Explain in detail biosynthesis and catabolism of catecholamine
Reading File
Finding Sources
Finding Sources
Searching Images
catecholamine biosynthesis pathway dopamine norepinephrine epinephrine
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
Searching Images
catecholamine degradation MAO COMT VMA vanillylmandelic acid pathway
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have all the information needed for a comprehensive, detailed answer. Let me compile it.
Biosynthesis and Catabolism of Catecholamines
Overview
The catecholamines — dopamine, norepinephrine (NE), and epinephrine (Epi) — share a common structural motif: a catechol ring (ortho-dihydroxybenzene) plus an ethylamine side chain. They are synthesized from the amino acid tyrosine through a sequential enzymatic cascade, and degraded primarily by two enzymes — monoamine oxidase (MAO) and catechol-O-methyltransferase (COMT).
Part I: Biosynthesis
Biosynthetic pathway diagram:
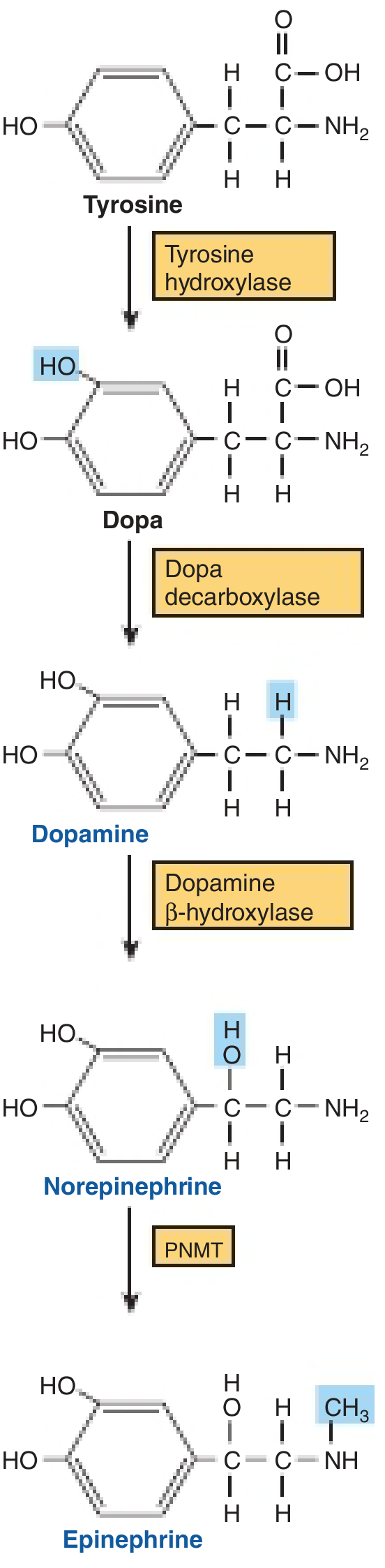
The conversion of tyrosine to epinephrine requires four sequential enzymatic steps.
Step 1 — Tyrosine → L-DOPA
Enzyme: Tyrosine hydroxylase (TH)
Location: Cytosol of catecholamine-producing neurons and adrenal chromaffin cells
Cofactor: Tetrahydrobiopterin (BH₄), molecular O₂, Fe²⁺
Reaction: Hydroxylation of the para-position of the phenyl ring of tyrosine → 3,4-dihydroxyphenylalanine (L-DOPA)
This is the rate-limiting step. TH is subject to feedback inhibition by dopamine and norepinephrine, which compete with the BH₄ cofactor — providing tight internal regulation of catecholamine output. Tyrosine is transported into neurons via a Na⁺-dependent carrier. Some tyrosine is derived from dietary phenylalanine via phenylalanine hydroxylase in the liver, but most is of dietary origin directly.
