Iron lungs ventillation
iron lung negative pressure ventilation mechanism history polio
iron lung mechanism negative pressure ventilation history
iron lung negative pressure ventilator polio
Iron Lung Ventilation
What Is the Iron Lung?
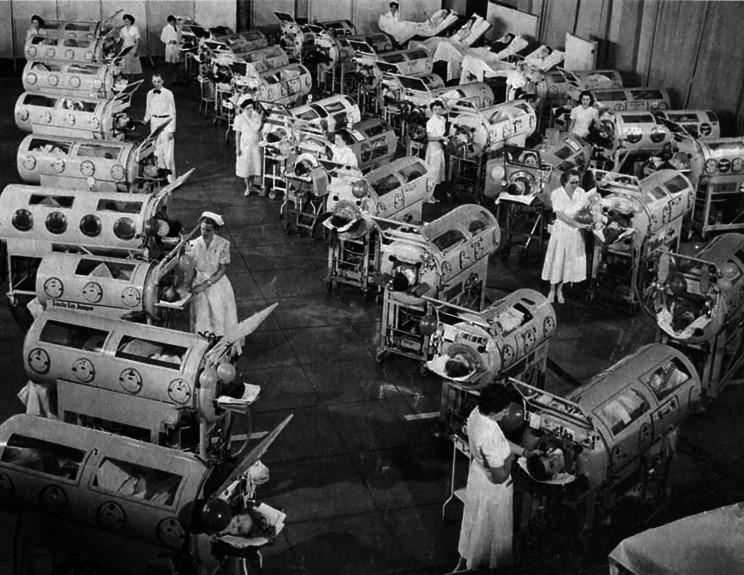
Mechanism: External Negative Pressure Ventilation (ENPV)
- Inspiration: A pump cyclically lowers the pressure inside the chamber to subatmospheric levels (typically −5 to −30 cmH₂O). This causes the thoracic cage and diaphragm to expand outward, reducing intrapleural and alveolar pressure → air flows passively into the lungs down a pressure gradient.
- Expiration: The chamber pressure returns to atmospheric. The elastic recoil of the lung and chest wall drives air back out passively.
"NPV involves generating subatmospheric pressure around the patient's chest, which causes chest wall and upper abdominal expansion, as well as downward diaphragmatic movement, resulting in inspiration. The negative pressure generated is proportional to the tidal volume delivered." — Fishman's Pulmonary Diseases and Disorders

Historical Development
| Year | Milestone |
|---|---|
| 1838 | Scottish physician John Dalziel described the first tank respirator using rhythmic negative pressure |
| 1905 | Davenport developed early versions with hand-operated bellows |
| 1918 | Chillingworth & Hopkins demonstrated electrically powered body plethysmographs to ventilate animals |
| 1928 | Philip Drinker, Charles McKhann, and Louis Shaw built the first electrically powered iron lung — threshold pressures of just 5–10 cmH₂O were found sufficient to override spontaneous breathing in normal subjects |
| 1937 | Portable plywood cabinet respirators introduced |
| 1952 | Copenhagen polio epidemic — anesthesiologist Björn Ibsen showed that invasive positive-pressure ventilation (PPV) improved survival, beginning the transition away from iron lungs |
| 1960 | Kelleher's rotatable iron lung introduced to treat atelectasis |
| Mid-1980s | Iron lungs finally displaced by noninvasive PPV (NPPV/BiPAP) for chronic respiratory failure |
Primary Clinical Application: Poliomyelitis
"Before the 1960s, the use of negative pressure ventilation in the form of a tank ventilator ('iron lung') was the most common form of mechanical ventilation outside the anesthesia suite." — Roberts and Hedges' Clinical Procedures in Emergency Medicine
Iron Lung vs. Positive-Pressure Ventilation (PPV)
| Feature | Iron Lung (NPV) | Modern PPV |
|---|---|---|
| Mechanism | Subatmospheric pressure around thorax | Positive pressure delivered to airway |
| Intubation required | No | Yes (IPPV) or no (NPPV mask) |
| Hemodynamic effect | Augments venous return (like normal breathing) | Reduces venous return / cardiac output |
| Barotrauma risk | Low | Present (especially volutrauma) |
| Airway resistance | Cannot overcome high resistance | Can overcome via pressure/flow settings |
| Poor compliance | Cannot compensate | Can compensate |
| Patient access | Very limited | Full access |
| Portability | Poor | Good (modern portable units) |
| Secretion clearance | Difficult | Can be managed via ETT |
"Although negative-pressure ventilation does not require tracheal intubation, it cannot overcome substantial increases in airway resistance or decreases in pulmonary compliance, and it also limits access to the patient." — Morgan and Mikhail's Clinical Anesthesiology, 7e
Related NPV Devices (Modern Descendants)
- Chest shell / cuirass ventilator — covers only the anterior chest
- Wrap ventilator (pneumosuit) — fabric wrap around trunk
