Breast cancer in pregnancy
Reading File
Finding Sources
Searching PubMed
"breast cancer" AND "pregnancy" AND management
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive information to synthesize a complete, well-structured answer.
Breast Cancer in Pregnancy
Definition
Gestational (pregnancy-associated) breast cancer (PABC) is defined as breast cancer diagnosed:
- During pregnancy
- In the first postpartum year
- At any time during lactation
— Creasy & Resnik's Maternal-Fetal Medicine, p. 1356; Current Surgical Therapy 14e, p. 785
Epidemiology
- Breast cancer is the most common non-gynecologic malignancy associated with pregnancy
- ~1 in 1,000 pregnancies complicated by breast cancer in the US; ~10,000 cases/year worldwide
- Incidence: 17.5–39.9 per 100,000 births (postpartum rates higher than during pregnancy itself)
- 10–20% of breast cancers in women in their 30s are discovered during pregnancy
- Incidence is increasing due to delayed childbearing and younger age of breast cancer diagnoses
- Women with first pregnancy after age 35 have an increased risk of breast cancer; those before age 20 have a 50% reduced risk
— Current Surgical Therapy 14e, p. 785; Creasy & Resnik's, p. 1356
Pathology / Histology
- Invasive ductal carcinoma (NOS) accounts for 78–88% of cases
- Triple-negative phenotype is disproportionately common, accounting for ~50–60% of PABC
- Higher histologic grade, larger tumor size, more frequent lymph node involvement, and lower ER/PR expression compared to non-pregnant patients
- ER negativity is likely due to downregulation of estrogen receptors during the hyperestrogenic state of pregnancy
- Higher proportion of inflammatory breast cancer
— Current Surgical Therapy 14e, p. 785; Sabiston Textbook of Surgery, p. 486
Clinical Presentation & Diagnosis
Presentation
- Most common: painless palpable breast mass
- Milk rejection sign — infant refuses to nurse from one breast; may indicate an occult carcinoma
- Diagnostic delays of ≥2 months are common due to physiologic engorgement and increased nodularity
- A 1-month delay in treatment increases risk of axillary metastasis by 0.9–1.8%
- Any discrete lump present for 2–4 weeks should be biopsied
Differential Diagnosis of a Breast Mass in Pregnancy
Breast cancer, lactating adenoma, fibrocystic disease, milk retention cyst, abscess, lipoma, hamartoma, leukemia/lymphoma, phyllodes tumor, sarcoma
Imaging
| Modality | Status | Notes |
|---|---|---|
| Ultrasound | First-line | Safe, distinguishes solid vs. cystic in 97%; also for axillary staging |
| Mammography | Safe (with shielding) | Fetal dose only 0.0004 cGy; high false-negative rate in pregnancy |
| MRI (without gadolinium) | Safe for staging | Gadolinium crosses placenta — avoid; no role in diagnosis during pregnancy |
| CT chest/abdomen | Avoid if possible | Fetal exposure ~0.25 cGy; MRI preferred |
| Low-dose bone scan | Acceptable if indicated | Fetal dose ~0.08 cGy vs. standard 0.19 cGy |
— Creasy & Resnik's, p. 1357
Biopsy
- Core-needle biopsy is preferred — safe, reliable, performed under local anesthesia
- Fine-needle aspiration is an alternative but requires experienced pathologist (pregnancy-related proliferative changes can cause false indeterminate results)
- Risk of milk fistula ~1.4%; reduced by emptying breast before procedure; breastfeeding cessation is not mandatory if fistula develops
- Always send for ER, PR, HER2 immunohistochemistry
Staging
- AJCC TNM staging is used, same as non-pregnant patients
- PABC typically presents at a later stage than non-pregnant women (attributed to diagnostic delay, not the biology of pregnancy per se)
- Axillary staging:
- Palpable nodes → ultrasound-guided FNA
- Sentinel lymph node biopsy (SLNB) with technetium-99m sulfur colloid is safe in all trimesters (fetal dose <20 μGy)
- Blue dyes (isosulfan blue, methylene blue) are contraindicated — risk of anaphylaxis and fetal methemoglobinemia
Management
Multidisciplinary Team
Breast surgeon, medical oncologist, radiation oncologist, maternal-fetal medicine specialist, anesthesiologist, obstetrician, neonatologist, and genetic counselor.
Termination of pregnancy does not improve maternal prognosis. Treatment should adhere as closely as possible to non-pregnant standards.
