Explain gastritis
Reading File
Finding Sources
Searching PubMed
gastritis classification pathogenesis treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching Images
gastritis histology H pylori chronic gastritis
I now have comprehensive content. Let me compose a thorough, well-structured answer.
Gastritis
Gastritis is histologically documented inflammation of the gastric mucosa. It should not be equated with mucosal erythema seen on endoscopy, nor used interchangeably with "dyspepsia." When inflammatory cells are absent or rare, the correct term is gastropathy. Crucially, the correlation between histologic gastritis, symptoms, and endoscopic appearance is poor — there is no single typical clinical presentation. — Harrison's Principles of Internal Medicine, 22nd Ed.
Classification
Gastritis is classified by:
| Axis | Categories |
|---|---|
| Time course | Acute vs. chronic |
| Histology | Neutrophilic, lymphocytic, eosinophilic, granulomatous, etc. |
| Anatomic distribution | Antral-predominant, body-predominant, pangastritis |
| Pathogenic mechanism | Infectious, autoimmune, chemical/reactive |
Major Types
- Acute gastritis (infectious & other causes)
- Chronic atrophic gastritis
- Type A: Autoimmune, body-predominant
- Type B: H. pylori–related, antral-predominant
- Uncommon forms: lymphocytic, eosinophilic, granulomatous (Crohn's, sarcoidosis), Russell body gastritis
Acute Gastritis
Acute gastritis is characterized by dense neutrophilic infiltration of the gastric mucosa. It is rare compared to chronic forms.
Causes
- H. pylori (most common infectious cause): sudden onset epigastric pain, nausea, vomiting; marked neutrophilic infiltrate with edema and hyperemia. Hypochlorhydria may persist up to 1 year. If untreated, evolves to chronic gastritis.
- Phlegmonous (suppurative) gastritis: rare, life-threatening. Infection of the gastric submucosa and muscularis propria. Organisms: streptococci, staphylococci, E. coli, Proteus, Haemophilus, anaerobes. Risk factors: alcohol excess, AIDS, immunosuppression, recent GI surgery. Presents with sepsis, acute upper abdominal pain, peritonitis, and hypotension.
