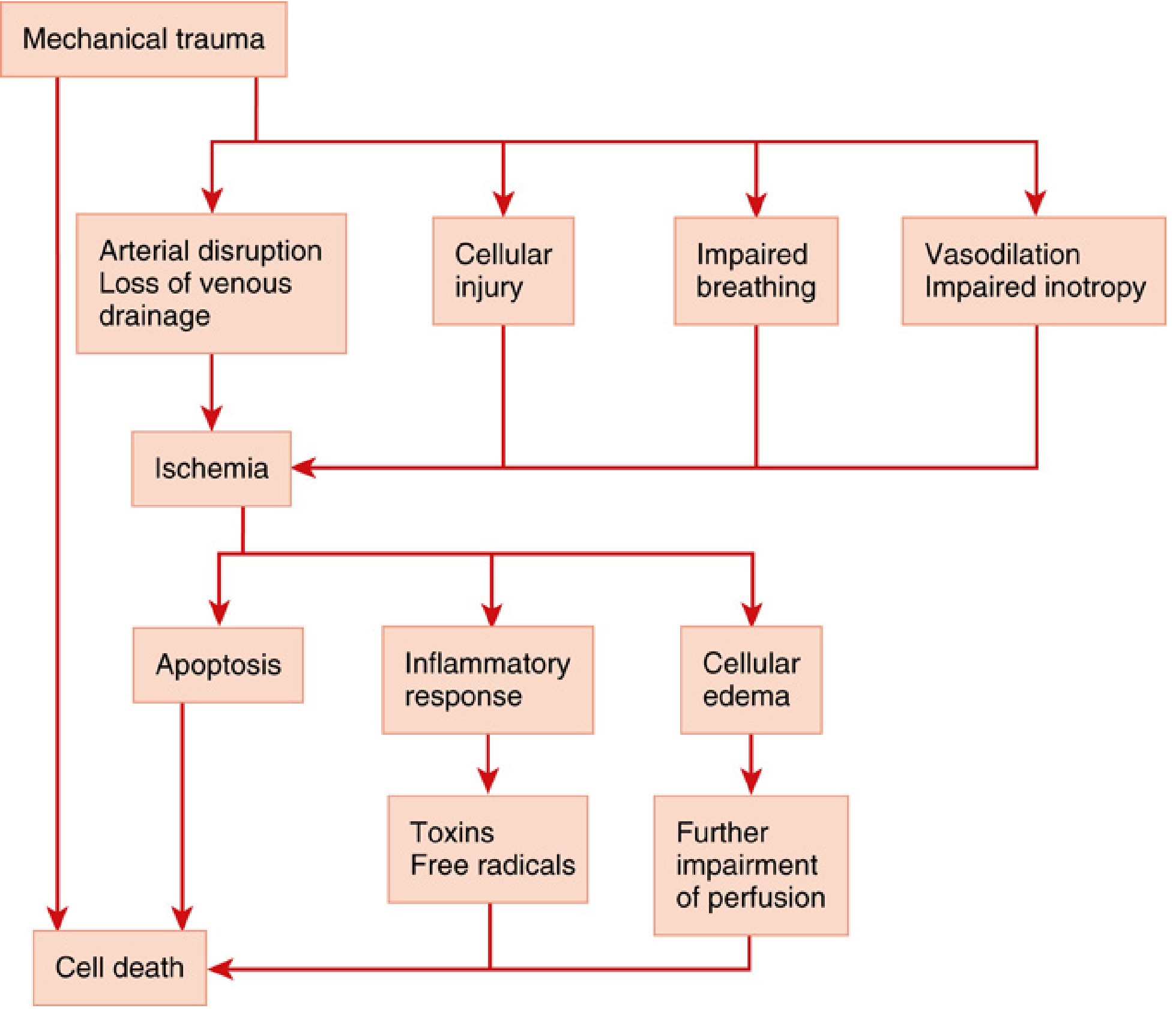
- Arterial disruption and loss of venous drainage → ischaemia
- Cellular injury
- Impaired breathing
- Vasodilation and impaired inotropy
- Ischaemia leads to: apoptosis, inflammatory response (toxins, free radicals), cellular oedema (further impairs perfusion) → cell death
PART III — CLASSIFICATION AND SCORING
1. ASIA Impairment Scale (AIS)
| Grade | Type | Clinical Presentation |
|---|---|---|
| A | Complete | No sensory or motor function preserved in sacral segments S4–S5 |
| B | Incomplete | Sensory but not motor function preserved below neurologic level, including S4–S5 |
| C | Incomplete | Motor function preserved below neurologic level; >half of key muscles below NLI have grade <3 (Grades 0–2) |
| D | Incomplete | Motor function preserved below neurologic level; ≥half of key muscles below NLI have grade ≥3 |
| E | Normal | Sensory and motor functions are normal |
Complete vs. incomplete: Complete deficits (ASIA A) represent total spinal cord disruption at one level and carry a more guarded prognosis, with generally slight improvement over time. Incomplete deficits (ASIA B–D) may be worse on one side and may improve rapidly in the first minutes after injury.
2. Three-Column Spinal Stability Model
Injuries to any two columns suggest biomechanical instability — these patients will often require urgent surgical stabilisation. — Miller's, p. 9445
3. Neurologic Level of Injury (NLI) — Functional Impact by Level
| Level | Functional Consequence |
|---|---|
| Above C3 | Apnoea; loss of all limb and trunk function |
| C3–C5 | Diaphragmatic function impaired (phrenic nerve C3–5); ventilatory failure inevitable |
| C5–C7 | Loss of chest wall innervation, paradoxical breathing, inability to clear secretions |
| C6–C7 | May require ventilatory support due to impaired respiratory muscle function |
| Above T4–T6 | Neurogenic shock — loss of cardiac accelerator fibers (T1–T4) |
| Above T7 | 85% risk of serious cardiovascular instability |
| T1–L2 | Peripheral sympathetic outflow disrupted |
4. Important Distinction: Neurogenic Shock vs. "Spinal Shock"
| Term | Meaning |
|---|---|
| Neurogenic shock | Correct term: cardiovascular consequence of SCI — hypotension + bradycardia from sympathectomy and cardiac accelerator denervation |
| "Spinal shock" | Misnomer in this context — refers to temporary loss of all reflexes below the level of injury (flaccidity, areflexia), not a haemodynamic state |
"Note the distinction between neurogenic shock and the incorrect term spinal shock, which refers to a loss of reflexes." — Miller's, p. 9446
5. Grading of TBI (often co-existing with SCI)
| Grade | GCS Score | Clinical Significance |
|---|---|---|
| Mild | 13–15 | Risk of post-concussive effects; unlikely to deteriorate if stable at 24h |
| Moderate | 9–12 | Intracranial lesions possible; early CT strongly indicated |
| Severe | ≤8 | Significant mortality risk; requires invasive ICP monitoring |
PART IV — ACUTE SCI: ANAESTHETIC MANAGEMENT
A. Initial Assessment & Resuscitation — ABCDE Approach
- Secure airway — avoid worsening spinal instability
- Prevent hypoxia and hypotension (secondary injury drivers)
- Maintain target MAP
- Immobilise spine until instability ruled out
- Identify and treat all injuries simultaneously (40%+ have concomitant injuries)
B. Airway Management in Acute Cervical SCI
When is Intubation Required?
- C5 and above: virtually ALL require intubation (Como et al. study: 100% at C5+).
- C6–C7: 71% required tracheostomy in published studies.
- Complete cervical SCI: 70% needed tracheostomy in a study of 178 patients.
- Ventilatory support almost always required for deficits above C4 (insufficient diaphragmatic function).
- Patients at C6–C7 may require support due to: (1) loss of chest wall innervation, (2) paradoxical respiratory motion, (3) inability to clear secretions, (4) decreased lung and chest wall compliance. — Miller's, p. 9447
Choice of Intubation Technique
"No technique for airway management in patients with unstable SCI is superior for preventing neurologic deterioration." — Miller's, p. 9450
| Technique | Indications / Notes |
|---|---|
| Direct laryngoscopy + in-line stabilisation (MILS) | Emergency setting, unconscious/combative/hypoxaemic patients, or when spine status is unknown |
| Awake fiberoptic intubation (AFOI) | Gold standard for cooperative, alert patients with known cervical instability. Nasal route easier but risks sinusitis with prolonged ventilation; oral route preferred if patient will remain ventilated |
| Video laryngoscopy | Acceptable; may have higher first-attempt failure rate vs. direct laryngoscopy in some studies |
| Blind nasal intubation | Acceptable but less common |
| Intubating LMA | Acceptable alternative |
| RSI | Required for obtunded, combative, or hypoxaemic patients — cannot wait for awake technique |
Manual In-Line Stabilisation (MILS)
- An assistant maintains axial alignment of the head and neck during laryngoscopy.
- Does not guarantee absence of cervical motion.
- Removes cervical collar to allow mouth opening, but an assistant maintains the head.
C. Haemodynamic Management: Neurogenic Shock
- Loss of sympathetic outflow to heart and vasculature
- Unopposed vagal tone → inappropriate bradycardia + vasodilation
- Loss of cardiac accelerator function (T1–T4)
- Loss of peripheral vasoconstriction (T1–L2)
"This situation can be difficult to distinguish from hypotension resulting from acute haemorrhage. A trial of fluid administration is still indicated." — Miller's, p. 9451
MAP Targets
Target MAP >85 mmHg for the first 7 days after SCI may improve functional recovery. — Miller's, p. 9451 and p. 12011
Vasopressor Selection
| Vasopressor | Role | Notes |
|---|---|---|
| Norepinephrine | First-line | α₁ + β₁ agonist — vasoconstrictive, inotropic, and chronotropic |
| Dopamine | Alternative | α₁ + β₁ agonist; both vasoconstrictive and chronotropic |
| Dobutamine | Avoid | β₂ receptor activation causes vasodilation — worsens neurogenic shock |
Fluid Therapy
- Use isotonic crystalloids (normal saline, Plasma-Lyte).
- Avoid hypotonic solutions: D5W, Ringer's lactate, 0.45% NaCl — all worsen cord oedema. — Miller's, p. 12011
- Monitor with cardiac output devices.
Bradycardia Management
- Severe or life-threatening bradycardia: atropine is first line.
- Refractory cases: consider cardiac pacemaker placement. — Miller's, p. 12011
D. Respiratory Management by Injury Level
| Level | Respiratory Muscles Affected | Clinical Consequence |
|---|---|---|
| C1–C2 | All respiratory muscles including diaphragm | Immediate apnoea; permanent ventilator dependence |
| C3–C5 | Diaphragm impaired (phrenic nerve) | Respiratory failure; likely ventilator dependence |
| C5–T1 | Intercostal + accessory muscles | Reduced FRC, paradoxical breathing, unable to cough |
| Below T6 | Abdominal muscles affected | Reduced cough efficacy, progressive atelectasis |
- Intubate early before hypoxia renders patient uncooperative.
- Wean from ventilation early once haemodynamically stable.
- If weaning complicated → early tracheostomy to: reduce mechanical ventilation days, decrease sedation need, facilitate pulmonary toilet. — Miller's, p. 12012
- 70% of complete cervical SCI patients require tracheostomy.
