Tympanic membrane perforation classification
Reading File
Finding Sources
Finding Sources
Searching the Web
tympanic membrane perforation classification types
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Searching Images
tympanic membrane perforation types central marginal
Loading Image
Loading Image
Loading Image
I now have comprehensive information to provide a thorough answer on tympanic membrane perforation classification. Let me compile this.
Tympanic Membrane Perforation — Classification
1. By Location (Most Clinically Important)
A. Central Perforation
- Involves only the pars tensa; the annulus (fibrous ring) is intact on all sides
- Does not reach the bony canal margin
- Typically caused by acute otitis media (AOM) or trauma
- Generally safe — lower risk of cholesteatoma
- Located in any quadrant of the pars tensa (anterior, posterior, inferior, or subtotal)
B. Marginal Perforation
- Reaches the annulus — the fibrous ring is disrupted
- The edge of the perforation abuts the bony canal wall
- Higher risk of cholesteatoma because squamous epithelium can migrate into the middle ear through the disrupted annular margin
- Most dangerous when in the posterosuperior quadrant (close to ossicular chain, facial nerve)
C. Attic (Pars Flaccida) Perforation
- Located in the pars flaccida (above the notch of Rivinus, superiorly)
- The pars flaccida is less rigid and lacks the fibrous layer of the pars tensa
- Strongly associated with primary acquired cholesteatoma — keratin debris accumulates as the membrane retracts into the epitympanum
- Even a small perforation here carries significant pathological significance
2. By Aetiology
| Cause | Typical Features |
|---|---|
| Acute otitis media (AOM) | Spontaneous rupture; pars tensa; heals in ~90% without intervention |
| Chronic otitis media (COM) | Persistent perforation; hallmark of chronic disease; depends on Eustachian tube function for repair |
| Trauma (blast/barotrauma/blunt) | Almost always pars tensa, usually anteriorly or inferiorly; often heals spontaneously |
| Penetrating trauma | Higher risk of ossicular chain injury; posterosuperior quadrant involvement warrants urgent ENT referral |
| Iatrogenic | e.g., during irrigation, myringotomy, tympanostomy tube extrusion |
3. By Size
Clinically graded to guide surgical planning:
| Grade | Description |
|---|---|
| Small | < 25% of pars tensa area (< 1 quadrant) |
| Medium | 25–50% (1–2 quadrants) |
| Large | 50–75% (2–3 quadrants) |
| Subtotal | > 75%; annulus still partially intact |
| Total | Complete destruction of the TM; annulus entirely absent |
Larger perforations cause greater conductive hearing loss (up to ~35–40 dB HL) due to:
- Loss of the areal ratio between TM and oval window
- Disruption of the catenary lever effect
- Sound reaching both windows simultaneously (phase cancellation)