- Emphysematous gastritis: gas-producing organisms (Clostridium perfringens, E. coli, S. aureus) → gas in the stomach wall and portal venous system (seen on CT/plain films). Potentially fatal.
- Other: mycobacterial, syphilitic, viral (e.g., CMV), parasitic, fungal.
- Chemical/reactive: NSAIDs, alcohol, bile reflux, corrosive ingestion.
Any agent that interferes with the gastric mucosal protective barrier (mucus layer, bicarbonate, prostaglandins, blood flow) can cause acute gastritis. — Robbins & Cotran Pathologic Basis of Disease
Chronic Gastritis
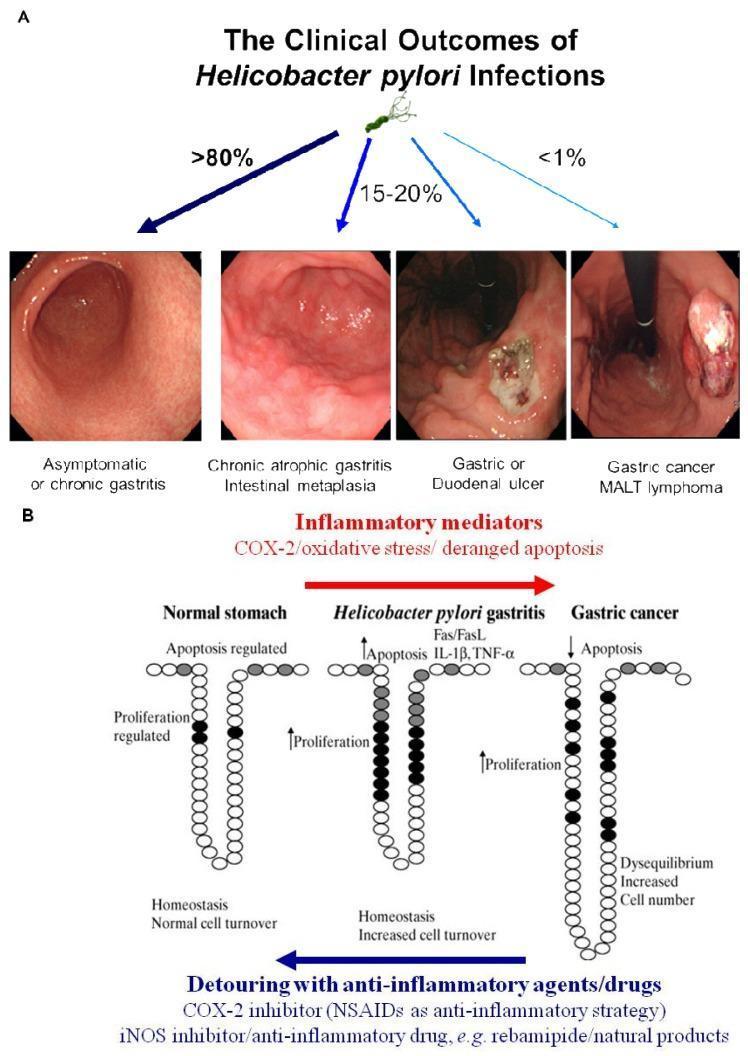
Much more common than acute gastritis, often clinically silent. Prevalence is declining in developed countries. Its primary clinical importance lies in being a risk factor for peptic ulcer disease (PUD), gastric adenocarcinoma, and MALToma (B-cell lymphoma).
1. H. pylori (Hp) Gastritis — Type B
H. pylori is the most common cause of chronic gastritis worldwide.
- The organism: Gram-negative, helical/spiral-shaped, flagellated bacterium.
- Epidemiology: >50% of the world's population is infected; 70–80% in developing countries. Childhood acquisition is the major risk factor, linked to low socioeconomic status and poor sanitation.
- Pattern: Diffuse antral gastritis initially (antral-predominant).
- Mechanism: Chemokines induced by Hp create a persistent mixed infiltrate — neutrophils (active inflammation) + lymphocytes, plasma cells, macrophages (chronic inflammation). Despite the robust immune response, bacteria persist in most people without treatment.
- Progression:
- Antrum → pangastritis → body involvement → glandular atrophy
- Atrophy → reduced acid (achlorhydria) in advanced disease
- Intestinal metaplasia → risk factor for gastric adenocarcinoma
- MALT acquisition → MALToma (B-cell lymphoma that can regress with Hp eradication)
- Associated with increased acid production early on → predisposing to peptic ulcer disease
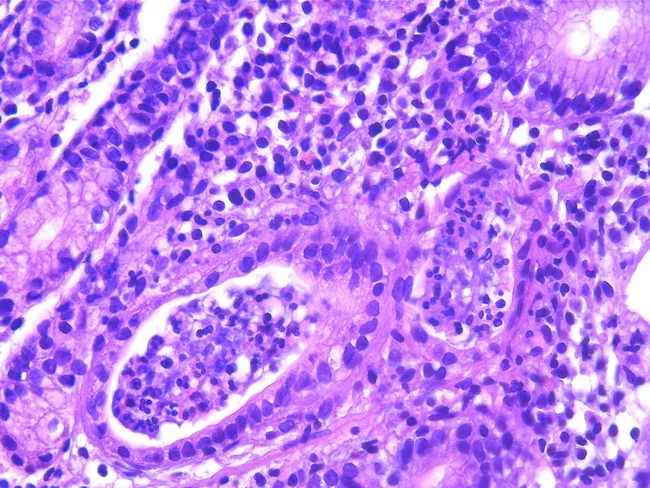
2. Autoimmune Metaplastic Atrophic Gastritis (AMAG) — Type A
- After H. pylori and NSAIDs, the most frequent cause of chronic gastritis.
