Hi
Hi! How can I help you today?
Multiple myeloma
Asking for Preferences
Reading File
Finding Sources
Searching PubMed
"multiple myeloma"[MeSH Terms] AND "therapy"[MeSH Terms]
Reading File
Reading File
Searching PubMed
"multiple myeloma" AND treatment
Finding Sources
Finding Sources
Searching Images
multiple myeloma bone marrow plasma cells histology
Reading File
Searching Images
multiple myeloma skull X-ray punched out lytic lesions
Reading File
Reading File
I now have comprehensive information to provide a thorough answer. Let me compile it.
Multiple Myeloma — Comprehensive Overview
Definition
Multiple myeloma (MM) is a malignancy of clonal plasma cells characterized by bone marrow infiltration and extensive skeletal destruction resulting in anemia, bone pain, and fractures. It is defined by all three of:
- ≥10% clonal plasma cells on bone marrow biopsy
- Monoclonal (M) protein in serum or urine (absent in nonsecretory myeloma)
- One or more myeloma-defining events (CRAB criteria or high-risk biomarkers)
— Goldman-Cecil Medicine
Epidemiology
- Accounts for ~1% of all malignancies and >10% of hematologic malignancies in the US
- Annual incidence ~4 per 100,000; ~30,000 new cases/year in the US
- Median age at diagnosis: 65–70 years; <2% of patients are under 40
- More common in males and in people of African descent (incidence nearly 2× that of Whites)
Pathogenesis
- Chromosomal translocations fusing the IgH locus (chromosome 14) to oncogenes (e.g., cyclin D1, cyclin D3) are common — dysregulation of D cyclins drives proliferation
- IL-6 (from marrow stromal fibroblasts and macrophages) is a major survival/proliferation cytokine for myeloma cells
- Late-stage disease may show MYC translocations, indicating aggressive phenotype
Bone Destruction Mechanism
Myeloma cells upregulate RANKL expression on bone marrow stromal cells → osteoclast activation + osteoblast inhibition → net bone resorption → hypercalcemia + pathologic fractures
— Robbins & Kumar Basic Pathology
Clinical Features (CRAB)