4. By Chronicity
- Acute — < 3 months duration; high rate of spontaneous healing
- Chronic — > 3 months; unlikely to heal without surgical intervention; associated with COM
5. Safe vs. Unsafe Perforation (Classic ENT Distinction)
| Safe (Tubotympanic) | Unsafe (Atticoantral) | |
|---|---|---|
| Location | Central, pars tensa | Marginal or attic, pars flaccida |
| Cholesteatoma risk | Low | High |
| Discharge | Mucoid, intermittent | Scanty, foul-smelling, persistent |
| Hearing loss | Moderate conductive | Variable; SNHL if complications |
| Management | Myringoplasty ± tympanoplasty | Modified radical or radical mastoidectomy |
| Complications | Less common | Intracranial complications more likely |
Clinical Images
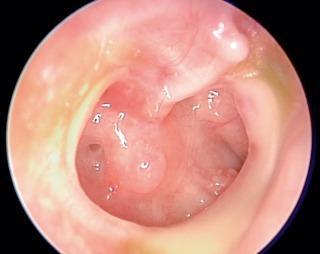
Central perforation — well-demarcated oval defect in the pars tensa with intact annulus:
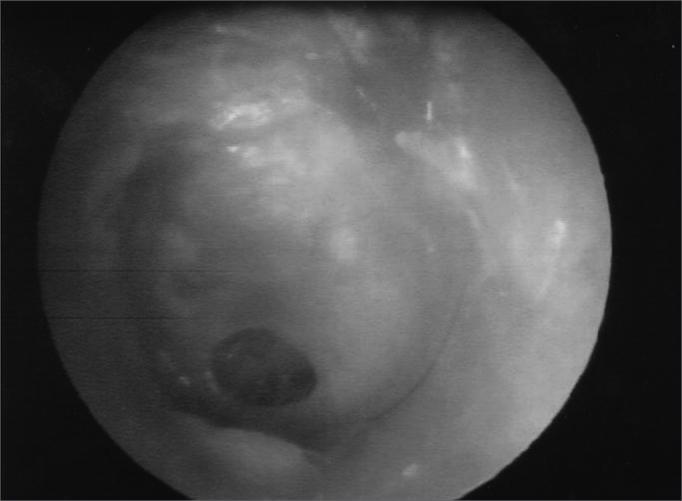
Large central mesotympanic perforation — intact annulus fibrosus, erythematous middle ear mucosa visible through the defect:
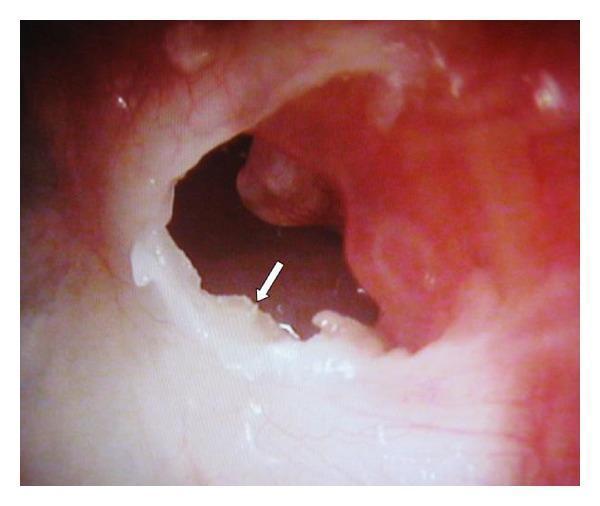
Marginal perforation — posterosuperior defect reaching the annulus with keratin debris (cholesteatoma):
Key Clinical Pearls
- Posterosuperior quadrant perforations — even if seemingly small — warrant urgent ENT referral due to proximity to the incudostapedial joint, facial nerve canal, and higher cholesteatoma risk
- Attic perforations can be deceptively small on otoscopy but harbour extensive cholesteatoma medially
- Successful myringoplasty/tympanoplasty depends critically on Eustachian tube function; ongoing dysfunction (OME, negative middle ear pressure in the contralateral ear) predicts poorer outcomes
- Spontaneous healing rates: traumatic > AOM-related > COM-related perforations
Sources: Cummings Otolaryngology Head and Neck Surgery; Tintinalli's Emergency Medicine; K.J. Lee's Essential Otolaryngology
Cholesteatoma
Finding Sources
Finding Sources
Searching the Web
cholesteatoma classification pathogenesis management 2024
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
cholesteatoma otoscopy middle ear erosion
Reading File
Loading Image
Loading Image
Reading File
Loading Image
Loading Image
I now have comprehensive material from Cummings Otolaryngology. Let me compile the full answer.
Cholesteatoma
Definition
An aural cholesteatoma is an epidermal inclusion of the middle ear or mastoid — a cystic or sheet-like accumulation of keratinizing squamous epithelium within the middle ear cleft that continuously desquamates keratin debris. The term was coined by Johannes Müller in 1838 because the white-yellow keratin flakes resemble cholesterol crystals (though cholesterol is only a minor component). Cruveilhier first described it as a "pearly tumor" of the temporal bone.
Annual incidence: 3–15/100,000 in children; 9–13/100,000 in adults.