- Target: Oxyntic (parietal cell–rich) glands of the gastric body/fundus.
- Autoantibodies: Anti-parietal cell antibodies (anti-H⁺/K⁺-ATPase) and anti-intrinsic factor antibodies.
- Consequences:
- Destruction of parietal cells → ↓ gastric acid (achlorhydria)
- ↓ Intrinsic factor → vitamin B₁₂ malabsorption → pernicious anemia
- Loss of negative feedback on G cells → antral G-cell hyperplasia and hypergastrinemia
- Intestinal metaplasia → risk of gastric adenocarcinoma
3. Environmental Metaplastic Atrophic Gastritis (EMAG)
- Associated with H. pylori, diet, and environmental factors.
- Patchy atrophy and intestinal metaplasia starting in the antrum, spreading proximally.
- Higher gastric cancer risk than antrum-only disease.
Intestinal Metaplasia — Shared Complication
Intestinal metaplasia develops in all forms of chronic gastritis and is a recognized precancerous lesion (Correa cascade: chronic gastritis → atrophic gastritis → intestinal metaplasia → dysplasia → adenocarcinoma). — Robbins & Cotran Pathologic Basis of Disease
Uncommon Forms
| Type | Key Features |
|---|---|
| Lymphocytic gastritis | Dense intraepithelial lymphocytosis; associated with celiac disease, H. pylori; endoscopy shows "varioliform" pattern |
| Eosinophilic gastritis | Eosinophilic infiltration; may be allergic/food-driven |
| Granulomatous | Crohn's disease, sarcoidosis, syphilis, fungal infections |
| Russell body gastritis | Plasma cells with Russell bodies; often H. pylori-associated |
| Ménétrier disease | Excessive TGF-α secretion → diffuse foveolar hyperplasia → protein-losing gastropathy; rare |
| Collagenous gastritis | Subepithelial collagen band deposition |
Associated Conditions
| Condition | Mechanism |
|---|---|
| Peptic ulcer disease | H. pylori–induced hyperchlorhydria → duodenal/gastric ulcers |
| Gastric adenocarcinoma | Chronic atrophic gastritis → intestinal metaplasia → dysplasia |
| MALToma | H. pylori–driven MALT acquisition → B-cell lymphoma |
| Pernicious anemia | AMAG → intrinsic factor loss → B₁₂ deficiency |
| Zollinger-Ellison syndrome | Gastrin-secreting tumor → parietal cell hyperplasia → acid hypersecretion (60–90% of gastrinomas are malignant) |
Management Principles
- H. pylori gastritis: Eradication therapy (triple or quadruple regimens with PPI + antibiotics). H. pylori eradication also leads to regression of gastric MALToma in early-stage disease.
- Autoimmune gastritis: Vitamin B₁₂ supplementation (intramuscular or high-dose oral); surveillance endoscopy for gastric cancer/neuroendocrine tumors.
- NSAID-induced: Discontinue offending agent; PPIs for mucosal healing.
- Acute phlegmonous/emphysematous gastritis: Broad-spectrum IV antibiotics; surgical intervention (gastrectomy) if refractory.
Recent evidence note: A 2024 meta-analysis (PMID: 39710669) reports globally rising H. pylori antibiotic resistance in children, which has implications for empiric triple-therapy regimens. A 2025 meta-analysis (PMID: 40822578) highlights significant H. pylori–gut microbiome interactions that may modulate disease outcomes beyond gastritis alone.
Sources: Robbins & Cotran Pathologic Basis of Disease; Harrison's Principles of Internal Medicine, 22nd Ed.; Sleisenger & Fordtran's Gastrointestinal and Liver Disease.
This is a shared conversation. Sign in to Orris to start your own chat.