1. Explain the Physiology of hearing. Discuss the tests done to assess vestibular functions in detail wth diagram
physiology of hearing external ear middle ear cochlea organ of corti auditory pathway detailed
vestibular function tests caloric test electronystagmography videonystagmography rotational chair posturography head impulse test vestibular evoked myogenic potentials
diagram physiology of hearing cochlea organ of corti auditory pathway vestibular tests schematic
Physiology of Hearing
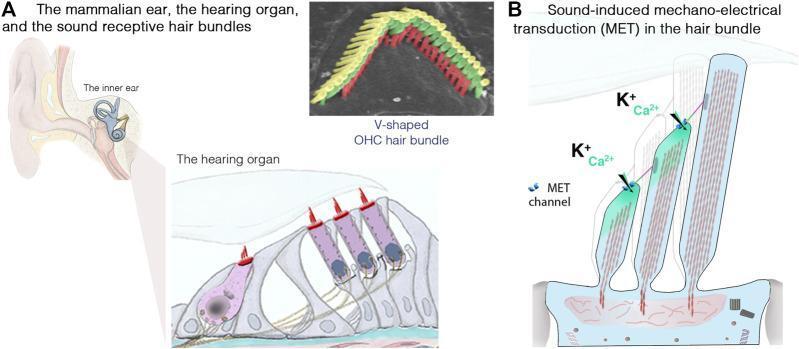
1) Sound collection and conduction
- Pinna + external auditory canal collect and funnel sound waves to the tympanic membrane.
- Tympanic membrane vibrates and transmits motion to ossicles (malleus, incus, stapes).
- Middle ear impedance matching: air-to-fluid transfer is normally inefficient, but tympanic membrane plus ossicles amplify pressure and improve transmission into the cochlea by about 200-fold. Without this mechanism, most sound energy would be reflected.
(Harrison’s Principles of Internal Medicine, 21e, p. 1019)
2) Cochlear mechanics
- Stapes footplate movement at oval window creates pressure waves in cochlear fluids.
- A traveling wave forms along the basilar membrane.
- Tonotopy:
- Base of cochlea (stiff, narrow) responds to high frequencies
- Apex (compliant, wide) responds to low frequencies
3) Organ of Corti and mechanotransduction
- Organ of Corti contains:
- 1 row inner hair cells (IHCs): main sensory transducers
- 3 rows outer hair cells (OHCs): cochlear amplifier (enhance sensitivity and tuning)
- Stereocilia are connected by tip links. Deflection toward tallest cilia opens mechano-electrical transduction channels.
- K+ and Ca2+ influx from endolymph depolarizes hair cells.
- Depolarization opens voltage-gated Ca2+ channels at hair cell base, releasing glutamate onto spiral ganglion afferents.
- Auditory nerve fibers fire action potentials proportional to frequency, intensity, and timing.
4) Central auditory pathway
- Cochlear nerve (CN VIII)
- Cochlear nuclei (pontomedullary junction)
- Bilateral projections (including superior olivary complex for binaural processing)
- Lateral lemniscus
- Inferior colliculus
- Medial geniculate body (thalamus)
- Primary auditory cortex (Heschl’s gyrus, temporal lobe)
5) Coding principles
- Frequency: place code (tonotopy) + temporal coding at low frequencies
- Intensity: firing rate + recruitment of more fibers
- Localization:
- Interaural time differences (low frequency)
- Interaural level differences (high frequency)
- Spectral cues from pinna
Vestibular Function Tests (Detailed)
(Benign Paroxysmal Positional Vertigo guideline text, p. 19)
A. Bedside clinical tests
- Head impulse test (bedside)
- Rapid unpredictable small-amplitude head thrust while patient fixates target.
- Corrective saccade suggests reduced vestibulo-ocular reflex (VOR) on side turned toward.
- Dynamic visual acuity
- Compare visual acuity with head still vs oscillating.
- Romberg/tandem gait/Fukuda stepping
- Functional screening of vestibulospinal deficits.
- Dix-Hallpike and supine roll test
- Positional tests, especially for BPPV.
B. Instrumented vestibular tests
1) Caloric test (ENG/VNG based)
- Primarily each lateral canal separately at very low frequency range.
- Best for unilateral vestibular hypofunction.
(BPPV guideline text, p. 19)
- Patient supine, head elevated ~30°.
- Sequential warm and cold stimulation each ear.
- Eye movements recorded (ENG/VNG), especially slow-phase velocity.
- Canal paresis (unilateral weakness)
- Directional preponderance
- Bilateral reduced responses (if globally low)
- Uncomfortable; can cause nausea.
- Tests low-frequency vestibular response only.
2) Electronystagmography (ENG) / Videonystagmography (VNG)
- ENG: corneo-retinal potential (electrodes)
- VNG: infrared video goggles (now common)
- Spontaneous/gaze nystagmus
- Saccade, smooth pursuit, optokinetic tests
- Positional/positioning nystagmus
- Caloric subtest
- Differentiate peripheral vs central patterns
- Objective documentation of vestibular deficit
3) Rotational chair testing
- Most sensitive/reliable for bilateral vestibular hypofunction quantification.