Classification
1. Congenital Cholesteatoma
- Arises from retained embryonic keratinizing epithelial rests within the middle ear — specifically from an epidermoid formation (Michaels' epidermoid) in the anterior tympanum that normally involutes by 33 weeks' gestation
- Appears as a white cystic structure medial to an intact tympanic membrane, without prior history of otitis media, perforation, or ear surgery
- More common in children; bilateral cases reported; possible genetic contribution
Potsic Staging System for Congenital Cholesteatoma:
| Stage | Description | Residual Risk |
|---|---|---|
| I | Limited to one quadrant | Low |
| II | Multiple quadrants, no ossicular involvement | Moderate |
| III | Ossicular involvement, no mastoid extension | Significant |
| IV | Mastoid involvement | 67% residual disease |
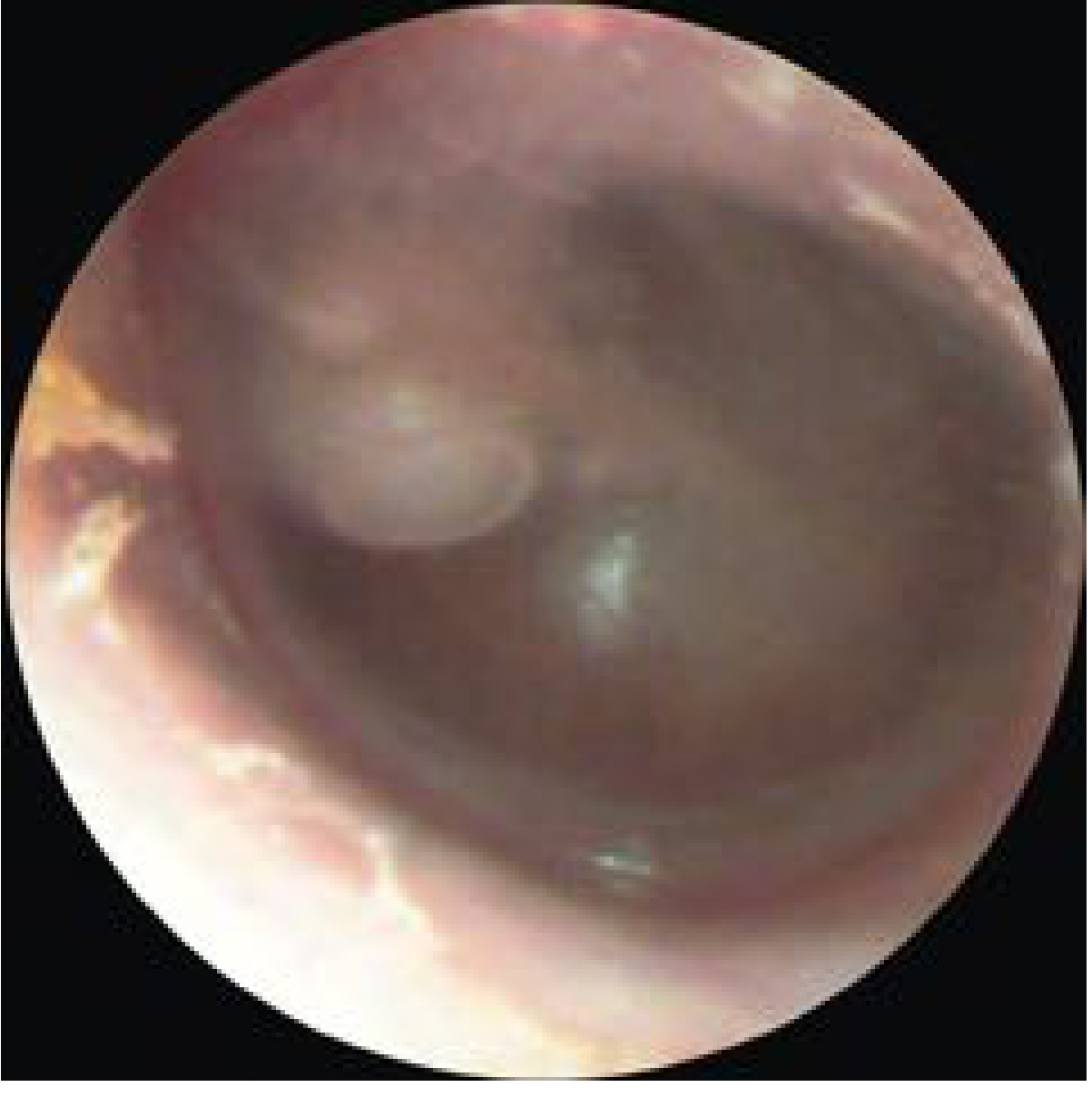
2. Acquired Cholesteatoma
A. Primary Acquired (Pars Flaccida / Attic Type)
- Arises from a retraction pocket of the pars flaccida due to chronic negative middle ear pressure (Eustachian tube dysfunction)
- Pocket deepens into the epitympanum → trapped keratin cannot migrate out → cholesteatoma forms
- Classic appearance: defect adjacent to the posterosuperior TM / attic with white keratin debris and scutum erosion
- Associated with cleft palate and underdeveloped mastoid pneumatization
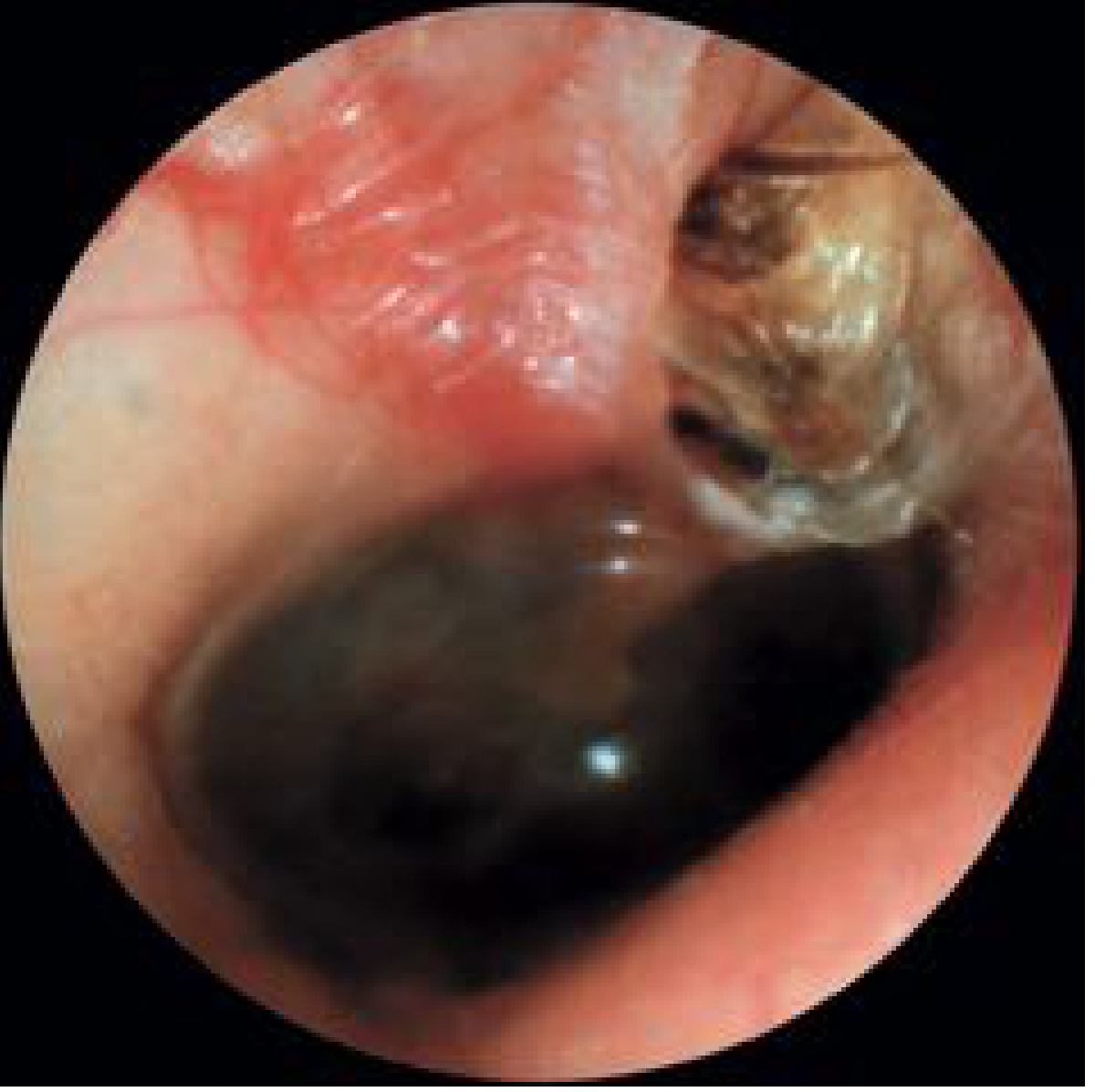
B. Secondary Acquired (Pars Tensa / Marginal Type)
- Keratinizing epithelium migrates through an existing perforation (usually marginal) into the middle ear
- More commonly associated with COM and longstanding TM perforations
- Often presents with a mass at the margin of a perforation
Pathogenesis Theories (Acquired)
Four classical theories, which are not mutually exclusive:
| Theory | Mechanism |
|---|---|
| Invagination (Retraction Pocket) | Pars flaccida retracts into attic under chronic negative ME pressure; trapped keratin → cholesteatoma. Most accepted mechanism for primary acquired type. |
| Basal Cell Hyperplasia | Basal cells of the TM proliferate and migrate into the middle ear cleft; supported by molecular markers (cytokeratin 13 & 16, EGF receptor overexpression) |
| Epithelial Ingrowth (Migration Theory) | Squamous epithelium migrates through a TM perforation; explains secondary acquired type |
| Squamous Metaplasia | Simple/cuboidal middle ear epithelium undergoes metaplastic transformation to keratinizing squamous epithelium (least supported; no human/animal model confirmation) |
| Mucosal Traction (Jackler) | Opposing mucosal surfaces propel pars flaccida into the attic via mucociliary flow and epithelial migration |
Clinical Features
Symptoms:
- Slowly progressive conductive hearing loss
- Chronic foul-smelling otorrhea (anaerobic infection of keratin debris)
- May be asymptomatic initially
- Vertigo, SNHL (labyrinthine fistula)
- Facial nerve palsy (erosion of bony fallopian canal — tympanic segment most vulnerable)
- Headache, meningismus (intracranial extension)
Otoscopic signs:
- Attic/posterosuperior defect with white keratin debris
- Aural polyp = granulation tissue at the eroding cholesteatoma margin — should be considered cholesteatoma until proven otherwise
- White mass behind intact TM (congenital)
Bone Erosion Mechanism
The classic "pressure necrosis" theory has been abandoned. Bone destruction is mediated by osteoclast activation via inflammatory cytokines. Key pathway:
- RANKL/RANK axis drives osteoclast recruitment
- Enzymes elaborated: acid phosphatase, collagenase, cathepsins, matrix metalloproteinases (MMPs)