| Feature | Details |
|---|---|
| C — HyperCalcemia | 15–20% at diagnosis; from osteoclast-driven bone resorption |
| R — Renal failure | Creatinine >2 mg/dL in 20% at diagnosis |
| A — Anemia | Marrow replacement by plasma cells |
| B — Bone lesions | Lytic lesions, osteoporosis, pathologic fractures |
Additional features: recurrent bacterial infections (due to impaired humoral immunity), hyperviscosity syndrome, peripheral neuropathy.
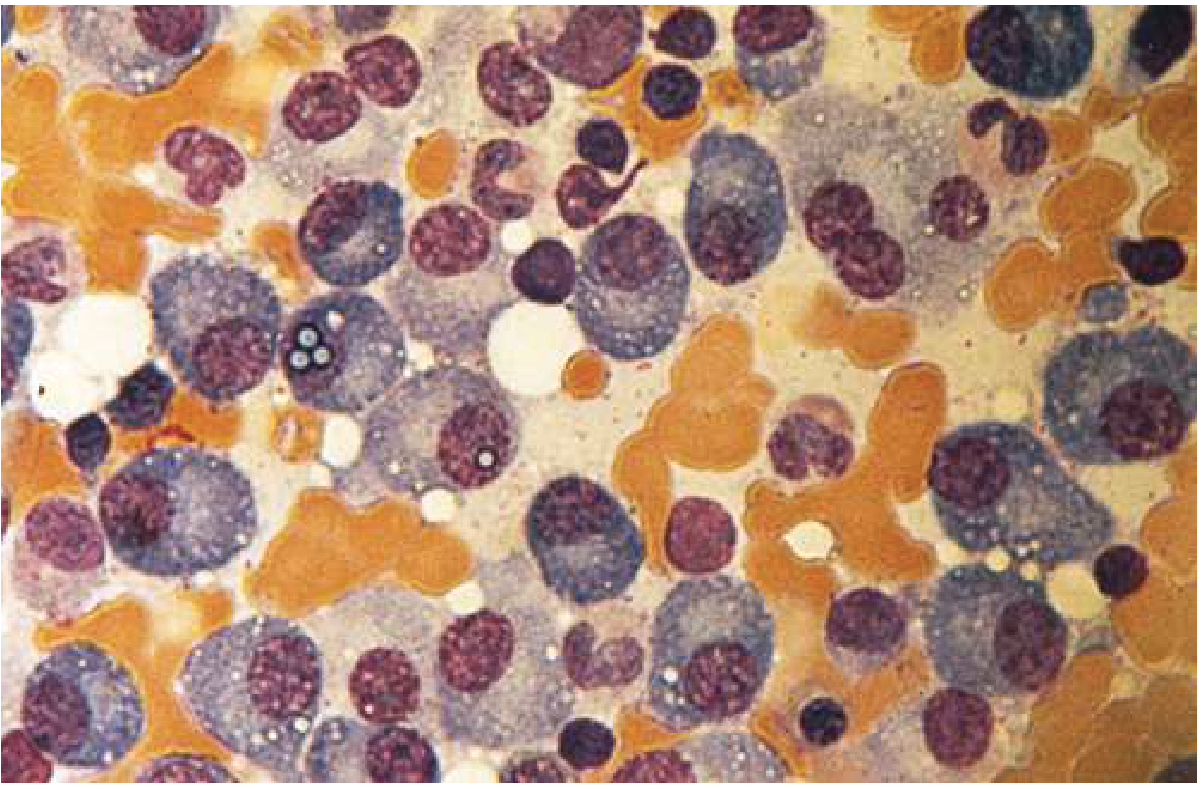
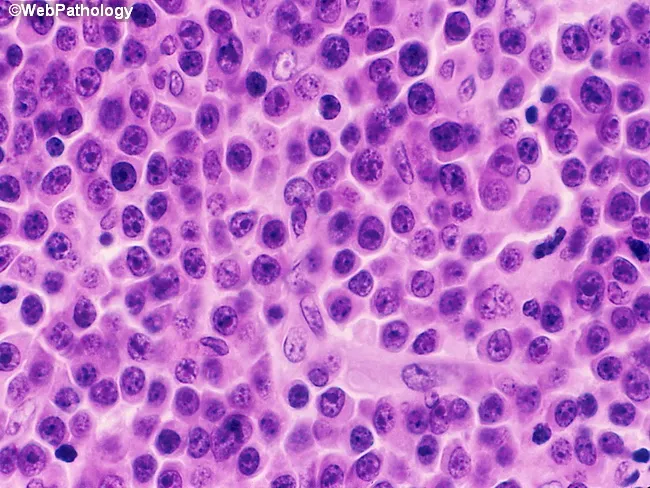
Histology & Morphology
Bone marrow shows >30% plasma cells (normal <5%). Myeloma cells may resemble normal plasma cells or show abnormal features: prominent nucleoli, binucleation, and Russell bodies (cytoplasmic immunoglobulin inclusions).
Bone marrow aspirate showing predominance of plasma cells:
H&E biopsy — sheets of malignant plasma cells replacing marrow:
M Protein Distribution
| Immunoglobulin | Frequency |
|---|---|
| IgG | 60% |
| IgA | 20–25% |
| Light chain only (κ or λ) | Remaining cases |
| IgM, IgD, IgE | Rare |
Diagnosis & Staging
Diagnostic Criteria
- ≥10% clonal plasma cells on BM biopsy
- M protein in serum/urine
- CRAB criteria OR SLiM (60% clonal plasma cells; serum FLC ratio ≥100; >1 focal lesion on MRI)
Revised International Staging System (R-ISS)
| Stage | Criteria | Median OS |
|---|---|---|
| I | β2-microglobulin <3.5 mg/L + albumin ≥3.5 g/dL; no high-risk cytogenetics; normal LDH | Not reached |
| II | Not I or III | ~83 months |
| III | β2-microglobulin >5.5 mg/L + high-risk cytogenetics [del(17p), t(4;14), t(14;16)] or elevated LDH | ~43 months |
MGUS → Smoldering MM → MM Spectrum
| Condition | Risk of Progression at 20 yr |
|---|---|
| Low-risk MGUS (IgG <1.5 g/dL, normal FLC ratio) | 5% |
| High-risk MGUS (all 3 risk factors abnormal) | 58% |
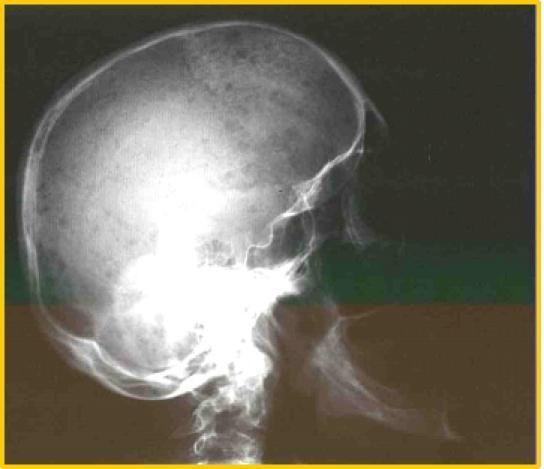
Radiologic Findings
Skeletal survey shows "punched-out" lytic lesions in ~80% of patients. Most common sites: vertebral column, skull, ribs, pelvis, proximal humerus and femur.
