L gbs
Reading File
search_medical_text
Guillain-Barré syndrome GBS pathophysiology clinical features diagnosis
search_medical_text
Guillain-Barré syndrome GBS treatment management IVIG plasmapheresis
search_medical_image
Guillain-Barré syndrome nerve conduction CSF findings
GradeResponse
GradeResponse
GradeResponse
Guillain-Barré Syndrome (GBS)
Overview
GBS is an acute, autoimmune polyradiculoneuropathy — frequently severe and potentially fulminant. It occurs year-round at 10–20 cases per million annually (~5,000–6,000 cases/year in the US). Males are slightly more affected than females; in Western countries, adults more than children. (Harrison's, p. 12718)
Pathophysiology
GBS is triggered by molecular mimicry: a preceding infection induces antibodies that cross-react with peripheral nerve gangliosides and myelin antigens.
Common precipitants (1–4 weeks prior):
- Campylobacter jejuni (most common, ~30%)
- Cytomegalovirus (CMV)
- Epstein-Barr virus (EBV)
- Mycoplasma pneumoniae
- COVID-19, Zika virus
- Vaccines (rare)
Subtypes
| Subtype | Mechanism | Key Feature |
|---|---|---|
| AIDP (most common in West) | Demyelinating | Slow conduction velocity |
| AMAN | Axonal, motor | Anti-GM1/GD1a antibodies |
| AMSAN | Axonal, motor + sensory | Worse prognosis |
| Miller Fisher Syndrome | Anti-GQ1b antibodies | Ophthalmoplegia, ataxia, areflexia |
| Bickerstaff's encephalitis | Anti-GQ1b, CNS involvement | Altered consciousness |
Clinical Features
Progression: Ascending weakness + sensory symptoms → nadir by 4 weeks
- Motor: Bilateral limb weakness (legs → arms), facial diplegia
- Sensory: Paresthesias, pain (back/leg pain common early)
- Autonomic: Tachycardia, BP fluctuation, urinary retention, ileus (life-threatening)
- Respiratory: Diaphragm weakness → respiratory failure in ~25–30%
- Reflexes: Areflexia/hyporeflexia is a hallmark
Plateau phase typically lasts 2–4 weeks, then gradual recovery.
Diagnosis
CSF (Lumbar Puncture)
- Classic finding: Albuminocytologic dissociation
- Elevated protein (>45 mg/dL, often >100)
- Normal or near-normal cell count (<10 WBC/µL)
- Seen in ~80% at 2 weeks
Electrodiagnostics (NCS/EMG)
- AIDP: Prolonged distal latencies, slowed conduction velocity, conduction block, prolonged F-waves
- AMAN/AMSAN: Reduced CMAP amplitudes with relatively preserved conduction velocity
Antibody Testing
- Anti-GQ1b → Miller Fisher Syndrome
- Anti-GM1 → AMAN
- Anti-GD1b → sensory GBS
MRI
Gadolinium enhancement and thickening of nerve roots (cauda equina) is a classic radiological finding:
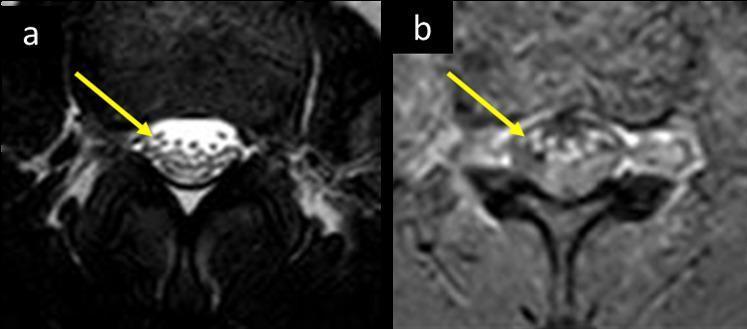
Management
ICU Monitoring Criteria (admit all suspected GBS)
- Serial FVC monitoring — intubate if FVC <20 mL/kg, MIP <30 cmH₂O, MEP <40 cmH₂O ("20-30-40 rule")
- Continuous cardiac monitoring (autonomic instability)
- DVT prophylaxis
Immunotherapy
Initiated as soon as possible — within ~2 weeks of first motor symptoms (Harrison's, p. 12730):
| Therapy | Dose | Notes |
|---|---|---|
| IVIg (preferred first-line) | 2 g/kg total over 5 days | Easy to administer; good safety profile |
| Plasmapheresis (PLEX) | 5 exchanges over 2 weeks | Equally effective |
| Combination | Not recommended | No added benefit over either alone |
Steroids (corticosteroids) are NOT effective in GBS and not recommended.
Supportive Care
- Pain management (gabapentin, carbamazepine, opioids for severe cases)
- Bowel/bladder care
- Physical and occupational therapy
- Nutritional support (NG/PEG if bulbar dysfunction)
Prognosis
- ~85% walk independently at 6 months
- ~5–10% mortality (respiratory failure, autonomic instability, sepsis)
- ~20% have significant residual disability at 1 year
- Predictors of poor outcome: older age, rapid progression, axonal subtype (AMAN/AMSAN), preceding C. jejuni infection, low CMAP amplitudes
EGOS/mEGOS score can estimate probability of independent ambulation at 6 months.
Key Differentiators from Mimics
| Feature | GBS | Transverse Myelitis | CIDP | MG |
|---|---|---|---|---|
| Reflexes | Absent | Absent/hyperreflexia | Reduced | Normal |
| Sensory level | No | Yes | Variable | No |
| CSF protein | High | Variable | High | Normal |
| Progression | <4 weeks | Hours–days | >8 weeks | Fatigable |
| Autonomic | Common | Bladder/bowel | Rare | Rare |
This is a shared conversation. Sign in to Orris to start your own chat.