Genetic testing (BRCA) is indicated; probability of pathogenic BRCA variant in young triple-negative patient is ~20%.
Surgery
Surgery is safe in all three trimesters. Safest period: second trimester (weeks 12–24).
Key intraoperative considerations:
- Avoid maternal hypoxia and hypotension; maintain MAP ≥60 mmHg for uterine perfusion
- After 20 weeks: left lateral tilt to displace uterus off IVC
- After 24 weeks: intraoperative fetal monitoring required
Mastectomy vs. Breast-Conserving Surgery (BCS):
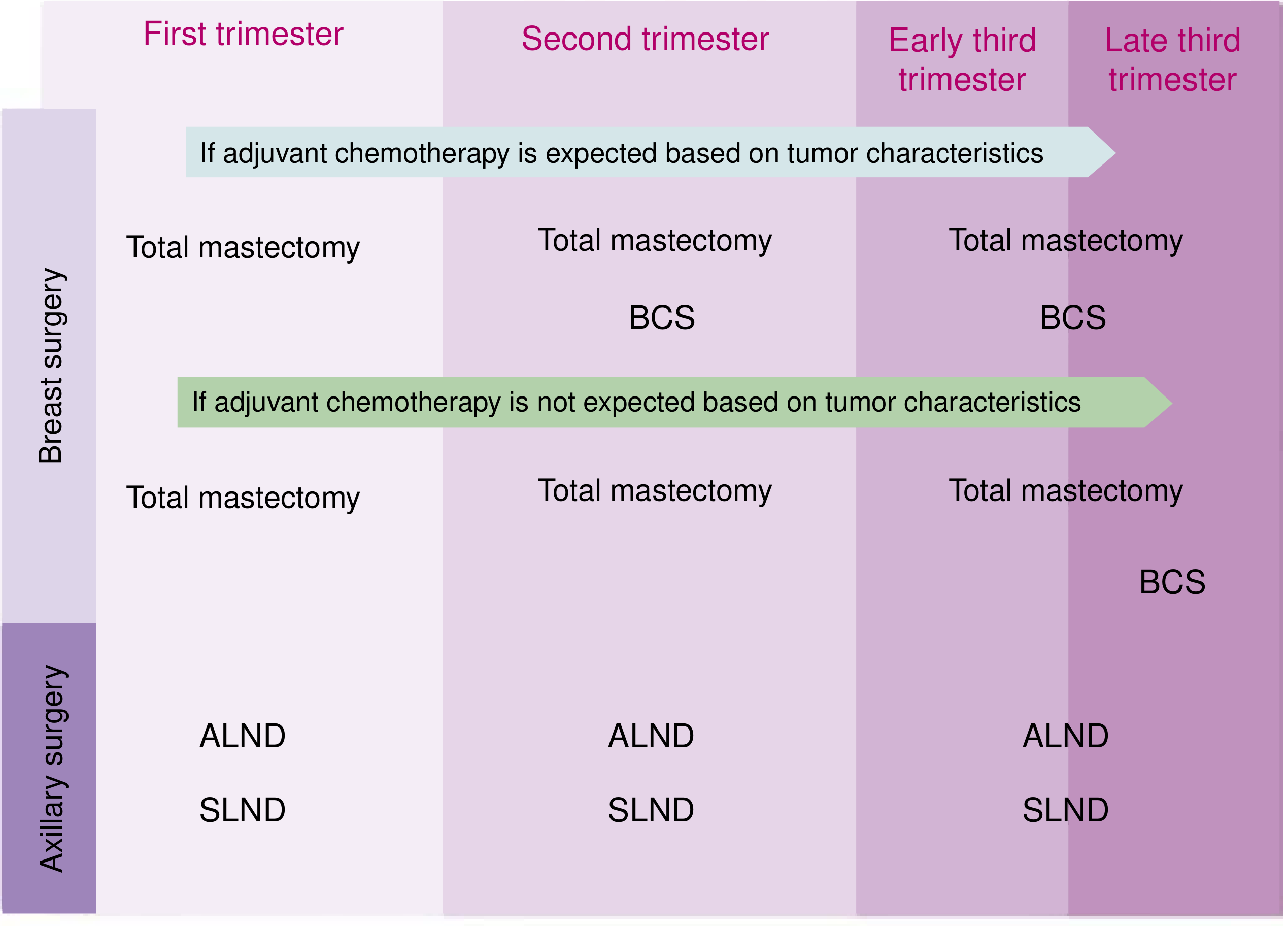
Surgical options for breast and axillary surgery by trimester. BCS = breast-conserving surgery; ALND = axillary lymph node dissection; SLND = sentinel lymph node dissection. — Current Surgical Therapy 14e, p. 787
- First trimester: Mastectomy is standard (BCS is not appropriate because radiation cannot follow immediately)
- Second trimester: Mastectomy or BCS; BCS must be followed by adjuvant chemo during remainder of pregnancy, then radiation postpartum
- Third trimester: Both mastectomy and BCS acceptable; postpartum radiation follows
- Immediate tissue expander reconstruction is safe and feasible in all trimesters
Chemotherapy
| Aspect | Recommendation |
|---|---|
| Timing | Safe from week 14 onward (2nd and 3rd trimester) |
| First trimester | Contraindicated — teratogenesis risk during organogenesis (weeks 4–12) |
| Preferred regimens | Anthracycline-based: doxorubicin + cyclophosphamide ± taxanes (paclitaxel/docetaxel) |
| Stop before delivery | No chemo after 35 weeks or within 3 weeks of planned delivery (myelosuppression risk) |
| Dosing | Same as non-pregnant patients; based on body surface area, adjusted for gestational weight gain |
| Supportive agents | Metoclopramide, ondansetron, granisetron, prochlorperazine (safe); methylprednisolone/hydrocortisone (preferred steroids); G-CSF and erythropoietin safe in 2nd/3rd trimester |
The placenta should be sent for pathologic evaluation at delivery (placental metastasis has been reported, but fetal metastasis has not).
Contraindicated Treatments During Pregnancy
| Treatment | Reason |
|---|---|
| Radiation therapy | Contraindicated at any point (fetal harm); defer to postpartum |
| Trastuzumab (HER2-targeted) | Oligohydramnios → pulmonary and renal insufficiency |
| Tamoxifen | Teratogenic — craniofacial malformations, ambiguous genitalia, fetal death |
| Methotrexate | Teratogenic |
| Bisphosphonates | Fetal underdevelopment, embryolethality, hypocalcemia, skeletal retardation |
| Isosulfan/methylene blue dye | Anaphylaxis risk; methemoglobinemia in fetus |
— Current Surgical Therapy 14e, p. 788
Endocrine Therapy
- Hormone receptor-positive tumors: tamoxifen deferred until after delivery