Classic "pepper-pot skull" on lateral X-ray:
PET scan showing extensive bony/extramedullary disease (A) and response to chemotherapy (B):
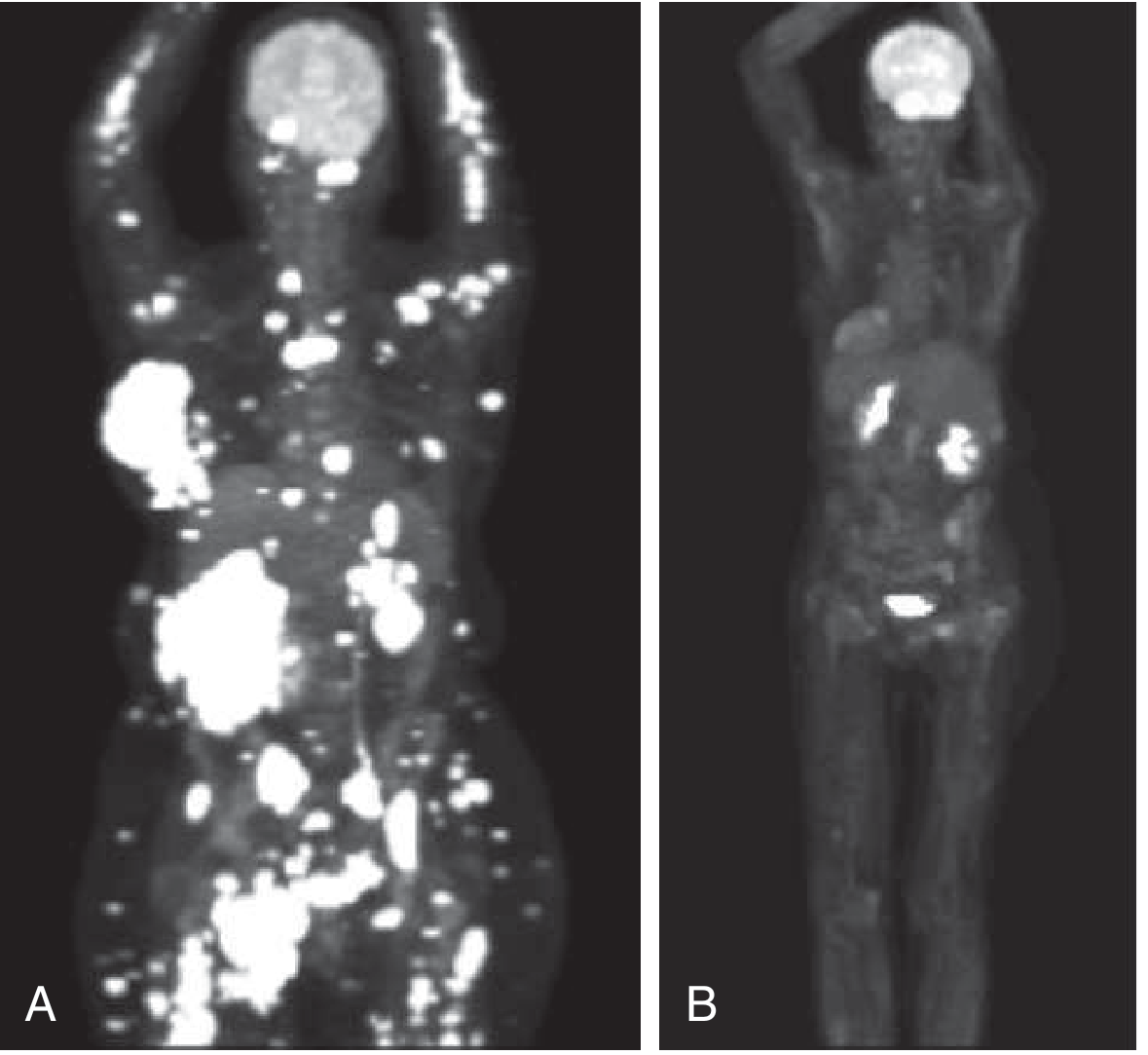
Low-dose whole-body CT and PET are now preferred over plain X-rays for assessing bone disease. MRI is used for spinal involvement or uncertain disease burden.