(BPPV guideline text, p. 19)
- VOR gain
- Phase
- Symmetry
- Better tolerated than calorics
- Good for bilateral loss and monitoring progression
4) Video Head Impulse Test (vHIT)
- Function of all 6 semicircular canals (right/left horizontal, anterior, posterior)
- Detects overt/covert corrective saccades and reduced VOR gain
(BPPV guideline text, p. 19)
- Quick, bedside-compatible instrumented test
- Complements caloric (different frequency domains)
5) Vestibular Evoked Myogenic Potentials (VEMP)
- cVEMP (sternocleidomastoid): mainly saccule + inferior vestibular nerve
- oVEMP (inferior oblique recording): mainly utricle + superior vestibular nerve
(BPPV guideline text, p. 19)
- Otolith dysfunction assessment
- Helpful in superior canal dehiscence, vestibular neuritis subtype localization, etc.
6) Computerized Dynamic Posturography (CDP)
- Functional effect of vestibular deficits on balance
- Sensory organization strategy and fall risk trends
(BPPV guideline text, p. 19)
- Not lesion-localizing by itself; best as complementary functional tool.
7) Additional useful tests
- Subjective visual vertical: utricular pathway bias
- Audiovestibular correlation: pure tone audiometry + speech + tympanometry in combined cochleo-vestibular disorders
- MRI/CT when central lesion or structural pathology suspected
Practical test selection (exam-style summary)
| Clinical suspicion | Most useful initial tests |
|---|---|
| Unilateral peripheral loss | VNG/ENG with caloric + vHIT |
| Bilateral vestibulopathy | Rotational chair + vHIT ± calorics |
| Otolith disorder | cVEMP + oVEMP |
| Functional balance impact | Dynamic posturography |
| Positional vertigo (BPPV) | Dix-Hallpike / Roll test (diagnostic), VNG if atypical |
- A clean flowchart diagram of auditory pathway and hearing physiology (text schematic for exam drawing), and
- A one-page viva answer on vestibular tests with labeled mini-diagrams (caloric setup, vHIT, VEMP pathways).
physiology of hearing fow chart
Physiology of Hearing - Flow Chart
Sound waves in air
↓
Pinna collects sound
↓
External auditory canal transmits sound
↓
Tympanic membrane vibrates
↓
Ossicles move (Malleus → Incus → Stapes)
↓
Stapes footplate pushes oval window
↓
Pressure waves in cochlear fluid (perilymph/endolymph)
↓
Traveling wave on basilar membrane
↓
Shearing movement between tectorial membrane and hair cells
↓
Stereocilia deflection in Organ of Corti
↓
Mechano-electrical transduction channels open
(K+ influx from endolymph, then Ca2+ entry)
↓
Hair cell depolarization
↓
Neurotransmitter (glutamate) release at hair cell-afferent synapse
↓
Action potentials in cochlear nerve (CN VIII)
↓
Cochlear nuclei (brainstem)
↓
Superior olivary complex (binaural processing)
↓
Lateral lemniscus
↓
Inferior colliculus (midbrain)
↓
Medial geniculate body (thalamus)
↓
Primary auditory cortex (Heschl gyrus, temporal lobe)
↓
Perception of hearing (pitch, loudness, localization, speech)
One-line exam version
tests to assess vestibuar functions
Tests to Assess Vestibular Functions
1) Clinical bedside tests
- Dix-Hallpike test: for posterior canal BPPV.
- Supine roll test: for horizontal canal BPPV.
- Head impulse test (HIT): checks vestibulo-ocular reflex (VOR); corrective saccade suggests peripheral hypofunction.
- Romberg/tandem gait/Fukuda stepping: gross vestibulospinal function.
2) Eye movement recording tests
- ENG (Electronystagmography) and VNG (Videonystagmography):
- Record spontaneous, gaze-evoked, positional, and caloric-induced nystagmus.
- Help differentiate peripheral vs central vestibular disorders.
3) Caloric test
- Warm/cold air or water stimulates each ear separately (mainly lateral semicircular canal).
- Very useful for unilateral vestibular weakness.
- Measured by nystagmus slow-phase velocity.
4) Rotational chair test
- Patient rotated in motorized chair; eye movements recorded.
- Best for bilateral vestibular hypofunction quantification.
- Measures VOR gain, phase, and symmetry.
5) Video Head Impulse Test (vHIT)
- High-speed camera measures eye response to rapid head thrusts.
- Assesses all 6 semicircular canals at high-frequency head motion.
- Detects overt and covert catch-up saccades.
6) Vestibular Evoked Myogenic Potentials (VEMP)
- cVEMP: mainly saccule + inferior vestibular nerve function (recorded from SCM muscle).