- Oncotype Dx 21-gene assay may still provide useful prognostic information in selected patients
Delivery & Monitoring
- Delivery planned 3–4 weeks after last chemotherapy cycle
- Timing based on maternal condition, treatment needs, and neonatal outcome expectations
- Women with PABC have a significantly higher rate of preterm delivery (61.2% vs. 8% in general population)
- Breastfeeding is contraindicated during chemotherapy or radiotherapy; lactation from a treated breast is not otherwise contraindicated
Prognosis
- When matched for stage and tumor characteristics, survival outcomes are similar to non-pregnant women
- The apparently worse prognosis historically reported reflects later stage at diagnosis, not pregnancy itself
- A 2.8-fold increase in metastasis and 2.7-fold increase in mortality has been observed in patients ≤40 years diagnosed within 5 years postpartum
— Current Surgical Therapy 14e, p. 785; Sabiston Textbook of Surgery, p. 486
Pregnancy After Breast Cancer
- Three large registry studies show no poorer prognosis in women who become pregnant after successful treatment
- Oncologists generally recommend delaying pregnancy 2–3 years after initial treatment (most recurrences occur within the first 2 years)
- Some reports even suggest an antitumor effect of subsequent pregnancy
— Creasy & Resnik's, p. 1359
Key Takeaways
- Any breast lump in pregnancy must be evaluated — ultrasound first, then core biopsy if solid
- Termination does not improve prognosis; standard treatment should proceed
- Surgery is safe in all trimesters; safest in the 2nd trimester
- Chemotherapy safe after week 14; stop ≥3 weeks before delivery
- Radiation, trastuzumab, tamoxifen, and methotrexate are contraindicated during pregnancy
- Blue dye for SLNB is contraindicated; use technetium-99m only
- Multidisciplinary management is essential
- Congenital abnormality rate in chemo-exposed infants (~3%) is comparable to the general population
chemotherapy in breast cancer in pregnancy
Finding Sources
Searching PubMed
"breast cancer" AND "pregnancy" AND chemotherapy
Finding Sources
Reading Article
Reading File
Reading File
Reading File
Reading File
I have comprehensive data from multiple textbooks plus recent systematic review evidence. Here is the full synthesis:
Chemotherapy in Breast Cancer in Pregnancy
General Principles
The decision to administer chemotherapy follows the same indications as in non-pregnant patients (node-positive disease, tumors >1–2 cm that are poorly differentiated), with adjustments for gestational age and overall treatment sequencing. Most pregnancy-associated breast cancers meet criteria for chemotherapy, as they tend to present with larger tumors and more frequent nodal involvement.