- Port-a-lung — more portable enclosure
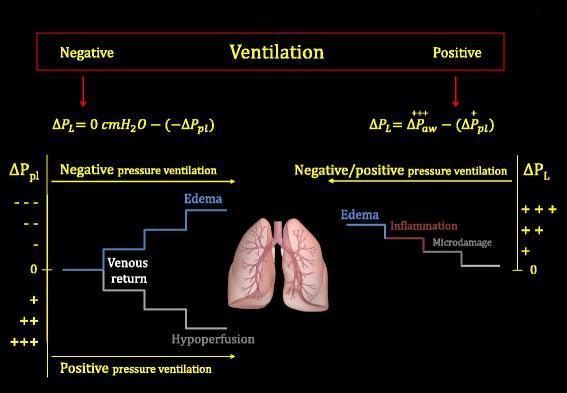
Physiological Comparison: NPV vs. PPV
Limitations Leading to Its Decline
- No airway access — suctioning, bronchoscopy, or emergency intubation is nearly impossible
- Cannot overcome secretion plugging or airway obstruction
- Obstructive apneas — upper airway muscles may not be paralyzed but may not generate enough tone to counteract the negative pressure, causing vocal cord collapse and paradoxical airway obstruction during "inspiration"
- Immobility and nursing challenges — patients cannot be easily turned (risk of pressure ulcers, atelectasis)
- Replaced by NPPV — BiPAP masks in the 1980s provided equivalent support for neuromuscular disease without these drawbacks
Retry
iron lung ventilation mechanism physiology history modern use
iron lung tank ventilator negative pressure polio ward
Iron Lung Ventilation
What Is the Iron Lung?

Mechanism of Ventilation
Inspiration
- Causes the chest wall and upper abdomen to expand outward
- Pulls the diaphragm downward
- Reduces intrapleural and alveolar pressure below atmospheric
- Air flows passively into the lungs down the resulting pressure gradient
Expiration
"A time-cycled negative pressure ventilator consisting of an airtight cylinder that encloses the patient up to his neck, leaving the head exposed to atmospheric pressure. Subatmospheric pressure is applied to the body rhythmically in phase with inspiration. When pressure inside the tank returns to atmospheric, the natural recoil of the lungs produces exhalation." — Milbank Memorial Fund
Historical Timeline
| Year | Event |
|---|---|
| 1838 | Scottish physician John Dalziel first described a tank respirator using rhythmic negative pressure |
| 1905 | Davenport (London) developed early hand-operated bellows versions |
| 1918 | Chillingworth & Hopkins ventilated tracheotomized animals using an electrically powered body plethysmograph — inspired Drinker's work |
| 1928 | Philip Drinker, Louis Agassiz Shaw (physiologist), and Charles McKhann (paediatrician) built the first electric-powered iron lung at Harvard. Threshold pressures of only 5–10 cmH₂O could override a normal subject's breathing |
| 1931 | John Emerson built a quieter, improved version (the "Emerson respirator") with portholes and leather bellows |
| 1937 | Portable plywood cabinet respirators introduced |
| 1948–1952 | Mass deployment during US polio epidemics; whole wards filled with iron lungs |
| 1952 | Copenhagen polio epidemic — anesthesiologist Björn Ibsen demonstrated that tracheotomy + manual PPV by medical students reduced mortality in bulbar polio from ~90% to ~25%, marking the beginning of the end for iron lungs |
| 1958 | First ICU established; concept of organ support directly descended from iron lung era |
| 1961 | Mildred Stahlman used a modified iron lung to save a premature infant → birth of the modern NICU |
| Mid-1980s | BiPAP/nasal CPAP displaces iron lungs as treatment for chronic respiratory failure in neuromuscular disease |
Primary Clinical Application: Poliomyelitis
"Respiratory motor nuclei may be directly involved, resulting in diaphragmatic or other respiratory muscle dysfunction... the central respiratory centers can be directly affected, resulting in irregular respirations." — Fishman's Pulmonary Diseases and Disorders
- 15–30% of adults with paralytic polio died even with the iron lung
- Post-polio syndrome: 20–30 years later, progressive muscle weakness, fatigue, and pain can recur — respiratory failure risk is highest in those who needed ventilation acutely