E. Surgical Decompression — Timing
Surgical decompression should be performed within 24 hours after SCI and is associated with improved neurologic outcome. — Miller's, p. 12009
- A patient with partial neurologic deficit and visible spinal canal impingement on imaging is considered a surgical emergency.
- Laminectomy in motor-complete thoracic SCI patients has significantly increased the rate of successful spinal cord decompression.
- Timing of surgery for cervical injuries is particularly important.
- Earlier decompression may improve outcomes, though exact timing remains controversial. — Miller's, p. 9451
F. Steroids — Current Position
Current guidelines do not recommend methylprednisolone for acute SCI.
G. Intraoperative Anaesthetic Management — Acute SCI Surgery
Monitoring
- Standard ASA monitoring + invasive arterial line (mandatory — allows beat-to-beat BP and MAP monitoring for neurogenic shock).
- Central venous line for vasopressor administration.
- Cardiac output monitoring (thermodilution PA catheter or non-invasive device).
- Spinal cord perfusion pressure (SCPP) monitoring via intradural catheter — still under investigation; benefits must be weighed against risk of injury and infection. — Miller's, p. 12011
- Intraoperative neuromonitoring (IONM): Somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) — used to detect intraoperative cord compromise during spinal surgery.
Positioning
- Prone positioning for posterior spinal surgery requires particular care:
- Ensure no pressure on eyes or face.
- Avoid cervical hyperflexion or hyperextension.
- Use frames (Wilson, Jackson) that permit abdominal wall excursion to reduce epidural venous pressure and blood loss.
- Inspect all pressure points meticulously.
Anaesthetic Agents
- Volatile agents: safe to use; dose-dependently suppress IONM signals — reduce MAC to minimise interference with MEPs/SSEPs.
- Total intravenous anaesthesia (TIVA) with propofol + remifentanil: preferred when robust IONM is required (less interference with MEPs/SSEPs than volatile agents).
- Opioids: safe for haemodynamic management; remifentanil ideal for titratable short-acting analgesia during TIVA.
- Ketamine: useful for haemodynamic support in neurogenic shock; low-dose may be neuroprotective.
Neuromuscular Blockade — CRITICAL CONSIDERATION
- Massive efflux of intracellular potassium from the entire muscle surface (not just the normal end-plate region)
- Can cause life-threatening hyperkalaemia (serum K⁺ may rise by 5–10 mEq/L)
- Can result in ventricular fibrillation and cardiac arrest
"Succinylcholine should not be administered to patients with Guillain–Barré syndrome [or any denervation state] because of the risk of severe hyperkalaemia." — Miller's, p. 4385
- Safe to use in the first 24–48 hours after acute SCI (before upregulation occurs) for RSI in the emergency setting.
- Risk begins 48–72 hours after SCI.
- Risk persists for months to years in chronic SCI.
- Use non-depolarising NMBDs (rocuronium, vecuronium, atracurium) at all times beyond the acute phase.
PART V — CORD TRANSECTION (COMPLETE SCI — ASIA A)
Pathophysiology of Complete Cord Transection
- "Spinal shock" phase: below the level of injury — complete flaccidity, areflexia, loss of all sensation, urinary retention, paralytic ileus.
- This phase lasts days to weeks.
- Neurogenic shock (cardiovascular): concurrent hypotension + bradycardia from sympathectomy (if level ≥T6).
- Spinal shock resolves: spasticity, hyperreflexia develop below level of injury.
- Isolated spinal cord resumes reflex activity independently of supraspinal control.
- Autonomic dysreflexia emerges — the most clinically dangerous chronic complication.
PART VI — CHRONIC SCI: ANAESTHETIC MANAGEMENT
Preoperative Assessment
- Detailed neurologic examination — document neurologic level and degree of deficit as baseline.
- Pulmonary evaluation and optimisation: particularly for high cervical lesions affecting ventilation or bulbar muscles.
- Level and timing of injury: determines autonomic dysreflexia risk and succinylcholine prohibition.
- Associated injuries and comorbidities: decubitus ulcers, urinary tract infections (common precipitants of autonomic dysreflexia).
- Ventilatory status: assess need for preoperative optimisation, physiotherapy, and whether the patient is ventilator-dependent.
- Cardiovascular evaluation: orthostatic hypotension, chronic dysrhythmias.
"The level and timing of spinal cord injuries, degree of paralysis, and associated injuries guide the preoperative evaluation." — Miller's, p. 4354
Autonomic Dysreflexia (AD)
Definition & Mechanism
- Noxious stimulus below injury level (bladder distension, rectal stimulation, surgical incision, catheterisation, decubitus ulcer, fracture)
- Afferent signals ascend to the level of injury but cannot reach supraspinal inhibitory centres
- Massive, unmodulated sympathetic discharge below the injury level → peripheral vasoconstriction and hypertension
- Baroreceptors in the carotid/aortic arch detect hypertension → parasympathetic response (bradycardia, flushing, nasal congestion) above the injury level
- The sympathetic storm cannot be inhibited by descending pathways (blocked by the injury)
- Result: Severe hypertension (may exceed 250–300 mmHg systolic) + bradycardia
"Autonomic dysreflexia is common in patients with lesions above the mid-thoracic level, but can occur with injuries at any level. Even if patients are insensate, they require anaesthesia to block noxious stimuli with surgeries and even urinary tract catheterisations to avoid severe haemodynamic perturbations." — Miller's, p. 4359
Trigger Stimuli (common perioperative)
- Most common: bladder distension or catheterisation
- Surgical skin incision below injury level
- Rectal distension or stimulation
- Decubitus ulcer debridement
- Fracture manipulation
- Tourniquet inflation
- Childbirth
Clinical Features of Autonomic Dysreflexia
- Below injury level: pallor, piloerection, vasoconstriction
- Above injury level: severe pounding headache, facial flushing, profuse sweating, nasal congestion, blurred vision, bradycardia
- Blood pressure crisis — potentially fatal (hypertensive encephalopathy, ICH, MI)
Prevention and Treatment
| Step | Action |
|---|---|
| Prevention | Adequate anaesthesia — even insensate patients require GA or neuraxial block to suppress the afferent stimulus |
| Identify and remove trigger | Drain bladder, decompress rectum, remove surgical stimulus immediately |
| Positioning | Elevate head of bed (promotes orthostatic reduction) |
| Pharmacologic treatment | Nitrates (sublingual GTN, topical nitropaste, IV nitroprusside); nifedipine (10 mg sublingual/oral); labetalol; hydralazine; phentolamine |
| Epidural/spinal block | Highly effective at blocking afferent noxious stimuli — recommended approach for all elective procedures in at-risk patients |
Anaesthetic Technique for Chronic SCI (Intraoperative)
"If patients have cervical spine disease, neck manipulation must be limited and awake intubation may be needed." — Miller's, p. 4359
| Concern | Management |
|---|---|
| Autonomic dysreflexia prevention | Neuraxial anaesthesia (spinal/epidural) preferred for procedures below injury level — blocks afferent trigger and autonomic response |
| Succinylcholine | Absolutely contraindicated — risk of fatal hyperkalaemia (extrajunctional nAChR upregulation) |
| General anaesthesia (volatile/TIVA) | Used when neuraxial not possible; ensure adequate depth before any stimulating procedure |
| Intraoperative hypotension | Common — chronic sympathectomy; have vasopressors immediately available |
| Temperature regulation | Impaired below lesion level — active warming essential; poikilothermia below lesion |
| Pressure areas | Meticulous positioning — chronic SCI patients have insensate skin and poor tissue perfusion; pressure injury occurs rapidly |
| Spina bifida/tethered cord | Neuraxial anaesthesia performed with caution; L5/S1 or lower generally does not impact spinal or epidural technique — Miller's, p. 4363 |
| Latex allergy | Patients with spina bifida and myelomeningocele may be sensitised to latex — use latex-free environment |
Neuraxial Anaesthesia in Chronic SCI
- Highly effective for autonomic dysreflexia prevention.
- A sensory block 2 segments above the injury level is generally sufficient to prevent AD.
- Technical challenges: prior surgery, scar tissue, hardware, or bone grafts may obstruct needle access and make LA spread unpredictable.
- Ultrasound guidance useful in challenging anatomy.
- If epidural placed: test dose essential; incremental dosing required.
General Anaesthesia in Chronic SCI
- All volatile agents are safe in principle.
- Depth of anaesthesia must be sufficient to suppress the sympathetic response — "awareness" in insensate patients can still trigger AD even without conscious experience.
- Processed EEG monitoring (BIS/PSI) useful to guide adequate anaesthetic depth.
- Avoid beta-blockers as primary agents for AD — may worsen bradycardia.
- Have short-acting antihypertensives (GTN, labetalol, hydralazine) immediately available throughout case.
Postoperative Management in Chronic SCI
- Pulmonary complications (PPCs) and VTE are the most common postoperative complications. — Miller's, p. 4368
- VTE prophylaxis: LMWH + mechanical compression — high risk due to immobility and impaired venous tone.
- Continued vigilance for autonomic dysreflexia — can occur in PACU from pain, urinary retention, or positioning.
- Incentive spirometry, chest physiotherapy, early mobilisation.
- Decubitus ulcer prevention — reposition frequently; appropriate pressure-relieving mattress.
- Monitor for urinary retention and ensure bladder drainage.
PART VII — NEUROSURGICAL ANAESTHETIC IMPLICATIONS
1. Spinal Decompression/Stabilisation (Acute SCI)
| Issue | Anaesthetic Implication |
|---|---|
| Cervical instability | AFOI or MILS-RSI; no neck manipulation under anaesthesia |
| Neurogenic shock | Invasive BP monitoring mandatory; vasopressors (NE/dopamine) infusion pre-positioned |
| Intraoperative MAP target | Maintain MAP >85 mmHg throughout case and for 7 days post-operatively |
| IONM (SSEPs/MEPs) | Prefer TIVA (propofol + remifentanil) — volatile agents interfere with MEPs at >0.5 MAC; avoid neuromuscular blockade during monitoring |
| Prone positioning | Careful frame positioning, protect eyes, avoid pressure on abdomen, check airway after turning |
| Blood loss | Spinal surgery can involve significant blood loss; cell salvage, TEG/ROTEM-guided transfusion |
| Hypotension | Particularly dangerous — worsens cord ischaemia; phenylephrine or norepinephrine infusion |
2. Intraoperative Neuromonitoring (IONM) — Drug Effects
| Drug | Effect on SSEPs | Effect on MEPs |
|---|---|---|
| Volatile agents (>0.5 MAC) | Reduce amplitude | Significantly suppress or abolish |
| Nitrous oxide | Reduces amplitude | Suppresses |
| Propofol (TIVA) | Minimal effect | Mild reduction only |
| Remifentanil/opioids | Minimal | Minimal |
| Ketamine | May enhance | Minimal to enhancing |
| Neuromuscular blocking drugs | No effect on SSEPs/MEPs | Abolish MEPs — avoid during monitoring periods |
3. Cervical Spine Surgery (Anterior vs. Posterior Approach)
- Risk of retractor-related airway oedema — delayed extubation may be required.
- Risk of recurrent laryngeal nerve (RLN) injury — assess voice before extubation.