- oVEMP: mainly utricle + superior vestibular nerve function (recorded from extraocular muscles).
7) Posturography (Computerized Dynamic Posturography)
- Evaluates balance under sensory conflict conditions.
- Shows functional impact of vestibular deficits on posture and fall risk.
8) Subjective Visual Vertical (SVV)
- Assesses utricular pathway dysfunction by ability to align a vertical line.
Quick exam classification
- Semicircular canal tests: Caloric, vHIT, Rotational chair
- Otolith tests: cVEMP, oVEMP, SVV
- Functional balance tests: Posturography, Romberg/Fukuda
- Positional vertigo tests: Dix-Hallpike, Supine roll
tests to assess vestibuar functions
Tests to Assess Vestibular Function
-
Dix-Hallpike test
- Diagnoses posterior canal BPPV (position-induced vertigo/nystagmus).
-
Supine Roll test (Pagnini-McClure)
- Diagnoses horizontal canal BPPV.
-
Head Impulse Test (HIT) / Video HIT (vHIT)
- Assesses vestibulo-ocular reflex (VOR).
- vHIT evaluates all 6 semicircular canals.
-
Romberg, Tandem gait, Fukuda stepping test
- Bedside screening of vestibulospinal balance function.
-
ENG / VNG (Electronystagmography / Videonystagmography)
- Records eye movements and nystagmus (spontaneous, positional, gaze-evoked).
- Helps peripheral vs central differentiation.
-
Caloric test
- Warm/cold stimulation of each ear separately (mainly lateral semicircular canal).
- Good for unilateral vestibular weakness.
-
Rotational chair test
- Quantifies bilateral vestibular hypofunction.
- Measures VOR gain, phase, symmetry.
-
Vestibular Evoked Myogenic Potentials (VEMP)
- cVEMP: saccule/inferior vestibular nerve
- oVEMP: utricle/superior vestibular nerve
-
Computerized Dynamic Posturography
- Functional impact of vestibular loss on posture and balance.
-
Subjective Visual Vertical (SVV)
- Assesses otolith (mainly utricular) pathway function.
in detai bout bppv
BPPV (Benign Paroxysmal Positional Vertigo) - Detailed
Definition
Pathophysiology
-
Canalithiasis (most common)
- Free-floating otoconia in canal endolymph
- Head movement causes particle lag and abnormal cupula deflection
- Produces transient vertigo + nystagmus (usually < 1 min)
-
Cupulolithiasis
- Otoconia attached to cupula
- Cupula becomes gravity-sensitive
- Symptoms may be more persistent
Risk Factors / Causes
- Idiopathic (most common)
- Head trauma
- Vestibular neuritis/labyrinthine insults
- Prolonged bed rest
- Increasing age
- Migraine association
- Post-ear surgery (occasionally)
Clinical Features
- Brief vertigo spells (seconds, usually <1 min)
- Triggered by:
- Turning in bed
- Looking up (top-shelf vertigo)
- Bending forward
- Getting up or lying down
- Associated nausea, imbalance
- Usually no hearing loss, no tinnitus, no focal neuro deficits (if present, think alternate diagnosis)
Diagnosis
1) Posterior canal BPPV: Dix-Hallpike test
- From sitting, head turned 45° to one side, then quickly laid back with neck extension.
- Positive:
- Latent onset (few seconds)
- Torsional upbeating nystagmus
- Fatigable, transient
- Reversal on sitting up
2) Horizontal canal BPPV: Supine roll test
- Supine with head flexed 30°, roll head to each side.
- Geotropic or apogeotropic horizontal nystagmus helps subtype.
Nystagmus Pattern by Canal (high-yield)
- Posterior canal: torsional + upbeating
- Horizontal canal: horizontal (geo/apogeotropic)
- Anterior canal: torsional + downbeating (rare, evaluate carefully to exclude central cause)
Treatment
A) Canal Repositioning Maneuvers (first-line)
-
Epley maneuver
- For posterior canal canalithiasis
- Most commonly used and highly effective
-
Semont (Liberatory) maneuver
- Alternative for posterior canal
-
Barbecue (Lempert) roll maneuver
- For horizontal canalithiasis
-
Gufoni maneuver
- Horizontal canal variants
-
Deep head-hanging maneuver
- For anterior canal BPPV
B) Medicines
- Vestibular suppressants (e.g., short course) only for severe nausea/vomiting.
- They do not treat root cause and should not be routine long-term therapy.
C) Surgery (rare)
- Posterior canal plugging or singular neurectomy in intractable refractory cases.
Follow-up and Prognosis
- Excellent prognosis; many improve after 1-2 maneuvers.
- Recurrence is common (about 15-50% over years).
- Repeat repositioning usually works.
- Vestibular rehab useful if residual dizziness/balance issues persist.