All chemotherapeutic agents used in breast cancer are classified by the FDA as Category D or X (teratogenic effects documented in humans). Decision-making must balance fetal risk against the consequences of withholding or delaying therapy for the mother.
— Creasy & Resnik's Maternal-Fetal Medicine, p. 1358; Current Surgical Therapy 14e, p. 786
Timing by Trimester
First Trimester (Weeks 1–12) — CONTRAINDICATED
Chemotherapy is contraindicated during weeks 4–12 (organogenesis). Risks include:
- Spontaneous abortion
- Congenital malformations
- Fetal death
The incidence of congenital malformation with first-trimester chemotherapy is approximately 16%, compared to only 1.3% when given in the second or third trimester.
Chemotherapy should be deferred during the first trimester as long as the mother's health is not compromised by the delay.
Second & Third Trimester (from Week 14 onward) — SAFE
Chemotherapy can be started as early as week 14. The risk of fetal malformation is not increased beyond baseline in the second and third trimesters.
However, approximately 50% of infants exposed to chemotherapy in the 2nd or 3rd trimester for breast cancer manifest fetal growth restriction, prematurity, and low birth weight.
Stop Before Delivery
Chemotherapy must be stopped at 35 weeks' gestation or at least 3 weeks before planned delivery. This interval is required to:
- Allow recovery from myelosuppression (neonatal pancytopenia, neutropenia, risk of sepsis at delivery)
- Prevent drug accumulation in the fetus
- Reduce risk of bleeding or infection in mother at delivery
The placenta should be sent for pathologic evaluation — placental metastasis has been reported, though fetal metastasis has not.
— Current Surgical Therapy 14e, p. 787; Creasy & Resnik's, p. 1358–1359
Pharmacokinetics in Pregnancy
Gestational physiologic changes alter drug handling and require attention to dosing:
| Change | Effect on Chemotherapy |
|---|---|
| Increased plasma volume | ↓ Peak plasma concentration, ↓ drug exposure |
| Increased glomerular filtration rate | ↑ Drug clearance |
| Decreased serum albumin | Altered free drug fraction |
| Decreased gastric emptying | ↑ Oral absorption |
| Altered hepatic function (hormonal) | Variable metabolism |
Net result: for doxorubicin, epirubicin, paclitaxel, and carboplatin there is decreased plasma drug exposure and increased distribution volume. Despite this pharmacokinetic concern, outcomes remain similar to non-pregnant patients when controlling for stage and tumor characteristics.
Dosing: Same as non-pregnant patients — based on body surface area (current weight, adjusted throughout pregnancy) and creatinine clearance.
— Current Surgical Therapy 14e, p. 787
Recommended Regimens
Safe Agents (2nd and 3rd Trimester)
| Drug Class | Agents | Safety Status |
|---|---|---|
| Anthracyclines | Doxorubicin, Epirubicin | Safe; most data available |
| Alkylating agents | Cyclophosphamide | Safe; long safety record |
| Fluoropyrimidines | 5-Fluorouracil | Safe |
| Taxanes | Paclitaxel (weekly or q3w), Docetaxel (q3w) | Safe after 1st trimester |
Standard Regimens Used
- AC — Doxorubicin + Cyclophosphamide
- FAC/FEC — 5-Fluorouracil + Doxorubicin/Epirubicin + Cyclophosphamide (the only prospective study of chemo in PABC used FAC in 24 patients — no birth defects, median delivery at 38 weeks)
- AC + Taxane — Doxorubicin + Cyclophosphamide followed by Paclitaxel or Docetaxel
Some investigators advocate weekly epirubicin (shorter nadir periods reduce risk if unexpected delivery occurs), but this is not standard — maternal prognosis should not be compromised to limit unproven fetal damage.