Iron Lung vs. Positive-Pressure Ventilation (PPV)
| Feature | Iron Lung (NPV) | Positive Pressure Ventilation (PPV) |
|---|---|---|
| Mechanism | Subatmospheric pressure around thorax | Positive pressure into airway |
| Intubation required | No | IPPV: yes; NPPV: no (mask) |
| Mimics normal breathing | Yes — thorax expands first | No — airway pressure drives inflation |
| Venous return | Augmented (↓ intrathoracic pressure) | Reduced (↑ intrathoracic pressure) |
| Cardiac output effect | Preserved/enhanced | Can reduce CO, ↑ risk of hypoperfusion |
| Barotrauma / volutrauma | Minimal | Present (especially with large Vt) |
| Overcomes high airway resistance | Cannot | Yes |
| Overcomes low compliance | Cannot | Yes |
| Patient access | Very limited (portholes only) | Full access |
| Secretion management | Extremely difficult | Via ETT/suction |
| Portability | Poor | Good (modern portable units) |
"Although negative-pressure ventilation does not require tracheal intubation, it cannot overcome substantial increases in airway resistance or decreases in pulmonary compliance, and it also limits access to the patient." — Morgan and Mikhail's Clinical Anesthesiology, 7e

- NPV → decreases pleural pressure → increases venous return → risk of pulmonary edema in volume-overloaded patients
- PPV → increases pleural pressure → reduces venous return → risk of hemodynamic compromise and hypoperfusion
Limitations and Why It Was Replaced
- No airway access — bronchoscopy, suctioning, and emergency intubation are nearly impossible
- Cannot overcome airway obstruction — bulbar polio with secretion pooling still caused death even with iron lung support; this is what killed patients in Copenhagen 1952
- Upper airway collapse — the negative pressure can pull vocal cords together, causing obstructive apneas, especially during sleep
- Immobility — patients cannot be turned easily; risk of pressure ulcers and atelectasis
- Bulk and noise — electric pumps were large and loud; early devices required continuous attention
- Displaced by NPPV — nasal CPAP (introduced 1980s for OSA) and BiPAP provided equivalent or better non-invasive support for neuromuscular patients without these drawbacks
"The clustering of paralyzed patients needing ventilatory support paved the way for development of medical intensive care units." — Fishman's Pulmonary Diseases and Disorders
Modern Descendants (NPV Devices Still in Use)
| Device | Description |
|---|---|
| Chest shell / cuirass ventilator | Hard shell covering anterior chest only |
| Wrap ventilator (pneumosuit) | Fabric wrap around the trunk |
| Port-a-lung | More portable partial enclosure |
| cNEP (continuous negative extrathoracic pressure) | Used for OSA research — same concept as iron lung applied to the upper airway |
Legacy
- The concept of organ support (keeping a patient alive while an underlying cause resolves)
- The modern ICU (Copenhagen 1952 created the first concentration of critically ill patients needing continuous monitoring)
- The NICU (Stahlman's 1961 adaptation)
- The field of respiratory physiology and gas exchange as a clinical discipline
"The invention of the iron lung irrevocably changed the relationship between humans and machines. For the first time, an individual struggling to breathe for an extended period could receive support, with the machine doing some or all of the work of breathing." — Hannah Wunsch, Weill Cornell Medical Center
Sigh phenomenon on ventillator - explain like a professor teaching a junior resident