- Carotid artery manipulation: baroreceptor stimulation may cause bradycardia.
- Tracheal deviation with large retractors — verify ET tube position after retractor placement.
- All prone positioning risks apply.
- Risk of cervical cord ischaemia from excess neck flexion or hypotension.
- Postoperative vision loss (POVL) risk — particularly with prolonged prone positioning, hypotension, and anaemia.
4. VTE in Neurosurgical SCI Patients
VTE prophylaxis should begin within 72 hours after SCI — administration is associated with substantially lower rates of deep vein thrombosis and pulmonary embolism. — Miller's, p. 12012
- Balance between thrombosis risk (high in SCI) and haemorrhage risk (perioperative + intradural procedures).
- ASRA/AANS guidelines specify timing intervals between anticoagulant administration and neuraxial procedures.
PART VIII — SUMMARY COMPARISON TABLE
| Feature | Acute SCI | Chronic SCI |
|---|---|---|
| Primary cardiovascular concern | Neurogenic shock (hypotension + bradycardia) | Autonomic dysreflexia (hypertension + bradycardia) |
| Airway concern | Cervical instability — avoid neck motion | Previous tracheostomy, difficult anatomy |
| Succinylcholine | Safe in first 24–48h only (emergency RSI) | Absolutely contraindicated (fatal hyperkalaemia) |
| Preferred NMB | Rocuronium/vecuronium (beyond 48h) | Rocuronium/vecuronium only |
| MAP target | >85 mmHg for 7 days | Avoid hypotension; prepare for hypertensive crises (AD) |
| Anaesthetic technique | TIVA preferred (IONM); MILS/AFOI intubation | Neuraxial preferred (AD prevention); TIVA if GA needed |
| Steroids | Not recommended (no benefit, increased harm) | Not applicable |
| Ventilation | Often required; early tracheostomy if weaning fails | Chronic ventilator dependence in high lesions |
| Fluid selection | Isotonic crystalloids; avoid hypotonic solutions | Standard; maintain euvolaemia |
| VTE | LMWH within 72h | Ongoing prophylaxis |
| Temperature | Active warming | Poikilothermia below lesion — active warming essential |
| Positioning | Careful spine immobilisation | Meticulous pressure care — insensate skin |
| Postop complications | Haemodynamic instability, respiratory failure | PPCs, VTE, AD in PACU, decubitus ulcers |
Using millers anaesthesiology book teach in detail neuroanaesthetic management, classification, scores, scales, and implications in posterior fossa surgery, SAH surgery, Aneurysm surgery, AV Malformation surgery
Neuroanaesthetic Management: Posterior Fossa, SAH, Aneurysm, and AVM Surgery
PART I — FOUNDATIONAL NEUROANAESTHESIA PRINCIPLES
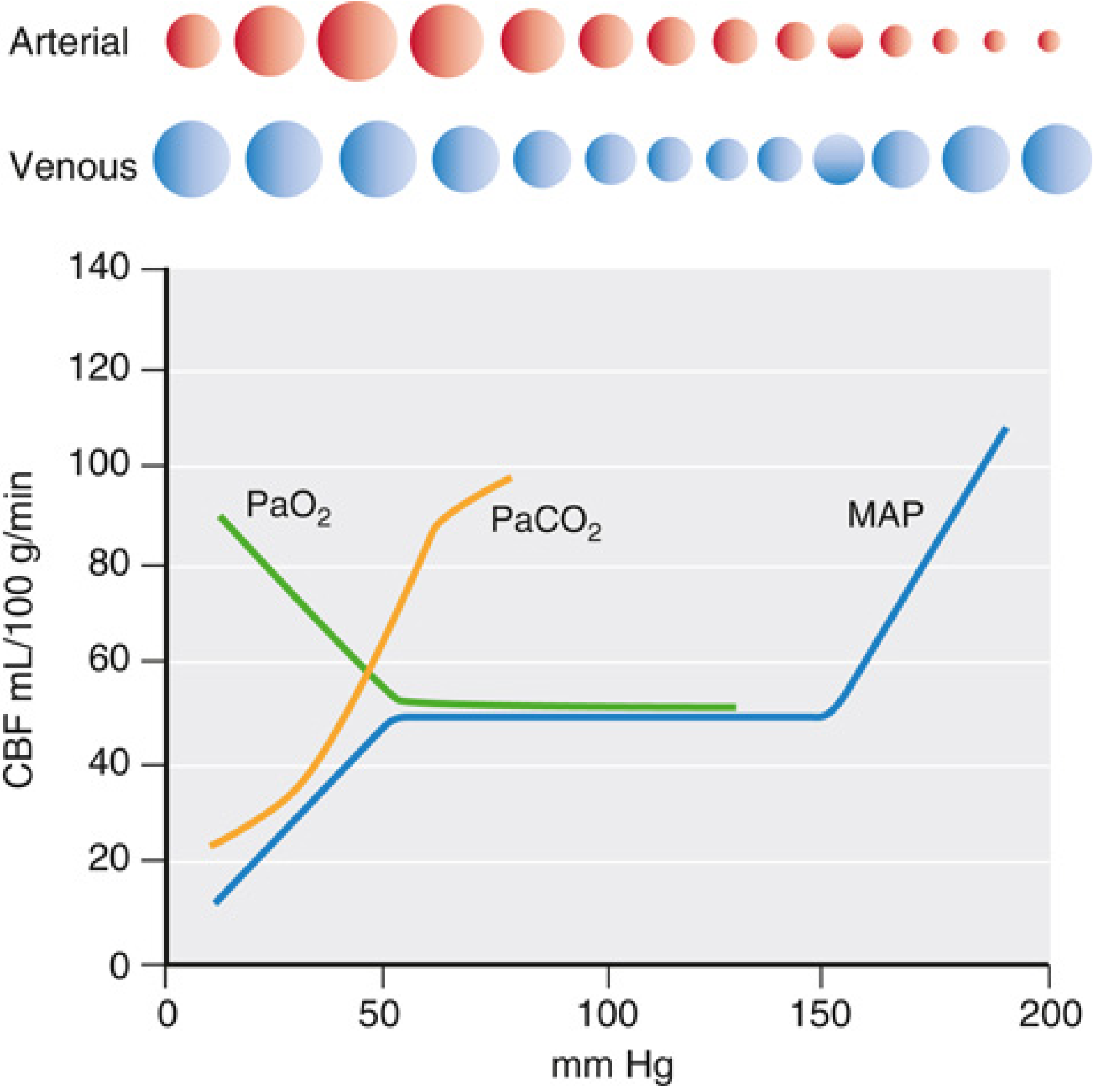
1. Cerebral Physiology — The Anaesthetist's Framework
| CBF (mL/100g/min) | Consequence |
|---|---|
| 50 | Normal |
| 20 | EEG slowing |
| 15 | Isoelectric EEG |
| 6–15 | Ischaemic penumbra (salvageable) |
| <6 | Neuronal death |
- Hypercapnia/hypoxia → cerebral vasodilation → ↑CBV → ↑ICP → ↓CPP → further CO₂ and O₂ derangement
- Hypotension → ↓CPP → vasodilation → ↑CBV → ↑ICP → further ↓CPP
"A reduction of cardiac output and CPP must be avoided to prevent further deterioration of the level of consciousness, which in turn leads to airway compromise and hypercapnia and hypoxia — perpetuating the vicious circle." — Miller's, p. 11974
2. Core Neuroanaesthetic Goals
| Goal | Target |
|---|---|
| ICP | <22 mmHg |
| CPP (CPP = MAP − ICP) | 50–70 mmHg |
| MAP | Within 10% of awake baseline (most surgical cases); >85 mmHg post-SCI/SAH |
| PaCO₂ | 35–40 mmHg (normocapnia); 30–35 mmHg mild hyperventilation only for acute ICP crises |
| PaO₂ | >60 mmHg (avoid hypoxia absolutely) |
| Glucose | Normoglycaemia (avoid hyperglycaemia) |
| Temperature | Normothermia (avoid hyperthermia); mild hypothermia selectively used |
3. Effects of Anaesthetic Agents on Cerebral Physiology
Intravenous Agents
- Propofol, barbiturates, benzodiazepines, opioids, etomidate: All reduce CMR and CBF. CBF–CMR coupling is generally preserved. Propofol and thiopental are the agents of choice for high ICP situations. — Miller's, p. 8125
- Ketamine: Once contraindicated for high ICP states; now considered acceptable when combined with appropriate agents.
Volatile Agents
- All volatile agents are dose-dependent cerebral vasodilators in isolation.
- Order of vasodilatory potency: Halothane > enflurane > desflurane > isoflurane > sevoflurane.
- At <1 MAC combined with hyperventilation, isoflurane and sevoflurane have minimal net effect on ICP.
- TIVA (propofol + remifentanil) is preferred when ICP is persistently elevated, the surgical field is tight, or robust IONM is required. — Miller's, p. 8125
Nitrous Oxide (N₂O)
- A cerebral vasodilator; effect is greatest as a sole agent.
- N₂O is not absolutely contraindicated but must be used with caution in neurosurgery.