Dose-Dense Regimens
Data in pregnancy are scarce. Small studies show no increased maternal or fetal complications, but routine use is not established.
— Current Surgical Therapy 14e, p. 786; Creasy & Resnik's, p. 1359; Sabiston Textbook of Surgery, p. 486
Agents Contraindicated During Pregnancy
| Agent | Reason |
|---|---|
| Methotrexate | Teratogenic; antimetabolite — high risk of spontaneous abortion even after 1st trimester; accumulates in amniotic fluid ("3rd spacing") |
| Trastuzumab (HER2-targeted) | Oligohydramnios → fetal pulmonary hypoplasia and renal insufficiency; 4 neonatal deaths from respiratory/renal failure reported; fetal renal epithelium strongly expresses HER2 |
| Tamoxifen | Craniofacial malformations, ambiguous genitalia, fetal death; deferred to postpartum |
| Other anti-HER2 agents / immunotherapy | Multiple fetal complications in case reports; considered contraindicated |
| Bisphosphonates | Fetal underdevelopment, embryolethality, hypocalcemia, skeletal retardation |
| Radiation therapy | Contraindicated at any stage of pregnancy; deferred postpartum |
| Blue dye (isosulfan blue, methylene blue) | Not chemotherapy, but relevant to management — methemoglobinemia risk; contraindicated |
For HER2-positive disease: chemotherapy (anthracycline/taxane backbone) proceeds during pregnancy; trastuzumab is deferred until after delivery.
— Current Surgical Therapy 14e, p. 788; Creasy & Resnik's, p. 1358
Neoadjuvant vs. Adjuvant Setting
- Chemotherapy may be given in either adjuvant or neoadjuvant settings during pregnancy
- If neoadjuvant: start at week 14, continue standard cycles as tolerated; surgical decision (mastectomy vs. BCS) follows postpartum if needed
- Neoadjuvant approach is particularly useful for locally advanced disease — allows definitive surgery to be deferred while systemic therapy proceeds during pregnancy
Supportive Care During Chemotherapy
| Drug | Safety |
|---|---|
| Antiemetics | Metoclopramide, ondansetron, granisetron, prochlorperazine — all safe |
| Steroids (pre-chemo) | Methylprednisolone, hydrocortisone — preferred (extensively metabolized in placenta); dexamethasone also used |
| G-CSF (e.g., filgrastim) | Safe in 2nd and 3rd trimester |
| Erythropoietin | Safe in 2nd and 3rd trimester |
Fetal & Neonatal Outcomes
- Congenital malformation rate ~3% — comparable to the national average for the general population
- ~50% of infants exposed to chemo in 2nd/3rd trimester show fetal growth restriction, prematurity, or low birth weight — normalizes over time
- No long-term cognitive or developmental deficits have been demonstrated
- Preterm delivery rate is significantly elevated (61.2% vs. 8% in the general population)
- Taxane systematic review (PMID 38166767, 2024): Among 159 patients (55% breast cancer, mostly paclitaxel), obstetric and fetal outcomes were generally similar to the general obstetric population when taxanes were used after the first trimester — live birth in all cases with known outcome; preterm delivery in 54.5%; SGA in 30.3%; 7 congenital malformations (5.3%)
Breastfeeding
Breastfeeding is contraindicated during chemotherapy — doxorubicin and cyclophosphamide both enter breast milk. Breastfeeding may resume after treatment is complete.
Key Points Summary
| Point | Detail |
|---|---|
| Contraindicated in | 1st trimester (organogenesis, weeks 4–12) |
| Safe from | Week 14 onward (2nd and 3rd trimester) |
| Stop before delivery | ≥3 weeks before delivery; no chemo after 35 weeks |
| Preferred backbone | Anthracycline + cyclophosphamide ± taxane (FAC/AC/AC-T) |
| Dosing basis | Body surface area (current weight), adjusted serially |
| Absolutely contraindicated | Methotrexate, trastuzumab, tamoxifen, radiation |
| Fetal malformation risk (2nd/3rd trimester) | ~1.3% (vs. 16% if 1st trimester) |
| Malformation rate vs. population | ~3% — not significantly elevated |
— Current Surgical Therapy 14e, p. 786–788; Creasy & Resnik's Maternal-Fetal Medicine, p. 1357–1359; Sabiston Textbook of Surgery, p. 486
thermal mammogram
Reading File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.