- Intermittent osteoclastic resorption explains why some temporal bone specimens show no active erosion
Structures commonly eroded (in order of frequency):
- Scutum (lateral attic wall) — earliest sign
- Ossicular chain — incus long process most common; causes conductive HL
- Tegmen tympani (roof) → intracranial extension
- Lateral semicircular canal → labyrinthine fistula, vertigo
- Facial nerve canal (tympanic segment) → facial palsy
- Sigmoid sinus → lateral sinus thrombosis
Imaging
HRCT temporal bone:
- Soft tissue density in attic/mastoid with bony erosion
- Scutum erosion (classic sign)
- Ossicular chain destruction
- Labyrinthine involvement, tegmen dehiscence
- Essential for preoperative planning and all revision/complex cases
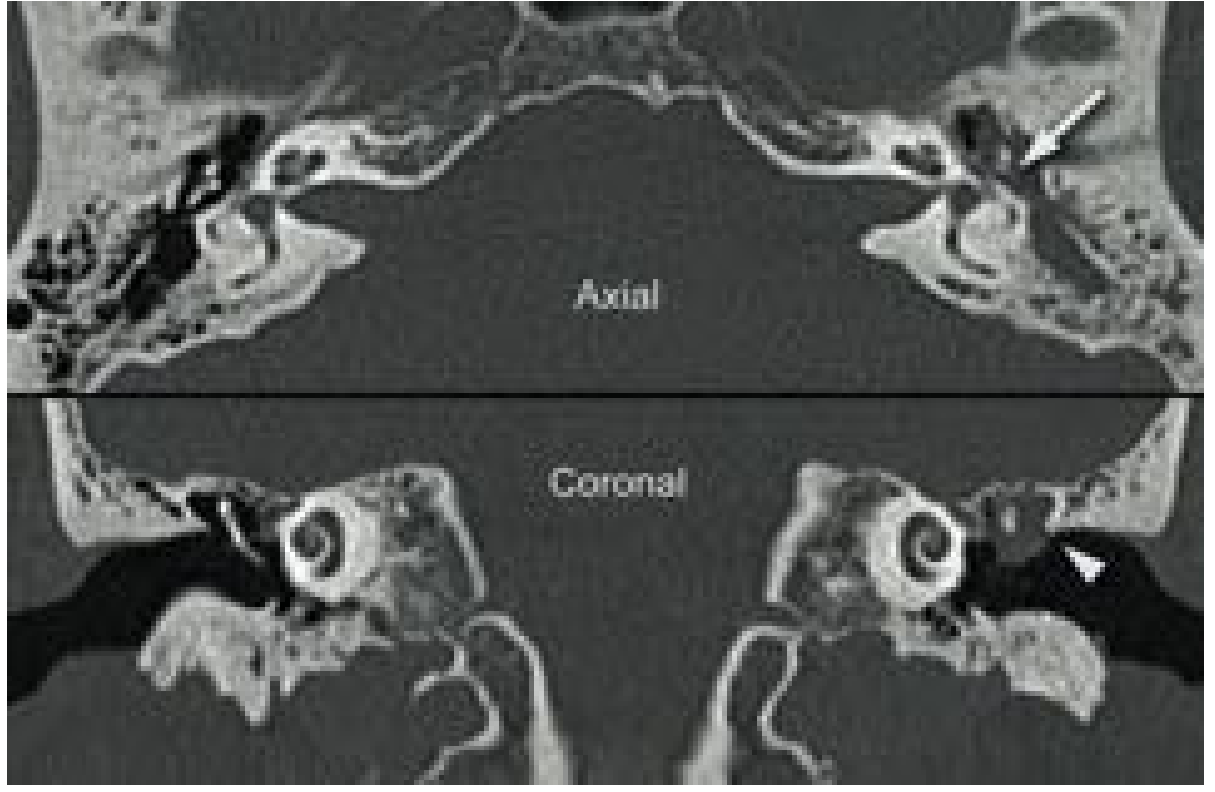
Non-echo-planar DWI MRI:
- Sensitivity and specificity ~94% for detecting cholesteatoma
- Bright signal on DWI + low ADC = true cholesteatoma
- Increasingly used for postoperative surveillance to detect residual/recurrent disease without second-look surgery
Complications
| Type | Example |
|---|---|
| Local | Ossicular erosion, TM perforation |
| Labyrinthine | Fistula (LSCC most common), SNHL, vertigo |
| Facial nerve | Paresis/paralysis (tympanic segment) |
| Intracranial | Meningitis, brain abscess, epidural abscess, lateral sinus thrombosis, otitic hydrocephalus |
| Petrous apex | Gradenigo syndrome (VI nerve palsy + retro-orbital pain + otorrhea) |
Management
Surgery is the only curative treatment — no medical therapy eliminates cholesteatoma.
Surgical Approach Options:
| Procedure | Description | Pros | Cons |
|---|---|---|---|