- Discontinue N₂O before dural closure in posterior fossa and other craniotomies — risk of contributing to tension pneumocephalus once intracranial space is sealed. — Miller's, p. 8152
4. Brain Relaxation — Methods
| Method | Mechanism | Notes |
|---|---|---|
| Normoventilation/mild hyperventilation | ↓PaCO₂ → cerebral vasoconstriction → ↓CBV | Use sparingly — risk of ischaemia at PaCO₂ <30 |
| Mannitol | Osmotic diuresis, rheological | 0.5–1 g/kg IV; most widely used |
| Hypertonic saline | Osmotic; reduces oedema | 3% NaCl; preferred in hypovolaemia; avoids rebound ICP |
| Head-up 15–30° | ↑Venous drainage | Verify venous return not compromised |
| TIVA (propofol) | ↓CMR, vasoconstriction | Preferred when volatile agents worsen tightness |
| CSF drainage | Lumbar drain/ventricular drain | Used for brain relaxation in aneurysm surgery |
| Dexamethasone | Reduces tumour oedema | Pre-operatively for 24–48h; NOT for TBI/SAH oedema |
PART II — CLASSIFICATION SCALES AND SCORES
1. Glasgow Coma Scale (GCS) — Table 80.2
| Category | Response | Score |
|---|---|---|
| Eye Opening | Spontaneous | 4 |
| To voice | 3 | |
| To pain | 2 | |
| None | 1 | |
| Verbal | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Sounds | 2 | |
| None | 1 | |
| Motor | Obeys commands | 6 |
| Localises | 5 | |
| Withdraws | 4 | |
| Abnormal flexion | 3 | |
| Extension | 2 | |
| None | 1 |
2. World Federation of Neurological Surgeons (WFNS) Scale — Table 53.3 / 80.3
| Grade | GCS Score | Motor Deficit |
|---|---|---|
| I | 15 | Absent |
| II | 13–14 | Absent |
| III | 13–14 | Present |
| IV | 7–12 | Present or absent |
| V | 3–6 | Present or absent |
3. Hunt–Hess Scale — Table 80.4 / 53.3
| Grade | Clinical Description |
|---|---|
| I | Asymptomatic or mild headache, slight nuchal rigidity |
| II | Moderate to severe headache, nuchal rigidity; no neurological deficit except CN palsy |
| III | Drowsy, confused, mild focal deficit |
| IV | Stuporous, moderate to severe hemiparesis, early decerebrate rigidity |
| V | Deep coma, decerebrate rigidity, moribund appearance |
"Higher Hunt and Hess and WFNSS grades are associated with worse clinical outcomes." — Miller's, p. 12014
4. Modified Fisher Scale (CT Appearance — SAH) — Table 80.9
| Grade | CT Appearance |
|---|---|
| 0 | No blood detected |
| 1 | Thin subarachnoid blood, no intraventricular haemorrhage (IVH) |
| 2 | Thin subarachnoid blood, with IVH |
| 3 | Thick subarachnoid blood (>1mm), no IVH |
| 4 | Thick subarachnoid blood, with IVH |
5. Marshall CT Classification and Rotterdam CT Score (TBI)
6. Spetzler–Martin AVM Grading Scale
- Size: Small (<3 cm) = 1; Medium (3–6 cm) = 2; Large (>6 cm) = 3
- Eloquence of adjacent brain: Non-eloquent = 0; Eloquent = 1
- Venous drainage pattern: Superficial only = 0; Deep = 1
PART III — POSTERIOR FOSSA SURGERY
1. Anatomy and Surgical Significance
"The posterior fossa is a narrow space around the brainstem that contains the cerebellum, ascending and descending sensorimotor pathways, cranial nerve nuclei, cardiorespiratory centres, reticular activating system, and the neural networks that underlie crucial protective reflexes such as eyeblink, swallowing, gag, and cough." — Miller's, p. 5264
"Surgery within the posterior fossa is therefore considered highly risky, and even small injuries can leave significant neurological deficits." — Miller's, p. 5264
- Disorders of consciousness
- Impaired respiratory drive
- Cardiovascular dysfunction
- Life-threatening brainstem compression
- Microvascular decompression (MVD) of CNs V, VII, IX (trigeminal neuralgia, hemifacial spasm, glossopharyngeal neuralgia)
- Vestibular nerve schwannoma (acoustic neuroma) resection
- Posterior fossa tumours (medulloblastoma, ependymoma, haemangioblastoma)
- Cerebellar and brainstem tumour resection
- Fourth ventricle surgery
2. Positioning — The Sitting Position
| Complication | Details |
|---|---|
| Venous Air Embolism (VAE) | Incidence detectable by precordial Doppler: ~40%; by TEE: up to 76% in sitting posterior fossa cases. Reduced to ~12% in non-sitting positions. — Miller's, p. 8156 |
| Paradoxical Air Embolism (PAE) | Air crosses interatrial septum via patent foramen ovale (PFO — present in ~25% adults) → systemic arterial embolism → stroke, myocardial ischaemia |
| Hypotension | Venous pooling in lower limbs; reduced cardiac preload |
| Pneumocephalus | Air enters supratentorial space during open craniotomy in head-up position |
| Macroglossia | Prolonged neck flexion → venous/lymphatic obstruction of tongue |
| Quadriplegia | Neck flexion causing cervical cord stretch/compression, particularly with osteophytes |
Alternative Positions (Reduce VAE Risk)
- Lateral (park bench) position: Good exposure, lower VAE risk
- Prone (concorde) position: Used for midline posterior fossa lesions
- Three-quarter prone: Compromise between lateral and prone
3. Venous Air Embolism (VAE) — Detailed Management
- Major cerebral venous sinuses (transverse, sigmoid, posterior sagittal — noncollapsible due to dural attachments)
- Emissary veins from suboccipital musculature
- Diploic space of skull (craniotomy + pin fixation)
- Cervical epidural veins
Detection — in order of sensitivity:
| Monitor | Sensitivity | Notes |
|---|---|---|
| Transoesophageal echocardiography (TEE) | Highest | Also detects PFO and right-to-left shunting; safety in prolonged use with neck flexion not established |
| Precordial Doppler | High | Standard practice at left/right parasternal 2nd–4th intercostal space; characteristic "mill-wheel" murmur |
| End-tidal CO₂ (ETCO₂) | Moderate | Sudden fall in ETCO₂ indicates reduced pulmonary blood flow from VAE |
| End-tidal N₂ | Low-moderate | Theoretically attractive but limited sensitivity except in catastrophic events |
| PA pressure, CVP | Indirect | Rise indicates haemodynamic compromise |
Standard practice: Precordial Doppler + ETCO₂ monitoring in combination. TEE is more sensitive and identifies right-to-left shunting but has practical limitations. — Miller's, p. 8157
Treatment Protocol for VAE (from Miller's):
- Notify surgeon — flood surgical field with saline, apply bone wax/pressure
- Jugular vein compression — increases venous back-pressure
- Lower the head (reduce venous-atmospheric pressure gradient)
- Aspirate right heart catheter (CVP or PA catheter) — multi-orificed catheter positioned 2 cm below SVC-atrial junction
- Discontinue N₂O immediately (prevents expansion of gas bubble)
- FiO₂ 1.0 (wash out N₂O; optimise oxygenation)
- Vasopressors/inotropes (support cardiac output)
- Chest compressions if haemodynamic collapse
- Multi-orificed catheter tip: 2 cm below SVC–atrial junction
- Single-orificed catheter: 3 cm above SVC–atrial junction
- Confirm by: radiography, intravascular ECG (biphasic P wave = intra-atrial position), or TEE
4. Pneumocephalus
- Air accumulates in the supratentorial space when the cranium is in a head-up position and intracranial volume is reduced (by hypocapnia, osmotic diuresis, CSF drainage, good venous drainage).
- When the patient returns to supine, CSF and venous blood reaccumulate and the air pocket becomes a mass lesion (nitrogen diffuses very slowly).
- Tension pneumocephalus: causes delayed awakening, severe headache, neurological deterioration.
- N₂O must be discontinued before dural closure in head-up posterior fossa procedures to prevent expansion of trapped gas.
- Diagnosis: brow-up lateral skull radiograph. — Miller's, p. 8152–8153
5. Brainstem Stimulation — Cardiovascular Responses
"Irritation of the lower pons and upper medulla... can result in several cardiovascular responses... bradycardia and hypotension, tachycardia and hypertension, or bradycardia and hypertension, and ventricular dysrhythmias." — Miller's, p. 8228
- Floor of fourth ventricle surgery
- Cerebellopontine angle surgery (acoustic neuromas, MVD of CNs V, VII, IX)
- Meticulous ECG monitoring and directly transduced arterial pressure must be maintained continuously during brainstem manipulation.
- Alert the surgeon immediately — the haemodynamic changes warn of adjacent cranial nerve nuclei and respiratory centre damage.
- Pharmacologically suppressing these warning dysrhythmias may eliminate the very signs that should prompt the surgeon to pause.
6. Intraoperative Neurophysiological Monitoring (IONM) in Posterior Fossa Surgery
| Modality | What It Monitors | Use in Posterior Fossa |
|---|---|---|
| BAEP (Brainstem Auditory Evoked Potentials) | CN VIII integrity; cochlear nerve | MVD for trigeminal neuralgia/hemifacial spasm; acoustic neuroma — increases chance of preserved hearing |
| Spontaneous EMG | Facial nerve, lower cranial nerves | Neurotonic discharges warn of impending stretch/compression injury |
| Stimulated EMG (lateral spread response) | Facial nerve decompression adequacy | Elimination of LSR in hemifacial spasm surgery confirms adequate decompression |
| SSEPs | Dorsal column–medial lemniscal pathway | Somatosensory pathway integrity |
| MEPs | Corticospinal tract | Motor pathway integrity |
- Neuromuscular blocking drugs: Abolish EMG responses — AVOID during EMG monitoring periods for cranial nerve preservation. Even sharp sectioning of a nerve may produce no EMG discharge.
- Volatile agents: Dose-dependently suppress MEPs and SSEPs — keep at ≤0.5 MAC; prefer TIVA.
- TIVA (propofol + remifentanil): Minimal interference with evoked potentials; gold standard for IONM cases.
7. Extubation Decisions After Posterior Fossa Surgery
"Irritation and injury of posterior fossa structures... should be considered in planning extubation and postoperative care." — Miller's, p. 8228
- Dissection on the floor of the fourth ventricle → risk of CN IX, X, XII dysfunction (loss of upper airway control, swallowing)
- Brainstem swelling → impaired respiratory drive and cardiac function
- A relatively small amount of swelling can cause life-threatening decompensation — the posterior fossa has very limited compensatory reserve compared to the supratentorial space.
- Was the floor of the fourth ventricle dissected?
- Are CN IX, X, XII functions intact (gag, swallow, tongue movement)?
- Is there brainstem oedema on intraoperative assessment?
- Is the patient responsive and following commands?
- Will postoperative monitoring be in ICU or standard ward?
PART IV — SUBARACHNOID HAEMORRHAGE (SAH)
1. Epidemiology and Risk Factors
- Incidence of aneurysmal SAH (aSAH): 6.1 per 100,000 person-years worldwide.
- Women > men (1.3-fold relative risk); most common after age 55.
- Case fatality has declined from 50% to 33% with modern endovascular techniques and ICU management.
- In-hospital mortality: 13%; Pre-hospital mortality: 26%. — Miller's, p. 12013
2. Pathophysiology of SAH — Early Brain Injury
- Transient global ischaemia (ICP spike at time of bleed may transiently equal MAP → global cerebral ischaemia)
- Elevated ICP (from haematoma, hydrocephalus)
- SAH toxicity — blood breakdown products directly injure neurons
- Microcirculatory changes, cerebral oedema, sympathetic surge
"The degree of neurological dysfunction and the amount of bleeding are the strongest predictors of clinical outcome." — Miller's, p. 12014
3. Complications of SAH
| Complication | Timing | Details |
|---|---|---|
| Rebleeding | First 24h (peak) | Mortality up to 70% if rebleed occurs. Most feared early complication |
| Acute hydrocephalus | First 24–72h | Develops in 15–87% of SAH patients; emergency CSF diversion (ventriculostomy/lumbar drain) may be lifesaving |
| Cerebral vasospasm | Day 4–14 | Delayed cerebral ischaemia (DCI); most common between days 7–10 |
| Hyponatraemia | First week | SIADH vs. cerebral salt wasting (different management) |
| Cardiac complications | Acute | ECG changes, Takotsubo cardiomyopathy, arrhythmias, troponin rise from catecholamine surge |
| Seizures | Acute/subacute | 7-day course anti-epileptics; avoid phenytoin |
4. Delayed Cerebral Ischaemia (DCI) and Vasospasm
- Vasospasm monitoring: TCD ultrasonography (Lindegaard index — MCA:ICA ratio; >3 = vasospasm), CTA, cerebral angiography, cEEG, invasive monitoring.
- Medical management of DCI: Augment arterial BP with vasopressors + goal-directed euvolaemia (not hypervolaemia).