Red Flags (consider central cause, not simple BPPV)
- Persistent non-fatigable nystagmus
- Pure vertical nystagmus without typical positional pattern
- Severe headache/new neuro deficits
- Ataxia out of proportion
- Diplopia, dysarthria, limb weakness/numbness
- New unilateral hearing loss with other neuro signs
Exam-oriented short note format
Symptoms: brief positional vertigo, nausea, no auditory symptoms.
Diagnosis: Dix-Hallpike (posterior), Roll test (horizontal).
Treatment: Epley/Semont/BBQ roll as per canal involved.
Prognosis: good but recurrent.
- Stepwise Epley maneuver diagram (exam drawing style)
- Table: posterior vs horizontal vs anterior BPPV
- Case-based viva questions with answers.
3. Explain Transient ischemic attack. Describe the acute management of stroke in detail
1) Transient Ischemic Attack (TIA)
Definition
Traditionally symptoms last <24 h, but most TIAs last minutes (often <1 h).
Common Clinical Features
- Unilateral weakness or numbness
- Speech disturbance (aphasia/dysarthria)
- Monocular vision loss (amaurosis fugax) or hemianopia
- Brainstem symptoms (diplopia, vertigo, ataxia, dysphagia)
Why TIA is important
- It is a warning stroke.
- Early stroke risk is highest in first 24-48 h.
- Needs urgent evaluation and secondary prevention.
Evaluation of Suspected TIA
- Immediate assessment
- ABC, vitals, glucose, neuro exam
- Brain imaging
- MRI with diffusion preferred (or urgent CT if MRI unavailable)
- Vascular imaging
- CTA/MRA/carotid duplex for carotid/intracranial stenosis
- Cardiac workup
- ECG, telemetry, echocardiography when indicated
- Labs
- CBC, electrolytes, renal function, coagulation, HbA1c, lipid profile
- Risk stratification
- ABCD2 score can support triage, but does not replace urgent specialist evaluation
TIA Management (Early Secondary Prevention)
- Antiplatelet therapy (if non-cardioembolic):
- Aspirin, or short-term dual antiplatelet (aspirin + clopidogrel) in selected high-risk minor stroke/TIA
- Anticoagulation if cardioembolic source (e.g., atrial fibrillation)
- High-intensity statin
- BP, diabetes, smoking, lifestyle control
- Carotid revascularization in suitable symptomatic high-grade carotid stenosis
2) Acute Management of Stroke (Detailed)
A. Initial Emergency Approach (First minutes)
- Recognize stroke and activate stroke code
- ABCs
- Airway protection, oxygen if hypoxic, cardiac monitoring
- Determine time last known well
- Rapid focused exam + NIHSS
- Immediate bedside glucose
- Treat hypoglycemia/hyperglycemia
- Urgent non-contrast CT head (or MRI where fast pathway exists)
- Differentiate ischemic vs hemorrhagic stroke
- Basic labs (do not delay reperfusion unless essential):
- CBC, PT/INR, aPTT, creatinine, electrolytes, troponin
B. Acute Ischemic Stroke Management
1) Reperfusion Therapy
i) Intravenous thrombolysis
- Drug: Alteplase (tPA) (or tenecteplase in selected protocols)
- Time window: usually within 4.5 h from onset (eligibility based on protocol)
- BP must be controlled (typically <185/110 before thrombolysis)
- Intracranial hemorrhage on CT
- Active major bleeding
- Recent major intracranial surgery/bleed
- Severe uncontrolled hypertension despite therapy
- Certain anticoagulation-related exclusions (protocol-based)
ii) Mechanical thrombectomy
- For large vessel occlusion (e.g., ICA, proximal MCA) with favorable imaging
- Standard window up to 6 h, extended up to 24 h in selected patients (perfusion mismatch criteria)
2) Antiplatelet and Anticoagulation
- If no thrombolysis: aspirin started early after imaging excludes hemorrhage.
- If thrombolysis given: avoid antiplatelet/anticoagulant for first 24 h, then repeat imaging before starting.
- Anticoagulation is generally not immediate unless specific indication and timing decision.
3) Blood Pressure in Ischemic Stroke
- If candidate for thrombolysis: lower BP to target threshold before treatment.
- If not candidate: permissive hypertension often allowed unless very high BP or comorbidity indications.