Complications
Renal
- Light chain cast nephropathy ("myeloma kidney"): waxy laminated casts in distal/collecting tubules from precipitated Bence Jones proteins
- Hypercalcemia-induced renal insufficiency
- AL amyloidosis (~10% of MM), light chain deposition disease, acquired Fanconi syndrome
Immune
- Humoral immune deficiency despite elevated total immunoglobulin (M protein)
- High risk of bacterial infections (e.g., S. pneumoniae, H. influenzae)
Neurologic
- Spinal cord compression from vertebral collapse or plasmacytoma
- Peripheral neuropathy (disease-related or bortezomib-induced)
- Hyperviscosity → headache, visual changes
Treatment
Transplant-Eligible Patients (generally <70 yrs, good performance status)
- Induction (3–4 cycles): VRd (Bortezomib + Lenalidomide + Dexamethasone) — the standard regimen; or Dara-VRd (Daratumumab added) for deeper responses
- Stem cell mobilization + Autologous HSCT (high-dose melphalan conditioning)
- Maintenance: Lenalidomide 10 mg/day (improves OS); add Bortezomib every 2 wk in high-risk disease
Transplant-Ineligible Patients (~50%)
- VRd for ~9 months → Lenalidomide maintenance, OR
- DRd (Daratumumab + Lenalidomide + Dexamethasone) until progression
Relapsed/Refractory MM
Combinations from these drug classes:
| Class | Examples |
|---|---|
| Proteasome inhibitors | Bortezomib, Carfilzomib, Ixazomib |
| Immunomodulatory drugs (IMiDs) | Lenalidomide, Pomalidomide, Thalidomide |
| Anti-CD38 monoclonals | Daratumumab, Isatuximab |
| Anti-SLAMF7 | Elotuzumab |
| Alkylating agents | Cyclophosphamide, Melphalan |
| CAR-T therapy | Idecabtagene vicleucel (ide-cel), Ciltacabtagene autoleucel (cilta-cel) — targeting BCMA |
| Bispecific antibodies | Teclistamab (BCMA × CD3) |
Re-induction with the original regimen is appropriate if relapse occurs >6 months after stopping therapy.
Supportive Care
| Problem | Management |
|---|---|
| Bone pain / lytic lesions | Bisphosphonates (zoledronic acid), denosumab; radiotherapy for focal lesions |
| Hypercalcemia | IV hydration, bisphosphonates, corticosteroids |
| Anemia | Erythropoiesis-stimulating agents, transfusion |
| Infections | Pneumococcal/influenza vaccination; IVIG for recurrent serious infections |
| VTE (from IMiDs) | Aspirin or LMWH prophylaxis |
| Neuropathy (bortezomib) | Switch to subcutaneous administration; dose adjustment |
Prognosis
Median overall survival has improved dramatically with modern therapy — from ~3 years in the pre-novel-agent era to >10 years for standard-risk patients with access to full treatment sequencing including CAR-T. High-risk cytogenetics [del(17p), t(4;14), t(14;16)] confer a significantly worse prognosis.
Recent Evidence
- CEPHEUS trial (2025) — Phase 3 RCT: Daratumumab + VRd showed superior outcomes over VRd alone in transplant-ineligible or transplant-deferred newly diagnosed MM (Nat Med, 2025) [PMID: 39910273]
- A 2024 Nature Reviews Disease Primers review [PMID: 38937492] provides an updated comprehensive overview of MM biology and management
Sources: Robbins & Kumar Basic Pathology; Goldman-Cecil Medicine; The Washington Manual of Medical Therapeutics
This is a shared conversation. Sign in to Orris to start your own chat.