- "Triple-H therapy" (hypertension–hypervolaemia–haemodilution): No longer recommended — no evidence of benefit and potential harm from RCTs. — Miller's, p. 12420
- Nimodipine: Level I evidence (RCT proven) — started as soon as possible after diagnosis, continued for 21 days. Reduces ischaemic deficit (modest effect); does not prevent angiographic vasospasm. — Miller's, p. 12412
- Endovascular therapy (balloon angioplasty, intra-arterial nicardipine/verapamil): For vasospasm refractory to medical management. — Miller's, p. 12414
5. Critical Care Management of SAH
- Acute pre-operative: No specific BP target for rebleeding prevention; avoid hypotension and BP variability.
- Post-operative: Individualised; nimodipine titration.
PART V — CEREBRAL ANEURYSM SURGERY
1. Overview and Timing of Intervention
"Contemporary management calls for intervention as early as feasible to reduce the rate of rebleeding." — Miller's, p. 8186
- Sooner clipping/coiling = less likelihood of rebleeding (principal cause of death post-SAH).
- Management of vasospasm (hypervolaemia + induced hypertension) can be given safely after aneurysm is secured.
- Avoids bed rest risk (DVT, pulmonary complications).
- Preferred: Within 24 hours of SAH. — Miller's, p. 12315
- If early intervention not feasible (poor grade, medical instability): Surgery may be delayed to 10–14 days — beyond the peak vasospasm period (days 4–10). — Miller's, p. 8186
- Endovascular coiling (ISAT data): Preferred when aneurysm anatomy is favourable; associated with higher odds of favourable outcome at 1 year.
- Surgical clipping: Preferred for anatomically complex aneurysms, wide-neck, large MCA aneurysms, those with associated haematoma requiring evacuation.
2. Preoperative Assessment for Aneurysm Surgery
| Parameter | Consideration |
|---|---|
| Neurological grade | WFNS/Hunt-Hess grade; guides urgency and induction technique |
| ICP status | Hydrocephalus? Ventriculostomy present? |
| Cardiac status | ECG (ST changes, deep T-wave inversions — "canyon T-waves"), echo (wall motion, Takotsubo) |
| Electrolytes | Hyponatraemia common; correct before GA |
| Medications | Nimodipine (vasodilatory effects under GA); anti-seizure drugs |
| Volume status | SAH patients are frequently hypovolaemic |
| Haematological | Coagulation — antifibrinolytics if used |
3. Anaesthetic Induction — Key Principle
"The prevention of paroxysmal hypertension is the only absolute requirement in patients undergoing aneurysm clipping." — Miller's, p. 8558
"The poorly organised clot over the aneurysms of patients undergoing early post-SAH clipping makes them particularly prone to rebleeding. A rebleed at induction is frequently fatal." — Miller's, p. 8558
- Hypertension (drives rebleeding through poorly organised clot) AND
- Hypotension (worsens ischaemia in a brain with impaired autoregulation and marginal CBF)
- Pre-induction arterial line (mandatory — avoid any unmonitored hypertension at laryngoscopy/pin placement).
- Smooth, controlled induction with propofol or thiopental + opioid (fentanyl/remifentanil) to blunt laryngoscopy response.
- Avoid rapid sequence induction if possible — risk of hypertensive surge.
- Lignocaine 1.5 mg/kg IV or esmolol to blunt laryngoscopy response.
- Maintain MAP at or slightly below the patient's awake baseline throughout induction.
- Vecuronium or rocuronium for intubation (not succinylcholine unless emergency).
4. Monitoring
- Invasive arterial line (radial or femoral): Pre-induction — mandatory.
- Central venous catheter: For vasopressor administration and right atrial access (for VAE aspiration if applicable).
- Precordial Doppler ± TEE: Particularly if sitting position used (rare for aneurysm surgery but relevant for posterior communicating artery approaches).
- SSEPs/MEPs: For monitoring during temporary clipping and dissection.
- Processed EEG (BIS): For depth of anaesthesia; can guide burst suppression induction if neuroprotection is desired during temporary occlusion.
- Temperature: Maintain normothermia (see hypothermia section below).
- Urinary catheter + glucose monitoring: Mannitol/diuretic use; tight glycaemic control.
5. Anaesthetic Maintenance
"Any technique that permits proper control of MAP is acceptable. However, in the face of increased ICP or a tight surgical field, an inhaled anaesthetic technique may be less suitable." — Miller's, p. 8558
| Situation | Preferred Technique |
|---|---|
| Normal ICP, elective | Balanced volatile (≤1 MAC) + opioid + air/O₂ |
| Elevated ICP, tight brain | TIVA (propofol + remifentanil) |
| IONM required (MEPs) | TIVA — volatile agents suppress MEPs |
| Ruptured aneurysm, early surgery | TIVA preferred — avoids vasodilation from volatile |
6. Intraoperative Blood Pressure Management
- Maintain MAP at or near patient's awake baseline.
- Avoid hypotension — autoregulation is impaired, low resting CBF; modest hypotension can cause ischaemia.
- Avoid hypertension — risk of aneurysm rupture.
- Induce relative hypertension (augment collateral CBF).
- Phenylephrine or norepinephrine.
- Confirm adequate clip placement (surgeon may puncture dome and request SBP of 150 mmHg).
- Manage vasospasm prophylaxis with nimodipine.
7. Temporary Clipping — Anaesthetic Implications
- Facilitate safe clip placement (deflates the aneurysm dome)
- Manage intraoperative rupture
- Induce relative hypertension to augment collateral CBF via leptomeningeal anastomoses.
- Cerebral metabolic protection with burst suppression (propofol infusion, thiopental bolus, or etomidate) — reduces metabolic demand during ischaemia.
- Limit occlusion time — ideally < 10 minutes.
- SSEPs/MEPs monitoring to detect ischaemia.
8. Intraoperative Rupture
- Surgeons apply temporary clips immediately
- Anaesthesiologist: Lower MAP rapidly to facilitate haemostasis (MAP 40–50 mmHg may be requested)
- This is very difficult in a hypovolaemic patient
- Maintain normovolaemia before rupture occurs — Miller's advocates normovolaemia
- Rapidly available hypotensive agents (adenosine bolus [for brief cardiac standstill] or esmolol or sodium nitroprusside)
- Blood transfusion readiness
- Intensify neurological monitoring
9. Hypothermia in Aneurysm Surgery
"An international multicenter trial of mild hypothermia in 1001 relatively good-grade patients undergoing aneurysm surgery revealed no improvement in neurologic outcome." — Miller's, p. 8177
- Risk of cardiac dysrhythmia and coagulation dysfunction if temperature too low.
- Rewarm adequately before emergence to avoid shivering, hypertension, or delayed awakening.
- Temperature monitoring: oesophageal, tympanic, pulmonary arterial, and jugular bulb — all reflect deep brain temperature well. Bladder temperature does NOT.
10. Emergence from Anaesthesia — Aneurysm Surgery
"Most practitioners of neuroanesthesia place a premium on a smooth emergence; that is, one free of coughing, straining, and arterial hypertension." — Miller's, p. 8325
- Avoid hypertension at emergence (risk of intracranial bleeding)
- Permit rapid neurological assessment
- Avoid coughing/straining (Valsalva → ↑ICP → venous bleeding)
- Remifentanil infusion continued to extubation point for blunted response.
- Lignocaine 1.5 mg/kg IV before tracheal extubation.
- Dexmedetomidine infusion for smooth emergence.
- Labetalol or esmolol to control hypertension.
- Consider deep extubation (selected cases) vs. awake extubation with pharmacological blunting.
PART VI — ARTERIOVENOUS MALFORMATION (AVM) SURGERY
1. Classification — Spetzler–Martin Grading Scale
| Feature | Points |
|---|---|
| Size | |
| Small (<3 cm) | 1 |
| Medium (3–6 cm) | 2 |
| Large (>6 cm) | 3 |
| Eloquence of adjacent brain | |
| Non-eloquent area | 0 |
| Eloquent area (sensorimotor, language, visual cortex, thalamus, hypothalamus, brainstem, cerebellar nuclei, deep cerebellar white matter) | 1 |
| Pattern of venous drainage | |
| Superficial only | 0 |
| Any deep component | 1 |
- Grade 1–2: Low surgical risk; surgery favoured.
- Grade 3: Intermediate; individualise.
- Grade 4–5: High surgical risk; endovascular, radiosurgery, or conservative management often preferred.
2. AVM Pathophysiology — Anaesthetic Implications
-
"Normal perfusion pressure breakthrough" (NPPB): After AVM resection, the previously high-flow, low-resistance AVM nidus is removed. Surrounding brain tissue, which was chronically hypoperfused due to "steal" from the AVM, now receives normal arterial pressure — but its autoregulation is impaired (functionally exhausted from chronic exposure to low pressures). The result is breakthrough oedema, hyperaemia, and haemorrhage.
- Prevention: Tight BP control post-resection; avoid hypertension.
-
"Steal phenomenon": Blood is diverted from normal brain to the low-resistance AVM nidus. Hypotension worsens steal and worsens surrounding brain ischaemia.
-
Venous outflow pressure: Draining veins are under arterial pressure; premature ligation of draining veins (before nidus obliteration) causes catastrophic haemorrhage and oedema.
3. Anaesthetic Management of AVM Surgery
- Review Spetzler–Martin grade.
- Understand angioarchitecture (feeding arteries, draining veins, relationship to eloquent cortex).
- Review any prior embolisation procedures — staged treatment; residual AVM anatomy.
- Check electrolytes, coagulation, blood type and crossmatch.
- Pre-operative steroids if significant surrounding oedema.
- Continuous invasive arterial line: Pre-induction.
- IONM (SSEPs/MEPs/EEG): Particularly important for eloquent cortex AVMs.
- Awake craniotomy: May be chosen for AVMs adjacent to language cortex — allows real-time language mapping during resection.
- Smooth induction with propofol/thiopental + opioid — avoid hypertension.
- Maintain MAP at or below awake baseline.
- TIVA preferred (particularly for IONM, or tight BP control).
- Avoid volatile agents if MEPs required.
- Maintain normocapnia (PaCO₂ 35–40 mmHg) — hypercapnia worsens steal.
| Phase | BP Goal | Rationale |
|---|---|---|
| Before nidus obliteration | Slightly below baseline MAP | Reduce transmural pressure on AVM; reduce bleeding |
| During temporary vessel occlusion | Relatively higher MAP | Augment collateral flow |
| After nidus obliteration | Strict MAP control — below pre-operative baseline | Prevent NPPB; avoid hyperaemic breakthrough haemorrhage |
"Hypothermia... use in the management of... arteriovenous malformations (AVMs) became widespread. However, an international multicenter trial... revealed no improvement in neurologic outcome." — Miller's, p. 8177 (hypothermia no longer routinely used)
- Mannitol (0.5–1 g/kg).
- Head-up positioning.
- Moderate hypocapnia (PaCO₂ 30–35) only when needed for brain relaxation (not routine).
- ICU admission mandatory.
- Tight BP control (target BP below pre-operative baseline) for 24–48 hours to prevent NPPB.
- Neurological monitoring: assess for breakthrough bleeding (sudden ↑BP, altered consciousness, worsening neurological deficit).
- Continue IONM/EEG monitoring in ICU.
- Avoid factors that increase cerebral perfusion pressure (pain, agitation, hypertension, hypercapnia).