4) Supportive Care
- Maintain normoxia, normoglycemia, normothermia
- Treat fever
- IV fluids (avoid hypotonic overload/dehydration)
- Swallow screening before oral intake (prevent aspiration)
- DVT prophylaxis
- Early mobilization and rehab planning
C. Intracerebral Hemorrhage (ICH) Acute Management
- Reverse anticoagulation urgently (agent-specific reversal)
- BP control (careful rapid reduction per protocol)
- Neurosurgical evaluation
- Cerebellar bleed with compression/hydrocephalus
- Large hematoma or deterioration
- Manage raised ICP
- Head elevation, osmotherapy as indicated, ventilation strategy
- Seizure management if clinical/electrographic seizures
- ICU-level monitoring for severe cases
D. Subarachnoid Hemorrhage (if suspected)
- Urgent CT/CTA and neurosurgical/neurointerventional referral
- Secure aneurysm (coiling/clipping)
- Nimodipine to reduce delayed ischemia risk
- Manage vasospasm, hydrocephalus, electrolyte disturbances
E. Stroke Unit Care (Critical for outcomes)
- Continuous neuro monitoring
- Early complication detection
- Multidisciplinary care (neurology, nursing, physio, speech/swallow, OT)
F. Early Secondary Prevention After Acute Phase
- Determine stroke mechanism (TOAST-style etiologic workup)
- Antiplatelet or anticoagulant as indicated
- High-intensity statin
- BP and diabetes control
- Carotid and cardiac source management
- Smoking/alcohol/diet/exercise counseling
Quick Flowchart (Acute Stroke)
→ Hemorrhage?
- Yes → ICH/SAH protocol (BP control, reversal, neurosurgery)
- No (ischemic) → assess for IV thrombolysis window/eligibility
→ large vessel occlusion imaging
→ mechanical thrombectomy if eligible
→ stroke unit supportive care + secondary prevention
- Time targets (door-to-CT, door-to-needle, door-to-groin)
- tPA inclusion/exclusion checklist
- BP targets in ischemic vs hemorrhagic stroke.
not satisfied answer in deatil
3. Transient Ischemic Attack (TIA) and Acute Management of Stroke (Detailed, Exam Style)
Part A: Transient Ischemic Attack (TIA)
1) Definition
- Older time-based definition: symptoms resolve within 24 hours
- Modern tissue-based definition (preferred): no infarction on diffusion imaging
2) Pathophysiology
- Artery-to-artery embolism
- Atherosclerotic plaque in carotid or vertebral arteries
- Cardioembolism
- Atrial fibrillation, valvular disease, LV thrombus
- Small vessel disease
- Lipohyalinosis in chronic hypertension/diabetes
- Hemodynamic causes
- Severe carotid stenosis with systemic hypotension
- Hypercoagulable states
- Less common; in selected younger patients/recurrent events
3) Clinical Features of TIA
Typical symptoms
- Unilateral weakness (face/arm/leg)
- Unilateral numbness
- Aphasia or dysarthria
- Monocular blindness (amaurosis fugax)
- Homonymous hemianopia
- Brainstem/cerebellar symptoms: diplopia, ataxia, dysphagia, vertigo (with focal signs)
Features favoring mimics rather than TIA
- Gradual march of symptoms
- Positive phenomena (flashing lights, tingling spread)
- Isolated syncope
- Isolated confusion
- Long headache-first events (migraine pattern)
- Seizure-like jerking with postictal state
4) Important Differential Diagnoses (TIA mimics)
- Hypoglycemia
- Focal seizure with Todd’s paresis
- Migraine aura
- Vestibular disorders (for isolated vertigo)
- Functional neurological disorder
- Demyelination
- Peripheral nerve palsy/Bell palsy
5) Risk of Stroke after TIA
- Highest in first 24-48 hours
- Hence TIA is a medical emergency, not a benign event
6) Evaluation of Suspected TIA (Urgent)
A. Immediate
- ABC, pulse, BP, oxygen saturation, temperature
- Bedside glucose
- Focused neurologic exam
- Time of onset / last known well
B. Brain imaging
- MRI brain with diffusion (preferred)
- If unavailable urgently: CT head first (exclude hemorrhage/major lesion)
C. Vascular imaging
- CT angiography/MR angiography of head-neck
- Carotid duplex for extracranial carotid stenosis
D. Cardiac evaluation
- 12-lead ECG
- Continuous rhythm monitoring (look for AF)
- Echocardiography when cardioembolic source suspected
E. Blood tests
- CBC, renal function, electrolytes
- Coagulation profile
- HbA1c, fasting lipid profile
- ESR/CRP in selected vasculitis scenarios
7) ABCD2 Score (for triage support)
- Age ≥60 = 1
- BP ≥140/90 = 1
- Clinical: unilateral weakness = 2; speech only = 1
- Duration: ≥60 min = 2; 10-59 min = 1
- Diabetes = 1
8) Management of TIA (Early Secondary Prevention)
A. Antithrombotic therapy
- Non-cardioembolic TIA
- Antiplatelet therapy
- In high-risk TIA/minor stroke: short-course DAPT (aspirin + clopidogrel), then single agent
- Cardioembolic TIA (e.g., AF)
- Oral anticoagulation (timing individualized by imaging/risk)
B. Lipid management
- High-intensity statin (unless contraindicated)
C. BP and glycemic control
- Long-term target-based treatment
D. Carotid intervention
- Symptomatic significant carotid stenosis: carotid endarterectomy/stenting in selected patients, ideally early
E. Lifestyle
- Stop smoking, reduce alcohol, weight optimization
- Salt reduction, Mediterranean-style diet, regular physical activity
Part B: Acute Management of Stroke in Detail
1) Stroke Types
- Ischemic stroke (most common)
- Intracerebral hemorrhage (ICH)