| Canal Wall-Up (CWU) / Closed | Posterior tympanotomy + mastoidectomy, EAC wall preserved | Physiological ear; no cavity; good hearing reconstruction | Residual 11–27%; recurrent 5–13%; often needs second-look |
| Canal Wall-Down (CWD) / Open | Posterior canal wall removed; mastoid exteriorized | Residual/recurrent only 2–10%; recurrence easily seen | Lifelong cavity cleaning; shallow ME; meatoplasty issues; temperature dizziness |
| Radical Mastoidectomy | All ME structures removed; obliteration of ME space | Complete exteriorization | Hearing sacrifice |
| Modified Radical (Bondy) | Disease limited to attic; ossicular chain preserved | Disease-free ear; some hearing preserved | Limited indications |
| Endoscopic Ear Surgery | Transcanal endoscopic approach | Improved visualization of hidden areas; less morbidity | Learning curve; one-handed technique |
Key factors guiding choice: extent of disease, complications present, mastoid pneumatization, ET function, hearing status (both ears), patient reliability, surgeon experience.
Follow-up after CWU: Second-look surgery at 12–18 months OR non-EPI DWI MRI surveillance.
Key Pearls
- Cholesteatoma in 36% of perforated ears vs. 4% of non-perforated ears in COM temporal bones
- An aural polyp = cholesteatoma until proven otherwise
- Poorer Eustachian tube function → greater risk of recurrence post-repair
- Underdeveloped mastoid pneumatization is near-universal in both acquired and congenital cholesteatoma
- In children, even stage IV congenital cholesteatoma carries 67% residual disease — second-look is essential
- Facial nerve palsy from cholesteatoma = surgical emergency
Source: Cummings Otolaryngology Head and Neck Surgery; The Developing Human (Moore & Persaud); K.J. Lee's Essential Otolaryngology
This is a shared conversation. Sign in to Orris to start your own chat.