PART VII — GENERAL NEUROANAESTHETIC CONSIDERATIONS ACROSS ALL PROCEDURES
Preoperative Preparation
- Steroids: For tumour-related oedema — dexamethasone 10 mg IV then 10 mg 6-hourly, ideally for 48 hours pre-operatively. NOT indicated for TBI or SAH oedema. — Miller's, p. 8186, 8137
- Sedative premedication: Usually avoided outside the OR — CO₂ retention in patients with abnormal ICP compliance. — Miller's, p. 8401
Monitoring
- Invasive arterial line: near-universally placed for craniotomies; preinduction for high-risk patients (SAH, mass effect, poor compliance).
- Central venous catheter: when blood loss potential is high or peripheral access limited.
- ICP monitoring: rarely warranted at induction — once cranium is open, the surgical field directly reveals intracranial compliance. — Miller's, p. 8406
Osmotherapy
- Mechanism: osmotic diuresis (draws water from brain cells via osmotic gradient) + rheological effect (reduces blood viscosity, improves microcirculation).
- Dose: 0.5–1 g/kg IV.
- Onset: 15–20 minutes.
- Risk: hypovolaemia (monitor urine output and electrolytes).
- Preferred in hypovolaemic patients.
- Does not cause systemic diuresis.
- Reduces ICP via osmotic shift across blood–brain barrier.
- Maintains or expands intravascular volume.
Glucose Management
- Avoid hyperglycaemia: Worsens ischaemic injury.
- Avoid hypoglycaemia: Brain has no glycogen stores.
- Target: normoglycaemia (4–10 mmol/L) with insulin infusion if needed.
Temperature Management
- Avoid hyperthermia: Even modest hyperthermia significantly worsens outcomes after ischaemic brain injury.
- Active forced-air warming to prevent hypothermia.
- If therapeutic hypothermia used (selected cases): target 32–34°C; monitor with oesophageal/tympanic thermometer (not bladder).
Summary Comparison Table
| Feature | Posterior Fossa | SAH (acute) | Aneurysm Surgery | AVM Surgery |
|---|---|---|---|---|
| Key classification | Location, Spetzler–Martin (if vascular) | Hunt-Hess, WFNS, Fisher | WFNS, Hunt-Hess | Spetzler–Martin Grade 1–5 |
| Primary surgical risk | Brainstem injury, CN damage, VAE | Rebleeding | Intraoperative rupture | NPPB, haemorrhage, steal |
| Position concern | Sitting → VAE, PAE, macroglossia | Supine/lateral | Supine/lateral | Supine/lateral/park bench |
| BP at induction | Near-normal | Avoid hypertension AND hypotension | Normotensive (no spike) | Slightly below baseline |
| BP during surgery | Maintain CPP | Avoid hypotension | Near-baseline; augment for temporary clipping | Below baseline; strict post-resection |
| IONM | BAEP, EMG (CN VII, IX, X, XII), SSEPs, MEPs | SSEPs/MEPs | SSEPs/MEPs | SSEPs/MEPs/EEG; awake craniotomy possible |
| Preferred technique | TIVA for IONM; balanced volatile acceptable | TIVA preferred | TIVA; balanced volatile if ICP normal | TIVA for IONM + BP control |
| N₂O | Discontinue before dural closure | Avoid | Avoid | Avoid |
| Brain relaxation | Mannitol + positioning | Mannitol + normoventilation + CSF drain | Mannitol + CSF drain + normoventilation | Mannitol + normoventilation |
| Hypothermia | Not routine | Not routine | Not routine (no RCT benefit) | Not routine |
| Steroids | If tumour/oedema | Not indicated | Not indicated | If surrounding oedema |
| Extubation | Delayed if CN or brainstem risk | ICU — assess before extubation | Smooth emergence; ICU | ICU — tight BP control post-op |
| Critical postop concern | Brainstem oedema; CN dysfunction | Vasospasm (Day 4–14), DCI | Vasospasm, rebleeding | Normal perfusion pressure breakthrough |
Using millers anaesthesiology book discuss in detail neuromuscular blocking agents, neuromuscular monitoring, residual neuromuscular blockade, and most important concepts
Neuromuscular Blocking Agents, Monitoring, Residual Block, and Key Concepts
PART I — HISTORY AND INTRODUCTION
- Clinical pharmacology of NMBDs
- Residual neuromuscular block postoperatively (not yet recognised)
- Importance of pharmacologically antagonising residual block
- Guidelines for monitoring muscle strength
Adverse reactions: NMBDs account for 10.8% of adverse drug reactions and 7.3% of deaths during anaesthesia (Committee on Safety of Medicines, UK). — Miller's, p. 3293
PART II — PHYSIOLOGY OF THE NEUROMUSCULAR JUNCTION
1. Anatomy of the NMJ
- Presynaptic (motor nerve) terminal: synthesises and stores acetylcholine (ACh) in quantal vesicles
- Synaptic cleft: site of ACh diffusion
- Postsynaptic (muscle end-plate) membrane: contains nicotinic acetylcholine receptors (nAChRs)
2. Nicotinic Acetylcholine Receptors (nAChRs)
| Receptor Type | Subunit Composition | Location | Properties |
|---|---|---|---|
| Adult (mature) nAChR | α₂βεδ (αεαδβ arrangement) | Postsynaptic end-plate | Short opening time; high conductance (Na⁺, K⁺, Ca²⁺) |
| Fetal (immature) nAChR | α₂βγδ (γ subunit instead of ε) | Extrasynaptic (perijunctional, extrajunctional) | Longer open-channel time; lower conductance |
| Presynaptic neuronal nAChR | α₃β₂ subtype | Prejunctional motor nerve | Positive feedback — mobilises ACh reserve; mediates fade phenomenon |
3. Mechanism of Neuromuscular Transmission
- Nerve action potential arrives → depolarises presynaptic terminal
- Voltage-gated Ca²⁺ channels open → Ca²⁺ influx
- ACh vesicles fuse with membrane → ACh released into synaptic cleft (quantal release)
- ACh binds to both α subunits of postsynaptic nAChR → ion channel opens → Na⁺ influx (+ K⁺ efflux) → end-plate depolarisation (end-plate potential, EPP)
- EPP triggers action potential in muscle fibre → muscle contraction
- ACh hydrolysed by acetylcholinesterase (AChE) in synaptic cleft → terminated signal
4. Presynaptic (Prejunctional) Effects of NMBDs
- Presynaptic nicotinic (α₃β₂) receptors serve as a positive-feedback loop: during high-frequency stimulation, they mobilise ACh from the reserve store to maintain availability.
- Non-depolarising NMBDs block α₃β₂ presynaptic receptors → impaired mobilisation of ACh → decreased ACh available for release during repetitive stimulation → FADE (progressive reduction in twitch height during repeated stimulation). — Miller's, p. 3227
- Succinylcholine does NOT block presynaptic α₃β₂ receptors → no fade with depolarising block (in clinical doses).
"The action of nondepolarizing versus depolarizing NMBDs at this neuronal cholinergic receptor explains the typical fade phenomenon after any nondepolarizing drugs, and the lack of such effect in the clinical dose range for succinylcholine." — Miller's, p. 3227
PART III — PHARMACOLOGY OF DEPOLARISING NMBDs
Succinylcholine (Suxamethonium)
| Property | Detail |
|---|---|
| Structure | Two ACh molecules joined end-to-end |
| Mechanism | Binds and activates both α subunits of nAChR → prolonged depolarisation (Phase I block); cannot be hydrolysed by AChE; hydrolysed rapidly by butyrylcholinesterase (pseudocholinesterase) in plasma |
| Onset | 60–90 seconds at intubating dose (1.0–1.5 mg/kg) — fastest onset of all NMBDs |
| Duration | 10–13 minutes (ultra-short) |
| RSI dose | 1.0–1.5 mg/kg IV |
| Paediatric dose | 2 mg/kg (infants); 1–1.5 mg/kg (children) |
Mechanism of Block — Phase I and Phase II
| Phase | Description | Features |
|---|---|---|
| Phase I (Depolarising) | Receptor occupied and activated → sustained depolarisation of end-plate → voltage-gated Na⁺ channels inactivated → no propagated action potential | Fasciculations, no fade on TOF, no post-tetanic facilitation |
| Phase II (Desensitising) | After prolonged or repeated succinylcholine → receptors become desensitised/uncoupled despite depolarisation | Begins to resemble non-depolarising block; fade on TOF; may reverse with neostigmine |
Side Effects of Succinylcholine — Critical Points
| Side Effect | Mechanism | Clinical Notes |
|---|---|---|
| Hyperkalaemia | Depolarisation → K⁺ efflux from extrajunctional receptors (upregulated in burns, SCI, denervation, immobilisation) | Normal rise: 0.5 mEq/L; in pathological states: 5–10 mEq/L → cardiac arrest |
| Bradycardia | Muscarinic activation at cardiac SA node; particularly with second dose in children | Prevented with atropine 0.02 mg/kg pre-treatment |
| Fasciculations | Generalised muscle contractions before block | Can be reduced with small defasciculating dose of non-depolarising NMBD (increases SCh dose requirement by ~30%) |
| Myalgia | Postoperative muscle pain; related to fasciculations | More common in young, muscular patients |
| Increased intraocular pressure (IOP) | Tonic contraction of extraocular muscles | Clinical relevance debated; historically avoided in open-eye injuries |
| Increased intragastric pressure | Fasciculations; offset by LOS tone increase | Net effect on aspiration risk unclear |
| Increased intracranial pressure | Small, transient; attenuated by good depth of anaesthesia | |
| Malignant hyperthermia (MH) | Triggering agent — absolute contraindication in MH susceptibility | |
| Prolonged block | Butyrylcholinesterase (BChE) deficiency — genetic variants (dibucaine number) | Dibucaine number: Normal ~80; heterozygous ~60; homozygous ~20 |
| Masseter muscle rigidity | Hallmark early sign of MH; also seen in children under halothane | |
| Cardiac arrest in children | Unmasking of Duchenne muscular dystrophy (unsuspected) → fatal hyperkalaemia, rhabdomyolysis | Routine use in children contraindicated for elective procedures — Miller's, p. 3316 |
Contraindications to Succinylcholine
- Known or suspected Malignant Hyperthermia
- Personal/family history of pseudocholinesterase deficiency
- Burns (>24–48h post-injury)
- Crush injury/rhabdomyolysis (>24–48h post-injury)
- Spinal cord injury (>48h post-injury)
- Denervation injuries (>48h post-injury)
- Prolonged immobility
- Stroke (after 24–48h)
- Neuromuscular diseases (myopathies, muscular dystrophies)
- Hyperkalaemia
- Routine use in healthy children — risk of unmasked Duchenne MD — Miller's, p. 3316
PART IV — PHARMACOLOGY OF NON-DEPOLARISING NMBDs
1. Classification
By Chemical Class
| Class | Examples |
|---|---|
| Aminosteroids | Pancuronium, vecuronium, rocuronium, pipecuronium |
| Benzylisoquinoliniums | dTc, metocurine, atracurium, cisatracurium, mivacurium, doxacurium |
By Duration of Action (Equipotent Doses)
| Duration | Agents | DT25 (Clinical Duration to 25% Recovery) |
|---|---|---|
| Long-acting | Pancuronium, pipecuronium, doxacurium | >60 minutes |
| Intermediate-acting | Vecuronium, rocuronium, atracurium, cisatracurium | 25–50 minutes |
| Short-acting | Mivacurium | 15–20 minutes |
| Ultra-short-acting | Succinylcholine | ~10–13 minutes |
2. Mechanism of Non-Depolarising Block
- Depression of single-twitch: caused mainly by postsynaptic nAChR blockade
- Fade (TOF fade, tetanic fade): caused mainly by presynaptic α₃β₂ nAChR blockade → impaired ACh mobilisation
- Progressive fade on TOF stimulation
- Post-tetanic facilitation (PTF): after tetanic stimulation, twitch response transiently enhanced due to Ca²⁺-mediated increase in ACh mobilisation
- No fasciculations before onset
- Reversed by anticholinesterases (neostigmine, edrophonium) or sugammadex
3. Potency — ED₅₀, ED₉₀, ED₉₅ and the Inverse Potency–Onset Relationship
- ED₅₀ = 50% suppression of twitch height
- ED₉₀ = 90% suppression
- ED₉₅ = 95% suppression (standard clinical reference for intubating dose = 2× ED₉₅)
"Onset time decreases as ED₅₀ increases. When a potent NMBD is administered, fewer molecules are administered than in the case of an equipotent dose of a less potent drug. Because of this lower concentration gradient, more time is required for sufficient molecules of a potent drug to be delivered to the neuromuscular junction; thus onset time is longer." — Miller's, p. 3271
| Drug | Molar Potency (ED₉₅ μM/kg) | Onset (min to max block at AP) |
|---|---|---|
| Cisatracurium | High | Slowest (~5–7 min) |
| Vecuronium | High | 3–5 min |
| Atracurium | Moderate | 3–5 min |
| Rocuronium | Low (13% of vecuronium; 9% of cisatracurium) | 1.5–2 min (fastest non-depolariser) |
| Mivacurium | Moderate | 2–3 min |
| Succinylcholine | — (depolariser) | 1 min (fastest of all) |
Rocuronium's low molar potency (ED₉₅ = 0.54 μM/kg) explains its rapid onset — more molecules delivered per dose. — Miller's, p. 3271–3272
4. Muscle Sensitivity — Onset Varies by Muscle Group
"Neuromuscular block develops faster, lasts a shorter time, and recovers faster in the more centrally located neuromuscular units (laryngeal adductors, diaphragm, masseter) than in the more peripherally located adductor pollicis muscle." — Miller's, p. 3220
| Muscle Group | Sensitivity | Clinical Implication |
|---|---|---|
| Laryngeal adductors | More resistant; faster onset/recovery | Good intubating conditions present before adductor pollicis shows full block |
| Diaphragm | More resistant than adductor pollicis | Breathing may recover while peripheral monitor still shows residual block |
| Adductor pollicis (AP) | Most sensitive; used for monitoring | Underestimates block at larynx; overestimates block at diaphragm |
| Orbicularis oculi | More sensitive than AP | Block onset faster; may overestimate intubating conditions |
5. Individual Drug Profiles
A. Long-Acting Agents
- Aminosteroid; renal elimination predominant (also hepatic: 3-OH metabolite ~50% potency).