- Subarachnoid hemorrhage (SAH)
2) Hyperacute Phase (First hour): “Stroke Code”
Step 1: Prehospital and triage
- Recognize FAST/BE-FAST signs
- Pre-notification to stroke center
- Record exact last known well time
Step 2: ED immediate priorities
- Airway, breathing, circulation
- Oxygen only if hypoxemic
- Two IV lines, blood sampling
- Bedside glucose immediately
- NIHSS scoring
- Non-contrast CT head within minutes
- CTA head-neck for vessel occlusion (where protocol allows)
3) Acute Ischemic Stroke Management
A. Reperfusion is core treatment
i) IV Thrombolysis
- Agent: alteplase (or tenecteplase in many centers)
- Typical window: up to 4.5 h in eligible patients
- Must exclude hemorrhage first
- BP should be below threshold before lysis (commonly <185/110)
Major inclusion points
- Measurable disabling neurologic deficit
- Onset within accepted window
- No hemorrhage on imaging
Common exclusion examples
- Intracranial hemorrhage
- Active internal bleeding
- Recent major intracranial event/surgery (as per protocol)
- Severe uncontrolled HTN not responding to treatment
- Coagulation/anticoagulation contraindications per protocol
Post-thrombolysis care
- Neuro and BP monitoring in ICU/stroke unit
- No antiplatelet/anticoagulant for first 24 h
- Repeat CT/MRI at 24 h before antithrombotics
ii) Mechanical Thrombectomy
- For anterior circulation large vessel occlusion (and selected posterior circulation cases)
- Strongest benefit when done early (within 6 h), but selected patients benefit up to 24 h with perfusion mismatch imaging
Eligibility factors
- LVO on CTA/MRA
- Clinical deficit severity
- Salvageable tissue on perfusion/core imaging
- Pre-stroke functional status considerations
B. Antiplatelet and anticoagulation in acute ischemic stroke
- If no thrombolysis: aspirin usually started within 24 h after hemorrhage exclusion
- If thrombolysis done: delay antiplatelet until 24 h scan confirms no bleed
- Anticoagulation for AF-related stroke is delayed based on infarct size/hemorrhagic risk
C. Blood pressure strategy in ischemic stroke
- Candidate for thrombolysis/thrombectomy
- Lower BP to treatment threshold
- Not reperfusion candidate
- Permissive hypertension often allowed initially unless extremely high or specific comorbidity (aortic dissection, ACS, etc.)
D. Glucose, temperature, oxygen, fluids
- Keep glucose in safe range (avoid hypo and severe hyperglycemia)
- Treat fever aggressively
- Maintain euvolemia with isotonic fluids
- Avoid routine oxygen unless saturation low
E. Swallow and nutrition
- Mandatory swallow screen before oral intake
- If unsafe swallow: NG feeding plan
- Prevent aspiration pneumonia
F. DVT prophylaxis and complications
- Intermittent pneumatic compression
- Pharmacologic prophylaxis when safe
- Prevent pressure sores, UTI, aspiration, contractures
4) Acute Intracerebral Hemorrhage (ICH) Management
A. Initial priorities
- Neurocritical care admission
- BP lowering with IV agents as per protocol
- Reverse anticoagulation immediately:
- Warfarin: PCC + vitamin K
- Dabigatran: idarucizumab
- Factor Xa inhibitors: andexanet alfa/PCC (as available)
B. Manage intracranial pressure (ICP)
- Head elevation
- Osmotherapy (mannitol/hypertonic saline) when indicated
- Controlled ventilation strategy in selected severe ICP states
C. Neurosurgical indications (selected)
- Cerebellar hematoma with brainstem compression/hydrocephalus
- Lobar hematoma with mass effect in suitable patients
- External ventricular drain for hydrocephalus/intraventricular extension
D. Seizure management
- Treat clinical/electrographic seizures
- Routine prophylactic AED not universal; individualized
5) Subarachnoid Hemorrhage (SAH) Acute Care
- Secure aneurysm early (coiling/clipping)
- Nimodipine for vasospasm prevention
- Control BP and rebleeding risk
- Treat hydrocephalus (EVD if needed)
- Monitor vasospasm with clinical/TCD surveillance
6) Malignant MCA Infarction / Space-occupying edema
- Monitor for herniation signs
- Consider decompressive hemicraniectomy in appropriate age/timing window
7) Stroke Unit Care (Very Important)
- Stroke physician/neurologist
- Specialized nurses
- Physiotherapist
- Occupational therapist
- Speech and swallow therapist
- Dietician, social worker
8) Early Rehabilitation
- Start early once medically stable
- Mobilization, limb positioning, spasticity prevention
- Speech/swallow/cognitive rehab
- Family counseling and discharge planning
9) Secondary Prevention After Acute Stroke
Determine etiology (TOAST approach)
- Large artery atherosclerosis
- Cardioembolism
- Small vessel occlusion
- Other determined
- Undetermined
Based on etiology:
- Antiplatelet vs anticoagulation
- Statin therapy
- BP control
- Diabetes and sleep apnea management
- Carotid revascularization if indicated
- Lifestyle risk reduction
10) Exam Flowchart (Write directly in answer sheet)
→ If hemorrhage: BP control + anticoagulant reversal + neurosurgical/neurocritical care
→ If ischemic: assess IV thrombolysis eligibility (≤4.5 h) + LVO screening for thrombectomy (up to 24 h selected)
→ stroke unit care (BP, glucose, fever, swallow, DVT prophylaxis, rehab)
→ etiologic workup and secondary prevention.