sigh breath mechanical ventilation physiology atelectasis prevention ICU
sigh breath mechanical ventilation atelectasis
sigh ventilation ARDS recruitment atelectasis
sigh breath ventilator mechanism physiology atelectasis surfactant compliance
sigh ventilation lung recruitment oxygenation clinical trial
sigh mode ventilator settings clinical use ARDS low tidal volume evidence 2023 2024
The Sigh on a Ventilator — A Teaching Session
Step 1 — Start With What You Already Know: The Normal Sigh
Step 2 — The Physiology: Why Monotonous Ventilation Causes Trouble
1. Surfactant Depletion → Alveolar Instability
2. Atelectasis → Shunting → Hypoxemia
- Without sighs: lung compliance falls ~15% and PaO₂ falls ~22%
- After a few minutes of deep, slow sustained sighs: PaO₂ rises ~150 mmHg
3. The "Open Lung" Problem — Why Alveoli Don't Stay Open
Step 3 — The Ventilator Sigh: What It Actually Does
| Parameter | Normal Breath | Sigh Breath |
|---|---|---|
| Tidal volume | 6–8 mL/kg IBW | ~9–16 mL/kg IBW (1.5–2×) |
| Frequency | Set RR | 3–10/hour |
| Purpose | Gas exchange | Alveolar recruitment + surfactant redistribution |
Step 4 — When Does This Matter Clinically?
The Original Context: Low Tidal Volume Ventilation in ARDS
Other Contexts Where Sigh Matters:
- Perioperative/anesthesia ventilation — paralyzed patients lose their sigh reflex; progressive atelectasis over hours of surgery is well-documented
- Neuromuscular disease — patients with weak respiratory muscles can't generate their own sighs (e.g., MND, GBS); NIV sigh settings compensate
- Neonates on HFOV — here the evidence inverts: sigh breaths on top of high-frequency oscillation increase pneumothorax risk in extremely premature neonates (born 22–25 weeks). You give with one hand, take with the other — always context-dependent.
Step 5 — The Counterargument: Why Don't We Always Use Sighs?
-
Barotrauma and volutrauma — a sigh breath is by definition a larger volume. In an already overdistended ARDS lung, a 2× tidal volume breath could cause regional overdistension in the more compliant lung units. Peak and plateau pressures will be higher during the sigh.
-
Hemodynamics — a large tidal volume transiently increases intrathoracic pressure, which can reduce venous return and cardiac output. In a hemodynamically unstable patient, this matters.
-
Patient–ventilator dyssynchrony — in spontaneously breathing patients, a machine-delivered sigh may conflict with the patient's own inspiratory effort.
-
No universal guideline — there is no RCT proving that routine sigh use in all-comers on mechanical ventilation improves mortality. The evidence base is better in ARDS-specific populations.
The Takeaway — How I Want You to Think About This
"This machine is doing something biology evolved to do automatically. What reflexes have I taken away by intubating this patient, and does the ventilator settings compensate for them?"
Quick Reference Summary
| What it is | Periodic delivery of 1.5–2× normal tidal volume, ~3–10×/hour |
| Why it matters | Maintains alveolar stability, prevents surfactant depletion, reduces atelectasis |
| Physiological basis | Mimics the normal spontaneous sigh reflex abolished by intubation/paralysis |
| Key effect | ↑ surfactant release, ↓ alveolar surface tension, ↓ V/Q mismatch, ↑ PaO₂ |
| Best evidence for use | ARDS on low Vt protective ventilation; perioperative ventilation |
| Risks | Barotrauma, hemodynamic compromise, dyssynchrony |
| Caution | Extremely preterm neonates on HFOV — increased pneumothorax risk |