- Cardiovascular: Vagolytic (tachycardia, hypertension) — blocks cardiac muscarinic receptors + sympathetic stimulation; autonomic margin of safety is moderate.
- Elimination prolonged in renal AND hepatic failure.
- Rarely used now; largely unavailable in many countries. — Miller's, p. 3289
B. Intermediate-Acting Agents
- Aminosteroid; primarily hepatic elimination (bile); minor renal excretion.
- 3-OH-vecuronium metabolite: 60–80% potency of parent, eliminated renally — can accumulate in renal failure causing prolonged block with repeated dosing.
- Minimal cardiovascular effects (no vagolysis, no histamine release) — gold standard for cardiac patients.
- Volume of distribution increased in cirrhosis → increased initial dose may be needed; but prolonged block possible with repeated doses. — Miller's, p. 3317
- Aminosteroid; primarily biliary and urinary excretion (no significant metabolism in humans; no active metabolites).
- Onset: 1.5–2 min at 0.6 mg/kg (2×ED₉₅); 90–120 seconds for RSI at 1.2 mg/kg (4×ED₉₅).
- Renal failure: clearance reduced 33–39%; elimination half-life prolonged (70 vs. 57 min); single-dose duration not significantly changed, but caution with repeated doses.
- Currently the preferred non-depolarising NMBD for RSI (when succinylcholine is contraindicated) — reversible with sugammadex 16 mg/kg.
- Minimal cardiovascular effects. — Miller's, p. 3328
- Benzylisoquinolinium; unique elimination via Hofmann degradation (spontaneous non-enzymatic breakdown at physiological pH and temperature) + ester hydrolysis by plasma esterases.
- Organ-independent elimination — preferred in liver failure, renal failure, multiorgan dysfunction.
- Laudanosine metabolite: CNS stimulant in large doses (seizures in animal models at high concentrations); clinically irrelevant at standard doses.
- Histamine release possible with rapid high doses → flushing, hypotension, bronchospasm.
- Isomer of atracurium (R-cis/R-cis stereoisomer); 4× more potent.
- Same Hofmann degradation pathway — organ-independent elimination; no active metabolites.
- Negligible histamine release — preferred in patients at risk for bronchospasm or cardiovascular instability.
- Slower onset than atracurium (higher potency, fewer molecules).
C. Short-Acting Agents
- Benzylisoquinolinium; hydrolysed by butyrylcholinesterase (same enzyme as succinylcholine) — short duration.
- Three stereoisomers: trans-trans (most potent, CL ~50–70 mL/kg/min) + cis-trans (CL ~100 mL/kg/min) + cis-cis (weak, long t½ ~55 min, minor contributor to duration).
- Prolonged block with pseudocholinesterase deficiency (same as succinylcholine).
- Histamine release possible with rapid bolus administration.
PART V — NEUROMUSCULAR MONITORING
"Quantitative neuromuscular monitoring is the only method of assessing whether a safe level of recovery of muscular function has occurred." — Miller's, p. 3221
"Subjective (qualitative) assessment is no longer considered sufficient for determining the depth of block or the adequacy of reversal; quantitative monitoring is recommended." — Miller's, p. 3276
1. Why Monitor? — The Safety Imperative
- Decreased upper oesophageal tone and coordination during swallowing
- Impaired hypoxic ventilatory drive
- Airway obstruction
- Postoperative pulmonary complications (aspiration, pneumonia, atelectasis)
- Increased healthcare costs, longer PACU stay, increased morbidity and mortality
"Approximately 30% to 50% of patients can have train-of-four ratios less than 0.90 following tracheal extubation in the absence of quantitative monitoring." — Miller's, p. 3221
2. Stimulation Patterns
- A single supramaximal stimulus (0.1–0.2 Hz)
- Baseline twitch is established before NMBD administration
- Not sensitive enough for early block detection — >70–75% receptor occupancy required for detectable twitch depression
- Not useful for detecting residual block (normal twitch height with significant residual block)
- 4 supramaximal stimuli at 2 Hz (0.5 second between stimuli)
- Most widely used clinically
- TOF Count: Number of twitches visible (0–4)
- TOF Ratio (TOFR): Height of 4th twitch / Height of 1st twitch
- Key threshold: TOFR ≥0.90 = adequate recovery (clinical recovery without appreciable residual block)
- Critical feature: Fade — progressive decrease in twitch height from T1 to T4 is caused by presynaptic receptor blockade
- No fade is seen with depolarising block (Phase I)
- Two short bursts of 3 tetanic stimuli (50 Hz), 750 ms apart
- More sensitive to fade than TOF subjectively
- Better than TOF for detecting residual block qualitatively (at TOFR 0.6–0.9)
- Sustained 50 Hz stimulation for 5 seconds
- Fade indicates non-depolarising block
- Sustained tetanic response (no fade) required for confidence in recovery at diaphragm/larynx
- Post-tetanic facilitation follows
- Used during deep block when no TOF response is elicitable
- Tetanic stimulus (50 Hz, 5 sec) → 3 second pause → single stimuli at 1 Hz
- Count of post-tetanic twitches predicts when TOF responses will return
- PTC of 1–2 = TOF count will return in approximately 15–20 min (for rocuronium/vecuronium)
3. Depth of Neuromuscular Block — Definitions (Table 24.6)
| Depth of Block | Objective (Quantitative) Criteria | Subjective PNS |
|---|---|---|
| Complete | PTC = 0 | PTC = 0 |
| Deep | PTC ≥1; TOF count = 0 | PTC ≥1; TOF count = 0 |
| Moderate | TOF count = 1–3 | TOF count = 1–3 |
| Shallow | TOFR <0.4 | TOF count = 4 with fade present |
| Minimal | TOFR 0.4–0.9 | TOF count = 4, fade absent subjectively* |
| Acceptable recovery | TOFR ≥0.90 | Cannot be determined subjectively |
Critical: "Absence of subjectively assessed fade has been reported with TOF ratio <0.3 and subjectively assessed presence of fade has been reported with TOF ratio >0.7." — Miller's, p. 3276
4. Monitoring Devices
| Device | Type | Method | Accuracy |
|---|---|---|---|
| Mechanomyography (MMG) | Quantitative | Measures force of thumb adduction; gold standard for research | High; bulky |
| Electromyography (EMG) | Quantitative | Measures compound muscle action potential | High; correlates well with MMG |
| Acceleromyography (AMG) | Quantitative | Measures acceleration of thumb using piezoelectric crystal | Most common clinical quantitative device; thumb must be free to move |
| Kinemyography (KMG) | Quantitative | Measures bending of sensor between thumb and finger | Moderate |
| Peripheral nerve stimulator (PNS) with tactile/visual assessment | Qualitative | Stimulator only — clinician tactile/visual assessment | Unreliable; not recommended for determining adequacy of reversal |
5. Monitoring Site — Adductor Pollicis
- Most sensitive muscle to NMBD effects
- Well-defined anatomy; easily accessible
- Most sensitive → first to show block, last to recover
- Caution: AP recovery overestimates block at the larynx/diaphragm; AP recovery underestimates block at the upper oesophagus.