High-yield Differences: TIA vs Stroke
| Feature | TIA | Stroke |
|---|---|---|
| Deficit duration | Transient | Persistent (>24 h usually) |
| Imaging infarct | Absent (by definition) | Present in ischemic stroke |
| Tissue injury | No permanent infarction | Infarction/hemorrhage |
| Urgency | Emergency (warning event) | Hyperacute emergency |
- Exact thrombolysis inclusion/exclusion checklist (exam table),
- BP targets in ischemic vs hemorrhagic stroke in one chart,
- Drug doses commonly asked in viva (alteplase, antiplatelets, statins, reversal agents).
multi[ple scerosis in detail causes defn pathophysiollogy investigation managment in destil
Multiple Sclerosis (MS) - Detailed
1) Definition
- Disseminated lesions in space (different CNS locations) and time (different time points),
- Variable axonal loss and gliosis,
- Episodes of neurologic dysfunction with or without progression.
2) Etiology and Causes (Risk Factors)
A. Genetic factors
- HLA association, especially HLA-DRB1*15:01
- Family history increases risk
- Polygenic susceptibility (immune-regulation genes)
B. Environmental factors
- Epstein-Barr virus (EBV) infection strongly associated
- Low vitamin D / low sunlight exposure
- Higher latitude residence
- Smoking (increases risk and worsens progression)
- Adolescent obesity (notably in females)
- Possible microbiome contributions
C. Sex and age
- More common in females (especially relapsing forms)
- Typical onset: 20-40 years
3) Pathophysiology (Detailed)
Core process
- Peripheral immune activation (autoreactive T and B cells)
- Blood-brain barrier disruption
- CNS infiltration by immune cells
- Inflammation against myelin and oligodendrocytes
- Demyelination and conduction block
- Axonal transection/neurodegeneration
- Gliosis and plaque (sclerosis) formation
Immune mechanisms
- CD4+ T cells (Th1/Th17) drive inflammation
- CD8+ T cells contribute to axonal injury
- B cells/plasma cells produce intrathecal antibodies (oligoclonal bands)
- Microglial activation and cytokines amplify damage
Why symptoms vary
- Optic nerve -> optic neuritis
- Brainstem/cerebellum -> diplopia, ataxia, vertigo
- Spinal cord -> weakness, sensory level, sphincter symptoms
- Periventricular/cortical pathways -> cognitive and motor deficits
Disease evolution
- Early: inflammatory relapses with partial recovery (remyelination possible)
- Later: neurodegeneration dominates, progressive disability
4) Clinical Types of MS
-
Clinically Isolated Syndrome (CIS)
- First demyelinating event suggestive of MS
-
Relapsing-Remitting MS (RRMS) (most common initially)
- Relapses (attacks) with remission
-
Secondary Progressive MS (SPMS)
- Starts as RRMS, then gradual progression with fewer clear relapses
-
Primary Progressive MS (PPMS)
- Progressive disability from onset (no clear early relapses)
5) Clinical Features
Common presenting syndromes
- Optic neuritis: painful visual loss, color desaturation, RAPD
- Partial transverse myelitis: limb weakness, sensory deficits, bladder symptoms
- Brainstem syndrome: diplopia, internuclear ophthalmoplegia
- Cerebellar signs: ataxia, intention tremor
- Sensory symptoms: paresthesia, numbness
- Lhermitte sign: electric shock sensation on neck flexion
- Uhthoff phenomenon: worsening with heat
Chronic symptoms
- Fatigue
- Spasticity
- Neuropathic pain
- Cognitive dysfunction
- Depression/anxiety
- Sexual dysfunction
- Bladder, bowel problems
- Gait impairment
6) Diagnosis
A. MRI brain and spinal cord (key investigation)
- Periventricular
- Juxtacortical/cortical
- Infratentorial
- Spinal cord (short segment, peripheral)
- T2/FLAIR hyperintense plaques
- Gadolinium-enhancing active lesions
- Simultaneous enhancing + non-enhancing lesions suggest DIT
B. CSF analysis
- Oligoclonal IgG bands (unmatched in serum) support MS
- Elevated IgG index may be present
C. Evoked potentials
- Visual evoked potential may show delayed conduction (optic pathway demyelination)
D. Blood tests (to exclude mimics)
- B12 deficiency
- ANA/autoimmune profile
- Aquaporin-4 IgG (NMOSD)
- MOG-IgG disease testing
- Infections (HIV, syphilis etc. as indicated)
- Thyroid and metabolic testing as needed
7) Differential Diagnosis
- NMOSD
- MOG antibody-associated disease
- Acute disseminated encephalomyelitis (ADEM)
- CNS vasculitis
- Neurosarcoidosis
- B12 deficiency myelopathy
- Lyme/HIV/syphilis-related CNS disease
- Small vessel ischemic white matter disease (older patients)
8) Management in Detail
- Treat acute relapse
- Disease-modifying therapy (DMT)
- Symptomatic treatment
- Rehabilitation and long-term monitoring
8.1 Acute Relapse Treatment
Definition of relapse
First-line
- High-dose corticosteroids
- IV methylprednisolone (commonly 1 g/day for 3-5 days) or equivalent oral high-dose regimen
- Goal: shorten relapse duration (does not change long-term course markedly)
If severe and steroid-refractory
- Plasma exchange (especially severe motor/brainstem/optic relapse)
Important
- Rule out pseudo-relapse (UTI, fever, heat exposure, metabolic causes)
8.2 Disease-Modifying Therapy (DMT)
- Reduce relapse rate
- Reduce MRI activity
- Delay disability progression
Classes and examples
A. Injectable
- Interferon beta preparations
- Glatiramer acetate
B. Oral
- Dimethyl fumarate
- Teriflunomide
- S1P modulators (e.g., fingolimod, siponimod, ozanimod, ponesimod)
- Cladribine (immune reconstitution strategy)
C. Monoclonal antibodies (high efficacy)
- Natalizumab
- Anti-CD20 therapies (ocrelizumab, ofatumumab, rituximab off-label in many settings)
- Alemtuzumab (selected use)
Choosing DMT depends on:
- Disease activity and prognostic risk
- MRI burden
- Comorbidities and infection risk
- Pregnancy planning
- Monitoring capability and patient preference
Escalation vs early high-efficacy approach
- Traditional: start moderate-efficacy and escalate
- Increasingly used: early high-efficacy in aggressive disease
8.3 Symptomatic Management (Very Important)
- Spasticity: physiotherapy, baclofen/tizanidine
- Neuropathic pain: gabapentin/pregabalin/duloxetine etc.
- Fatigue: energy conservation, sleep optimization, selected pharmacologic options
- Bladder dysfunction:
- urgency: antimuscarinic/beta-3 agonist
- retention: intermittent self-catheterization
- Gait difficulty: rehab, assistive devices, fall prevention
- Depression/anxiety: counseling + pharmacotherapy
- Cognitive issues: cognitive rehab, routines
- Sexual dysfunction: targeted therapy and counseling
8.4 Rehabilitation and Supportive Care
- Multidisciplinary team care:
- Neurology, physiotherapy, occupational therapy, speech therapy, psychology, urology
- Exercise program and balance training
- Vitamin D optimization
- Smoking cessation
- Vaccination planning (timed around immunotherapy)
- Bone health and fall-risk reduction
9) Monitoring
Clinical
- Relapses, disability progression (e.g., EDSS), mobility, cognition, QoL
MRI
- Baseline and periodic follow-up for new/enhancing lesions
Safety labs (drug-specific)
- CBC, liver function, renal function
- Lymphocyte counts
- Infection screening (HBV, TB, JCV antibody depending on DMT)
- Immunoglobulin levels for B-cell depleting therapy
10) Complications and Prognosis
Complications
- Progressive disability
- Spastic paraparesis
- Bladder complications/recurrent UTIs
- Cognitive decline
- Depression
- Treatment-related infections/PML risk (drug-specific)
Prognosis
- Relapsing onset
- Sensory/optic onset
- Good recovery from early relapses
- Lower early lesion burden
- Progressive onset
- Frequent early relapses
- Early motor/cerebellar involvement
- High MRI lesion load/spinal lesions
11) Pregnancy and MS (high-yield)
- Relapse rate often decreases during pregnancy, rises postpartum.
- Many DMTs require preconception planning.
- Individualized counseling for contraception, conception, breastfeeding, and postpartum restart of therapy.
12) Exam-ready short summary
Diagnosis is based on clinical syndromes + MRI ± CSF oligoclonal bands after excluding mimics.
Treatment includes:
- Acute relapse steroids,
- Long-term DMTs to prevent relapses/progression,
- Symptomatic and rehabilitative care.
- McDonald criteria in a simple table,
- Drug-wise DMT comparison chart (mechanism, route, major adverse effects, monitoring),
- Flowchart for diagnosis and management for exam writing.