PART VI — REVERSAL OF NEUROMUSCULAR BLOCKADE
1. Anticholinesterases (Neostigmine, Pyridostigmine, Edrophonium)
"There is a 'ceiling' effect to the maximal concentration of ACh that can be achieved with these drugs." — Miller's, p. 3221
| Agent | Dose | Onset | Duration | Notes |
|---|---|---|---|---|
| Neostigmine | 30–50 mcg/kg (max ~70 mcg/kg) | 3–7 min | 45–60 min | Must always be given with anticholinergic (glycopyrrolate 0.2 mg per 1 mg neostigmine) |
| Edrophonium | 0.5–1 mg/kg | 1–2 min | 30–45 min | Shorter onset; glycopyrrolate or atropine |
| Pyridostigmine | 0.1–0.25 mg/kg | 5–10 min | 60–90 min | Longest duration |
- Muscarinic stimulation → bradycardia, increased secretions, bronchoconstriction
- Must always be combined with an anticholinergic agent
- Glycopyrrolate preferred (quaternary ammonium — does not cross BBB, minimal CNS effects)
- Atropine: crosses BBB, causes tachycardia and CNS effects (confusion in elderly)
2. Sugammadex — Selective Relaxant-Binding Agent (SRBA)
"Sugammadex shows a high affinity for the steroidal neuromuscular blocking drugs rocuronium and vecuronium... forms a tight inclusion complex... thereby inactivating their effects and facilitating rapid reversal." — Miller's, p. 3221
| Clinical Situation | Depth of Block | Dose |
|---|---|---|
| Moderate block (TOF count 2+ responses) | TOF count ≥2 | 2 mg/kg |
| Deep block (PTC ≥1, TOF count = 0) | Deep | 4 mg/kg |
| Immediate reversal (RSI — within 3–5 min of rocuronium 1.2 mg/kg) | Complete | 16 mg/kg |
"Sugammadex reverses both moderate and deep neuromuscular block with a dose of 2.0 mg/kg and 4.0 mg/kg, respectively. An immediate reversal of neuromuscular block induced by rocuronium (given in doses up to 1.2 mg/kg) is possible when using a dose of sugammadex of 16 mg/kg." — Miller's, p. 3222
- No ceiling effect — can reverse any depth of block including complete block
- Faster reversal (minutes vs. 10–15 min with neostigmine)
- No need for anticholinergic co-administration (no muscarinic effects)
- Can reverse deep block safely (neostigmine cannot)
- Immediate reversal possible at 16 mg/kg — enables "cannot intubate, cannot oxygenate" scenarios where rocuronium is used for RSI
- Works ONLY on aminosteroid NMBDs (rocuronium > vecuronium); does NOT work on benzylisoquinoliniums (atracurium, cisatracurium, mivacurium)
- Hypersensitivity reactions — including anaphylaxis (rare, ~0.3%)
- Caution in renal failure — sugammadex–NMBD complex is primarily renally excreted; in severe renal failure, slow elimination may allow NMBD to re-dissociate (recurrence of block)
- Cost significantly higher than neostigmine
"Reversal of neuromuscular block by sugammadex is frequently, but not always, rapid." — Miller's, p. 3222
PART VII — RESIDUAL NEUROMUSCULAR BLOCKADE (RNMB)
1. Definition and Magnitude of the Problem
"Residual neuromuscular paralysis decreases upper oesophageal tone, coordination of the oesophageal musculature during swallowing, and hypoxic ventilatory drive. Residual paralysis can increase healthcare costs and the patient hospital length of stay, morbidity, and mortality." — Miller's, p. 3221
"Approximately 30% to 50% of patients can have train-of-four ratios less than 0.90 following tracheal extubation in the absence of quantitative monitoring." — Miller's, p. 3221
2. Clinical Consequences of RNMB (TOFR <0.90)
| TOFR | Clinical Consequence |
|---|---|
| <0.90 | Subclinical: impaired swallowing, impaired hypoxic ventilatory drive |
| <0.70 | Impaired upper airway obstruction prevention; diplopia, facial muscle weakness |
| <0.50 | Visible weakness; unable to sustain head lift for 5 seconds |
| <0.30 | Respiratory distress; inability to cough; dangerous hypoxia risk |
| <0.20 | Severe respiratory compromise |
- Studies have demonstrated that all signs of clinical recovery (5-second head lift, handgrip, tongue depressor test) can be present with TOFR as low as 0.60–0.70.
- However, subtle deficits in pharyngeal function, swallowing coordination, and hypoxic ventilatory drive persist until TOFR reaches 0.90.
- The "5-second head lift" test — historically considered gold standard for recovery — cannot detect residual block at TOFR 0.6–0.8.
3. Risk Factors for RNMB
- Use of long-acting NMBDs (pancuronium)
- Use of intermediate NMBDs without TOF monitoring
- No quantitative monitoring used
- Reversal with neostigmine at deep block
- No reversal given
- Renal/hepatic dysfunction (impaired elimination)
- Hypothermia (reduced metabolism, Hofmann degradation)
- Drug interactions (aminoglycosides, volatile agents, magnesium, local anaesthetics)
- Elderly patients (reduced clearance)
- Obesity (dosing errors on actual vs. lean body weight)
4. Prevention of RNMB
- Use quantitative monitoring routinely — TOF ratio measurement, not just TOF count
- Use intermediate or short-acting NMBDs (not pancuronium)
- Appropriate reversal at appropriate depth:
- Neostigmine only at TOF count of 4 with minimal fade (TOFR >0.4 ideally)
- Sugammadex for deep block or when rapid, reliable reversal is needed
- Confirm TOFR ≥0.90 before extubation using quantitative monitor
- Avoid extubation until TOFR ≥0.90 confirmed objectively
"Appropriate management of neuromuscular block based on quantitative monitoring will eliminate residual block and reduce the risk of postoperative complications." — Miller's, p. 3221
"After the administration of nondepolarising neuromuscular blocking drugs, it is essential to ensure adequate return of normal neuromuscular function using objective (quantitative) means of monitoring." — Miller's, p. 3221
PART VIII — DRUG INTERACTIONS
With Inhaled Anaesthetics
- Mechanism: enhanced presynaptic inhibition of ACh release + direct postjunctional muscle effects
- Order of potentiation: Isoflurane > sevoflurane/desflurane > N₂O > no volatile
- Effect: reduces ED₉₅ by ~25–30%; prolongs duration of block
- Clinically relevant: dose reduction needed when volatile agents used
With Other Drugs (Summary)
| Drug | Effect on NMBDs | Mechanism |
|---|---|---|
| Aminoglycosides | Potentiate | ↓ ACh release (presynaptic) + block receptor |
| Magnesium | Potentiate | ↓ ACh release; ↓ motor nerve excitability |
| Local anaesthetics | Potentiate | Channel block |
| Calcium channel blockers | Potentiate | ↓ Ca²⁺-mediated ACh release |
| Furosemide | Complex (low doses potentiate, high doses inconsistent) | ↓ cAMP; ↓ ACh output |
| Corticosteroids | Antagonise NMBDs acutely | Facilitate ACh release; chronic use → CIM if combined with NMBDs |
| Dantrolene | Potentiate | ↓ Ca²⁺ release from SR → ↓ muscle contraction |
| Azathioprine | Minor antagonism | Unclear |
| Antiestrogenic drugs (tamoxifen) | Potentiate | Unclear |
Defasciculating Doses (Pre-treatment)
- Reduces fasciculations and post-operative myalgia
- Antagonises the development of succinylcholine block — therefore increase succinylcholine dose by ~30% (from 1.0 to 1.3–1.5 mg/kg) when pre-treatment given. — Miller's, p. 3306
Drug Interactions Between Non-Depolarising NMBDs
- Same chemical class (e.g., atracurium + mivacurium; rocuronium + vecuronium): Additive interaction
- Different chemical classes (e.g., steroidal + benzylisoquinolinium): Synergistic interaction
- Example: rocuronium + mivacurium → rapid onset AND short duration — the ideal combination for some clinical scenarios
- Approximately 3 half-lives are required for a clinical changeover — until then, duration of block reflects the drug given first. — Miller's, p. 3305
PART IX — SPECIAL POPULATIONS AND CONSIDERATIONS
Renal Failure
| Drug | Effect | Safe? |
|---|---|---|
| Pancuronium | Markedly prolonged | Avoid |
| Vecuronium | Single dose OK; repeated doses → prolonged (3-OH metabolite accumulates) | Use caution with infusion |
| Rocuronium | Single-dose duration not significantly changed; caution with repeated doses | Acceptable; reversal with sugammadex reassuring |
| Atracurium/Cisatracurium | Organ-independent elimination — safest choice | Preferred |
| Mivacurium | OK (esterase metabolism); pseudocholinesterase may be low in renal failure | Acceptable |
| Succinylcholine | Avoid if hyperkalaemia present | Caution |
Hepatic Disease (Cirrhosis)
- Increased volume of distribution → apparent resistance (requires higher initial dose) — dilution effect, not decreased receptor sensitivity
- Prolonged elimination half-life for hepatically-eliminated drugs (vecuronium, pancuronium)
- Atracurium/cisatracurium: preferred — organ-independent elimination unaffected by liver failure
Neonates and Infants (<2 years)
- Neuromuscular junction maturation incomplete (fetal nAChRs extrajunctionally present up to 2 years)
- Children require HIGHER doses of non-depolarising NMBDs than adults (except infants <1 year, whose ED₉₅ at AP is ~30% less than adults due to larger volume of distribution)
- Succinylcholine contraindicated routinely in healthy children — risk of unmasked Duchenne/Becker MD → fatal hyperkalaemia/rhabdomyolysis. — Miller's, p. 3316
ICU and Prolonged NMB Use
- Drug-specific side effects
- Inadequate ventilation during ventilator failure/circuit disconnection
- Inadequate analgesia and sedation (paralysed patient may be awake and aware)
- Complications of immobility: DVT/PE, pressure injuries, peripheral nerve injuries
- Inability to cough, secretion retention, pulmonary infection
- Dysregulation of nAChRs
- Critical illness myopathy (CIM)
- Critical illness polyneuropathy (CIP)
- Prolonged paralysis after stopping relaxant
- Especially in patients with status asthmaticus treated with corticosteroids + NMBDs
- Primary cause: loss of myosin in myocytes → loss of contractile capacity
- Risk factors: corticosteroid treatment, immobility, sepsis, organ failure
- Prolonged immobility appears to be the key risk factor in corticosteroid-treated patients
- Occurs even without NMBDs or steroids in septic critically ill patients
PART X — MOST IMPORTANT CONCEPTS (SUMMARY)
| Concept | Key Statement from Miller's |
|---|---|
| Mechanism of nondepolarising block | Competitive antagonism at both α subunits; presynaptic block causes fade |
| Mechanism of succinylcholine block | Prolonged depolarisation → nAChR desensitisation + Na⁺ channel inactivation |
| Potency–onset inverse relationship | Low potency → faster onset (rocuronium) |
| Muscle sensitivity | Larynx > diaphragm > adductor pollicis (AP is most sensitive to block, last to recover) |
| TOF ratio threshold | TOFR ≥0.90 = adequate recovery; clinically cannot be determined subjectively |
| RNMB incidence | 30–50% incidence at extubation without quantitative monitoring |
| Succinylcholine hyperkalaemia | Extrajunctional fetal nAChR upregulation in burns, SCI, denervation → K⁺ surge |
| Succinylcholine in children | Contraindicated routinely — risk of unmasked Duchenne MD → cardiac arrest |
| Neostigmine ceiling effect | Only reverses shallow/minimal block; ineffective for deep block |
| Sugammadex | Reverses any depth of aminosteroid block; 16 mg/kg for immediate reversal of 1.2 mg/kg rocuronium |
| Hofmann degradation | Atracurium/cisatracurium — organ-independent elimination; preferred in organ failure |
| Drug interactions | Same class = additive; different class (steroidal + benzylisoquinolinium) = synergistic |
| ICU NMBDs | Risk CIM; corticosteroids + NMBDs particularly dangerous; always ensure adequate analgesia/sedation |
| Quantitative monitoring | The ONLY reliable method to confirm safe recovery; qualitative PNS assessment is insufficient |