Clinically: α-methyl-p-tyrosine (metyrosine) competitively inhibits TH and is used to reduce catecholamine synthesis in patients with pheochromocytoma. Catecholamines cannot cross the blood–brain barrier, so they must be synthesized locally in the CNS. L-DOPA does cross the blood–brain barrier, which is the basis for its use in Parkinson disease. — Harper's Illustrated Biochemistry, 32nd Ed; Goldman-Cecil Medicine
Step 2 — L-DOPA → Dopamine
Enzyme: Aromatic L-amino acid decarboxylase (AADC), also called dopa decarboxylase
Location: Cytosol; present in virtually all tissues
Cofactor: Pyridoxal phosphate (vitamin B₆)
Reaction: Decarboxylation of L-DOPA → 3,4-dihydroxyphenylethylamine (dopamine)
This enzyme is widely expressed and not tissue-specific. Dopamine neurons express TH + AADC but lack DBH (the next enzyme), so dopamine is their final product. Competitive inhibitors (e.g., α-methyldopa) are effective antihypertensives. Carbidopa, a peripheral AADC inhibitor that does not cross the BBB, is co-administered with L-DOPA to reduce peripheral side effects. — Harper's Illustrated Biochemistry; Kaplan & Sadock's Comprehensive Textbook of Psychiatry
Step 3 — Dopamine → Norepinephrine
Enzyme: Dopamine β-hydroxylase (DBH)
Location: Inside secretory/storage vesicles (granule-bound enzyme) — the only transmitter synthesized inside a vesicle
Cofactors: Ascorbate (electron donor), Cu²⁺ at the active site, fumarate as modulator
Reaction: β-hydroxylation of dopamine's side chain → norepinephrine
After synthesis in the cytoplasm, dopamine is actively transported into the vesicle by the vesicular monoamine transporter (VMAT), driven by a H⁺ gradient. DBH then converts it to NE inside the vesicle. DBH is a monooxygenase. Reserpine blocks VMAT and depletes catecholamine stores. — Ganong's Review of Medical Physiology, 26th Ed; Harper's Illustrated Biochemistry
Step 4 — Norepinephrine → Epinephrine
Enzyme: Phenylethanolamine N-methyltransferase (PNMT)
Location: Cytoplasm of adrenal medullary chromaffin cells and organ of Zuckerkandl; absent from most peripheral sympathetic neurons
Methyl donor: S-adenosylmethionine (SAM)
Reaction: N-methylation of norepinephrine → epinephrine
Because PNMT is a cytoplasmic enzyme, NE must exit the storage vesicle into the cytoplasm for methylation, then re-enter a separate vesicle. PNMT expression is induced by glucocorticoids delivered via the unique intra-adrenal portal system, which creates a ~100-fold steroid concentration gradient over systemic levels. This is why adrenal medullary tissue produces ~80% epinephrine, while extra-adrenal paragangliomas, which lack this high cortisol exposure, synthesize primarily norepinephrine. — Goldman-Cecil Medicine; Sabiston Textbook of Surgery
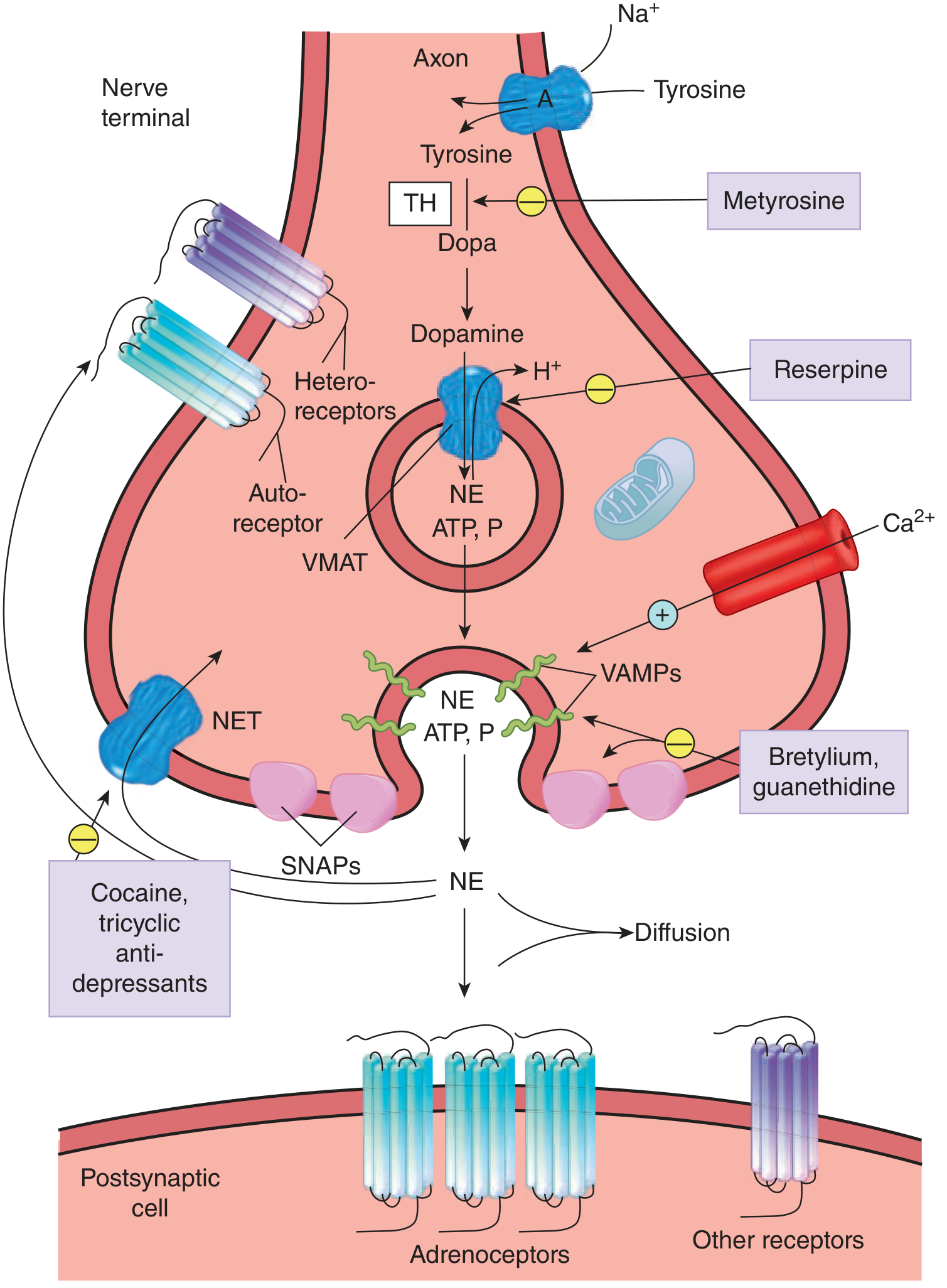
Noradrenergic synapse showing biosynthetic and release machinery:
Summary Table: Biosynthetic Enzymes
| Step | Substrate | Product | Enzyme | Location | Cofactor |
|---|---|---|---|---|---|
| 1 (rate-limiting) | Tyrosine | L-DOPA | Tyrosine hydroxylase | Cytosol | BH₄, O₂, Fe²⁺ |
| 2 | L-DOPA | Dopamine | Dopa decarboxylase (AADC) | Cytosol | Pyridoxal-P (B₆) |
| 3 | Dopamine | Norepinephrine | Dopamine β-hydroxylase | Vesicle lumen | Ascorbate, Cu²⁺ |
| 4 | Norepinephrine | Epinephrine | PNMT | Cytosol (adrenal only) | SAM |
Part II: Storage and Release
Catecholamines are stored in chromaffin granules (adrenal medulla) or synaptic vesicles (neurons), co-packaged with ATP, chromogranins, and neuropeptides. Release occurs by Ca²⁺-dependent exocytosis triggered by action potentials. The process involves SNAPs and VAMPs (vesicle-associated membrane proteins). Sympathetic stimulation can increase adrenal catecholamine secretion up to 50-fold above basal levels. — Sabiston Textbook of Surgery
Part III: Catabolism
Catecholamines are potent but short-lived: plasma half-life is 10–100 seconds, with NE/Epi having a half-life on the order of ~1 minute. Inactivation occurs through three mechanisms:
- Reuptake (dominant mechanism)
- Enzymatic degradation (MAO and COMT)
- Diffusion away from the synapse
3.1 Reuptake
Almost 90% of NE released at sympathetic synapses is removed by Uptake 1 (neuronal reuptake) via the norepinephrine transporter (NET). This is blocked by cocaine, tricyclic antidepressants, and phenothiazines. Extraneuronal tissues also take up catecholamines via Uptake 2, where most are metabolized by COMT. — Goldman-Cecil Medicine
3.2 Enzymatic Degradation: The Two Key Enzymes
Monoamine Oxidase (MAO)
- Location: Outer mitochondrial membrane; present in nerve terminals, liver, gut wall, and many other tissues
- Reaction: Oxidative deamination of the amine side chain → reactive aldehyde intermediate
- Subtypes: MAO-A (preferentially deaminates NE, Epi, serotonin); MAO-B (preferentially deaminates dopamine, phenylethylamine)
- The aldehyde intermediate from dopamine is further oxidized by aldehyde dehydrogenase → DOPAC (3,4-dihydroxyphenylacetic acid)
- The aldehyde intermediate from NE and Epi (β-hydroxylated) is preferentially reduced by aldehyde reductase → DHPG (3,4-dihydroxyphenylglycol)
- MAO inhibitors (MAOIs) (e.g., phenelzine, selegiline) are used as antidepressants and in Parkinson disease
Catechol-O-Methyltransferase (COMT)
- Location: Cytoplasm and membranes of most extraneuronal tissues — liver, kidney, smooth muscle, glial cells; absent from presynaptic noradrenergic neurons
- Reaction: O-methylation (meta-position) using SAM as methyl donor
- Converts: NE → normetanephrine; Epi → metanephrine; dopamine → methoxytyramine
- COMT inhibitors (e.g., tolcapone, entacapone) are used adjunctively in Parkinson disease
3.3 Detailed Catabolic Pathways
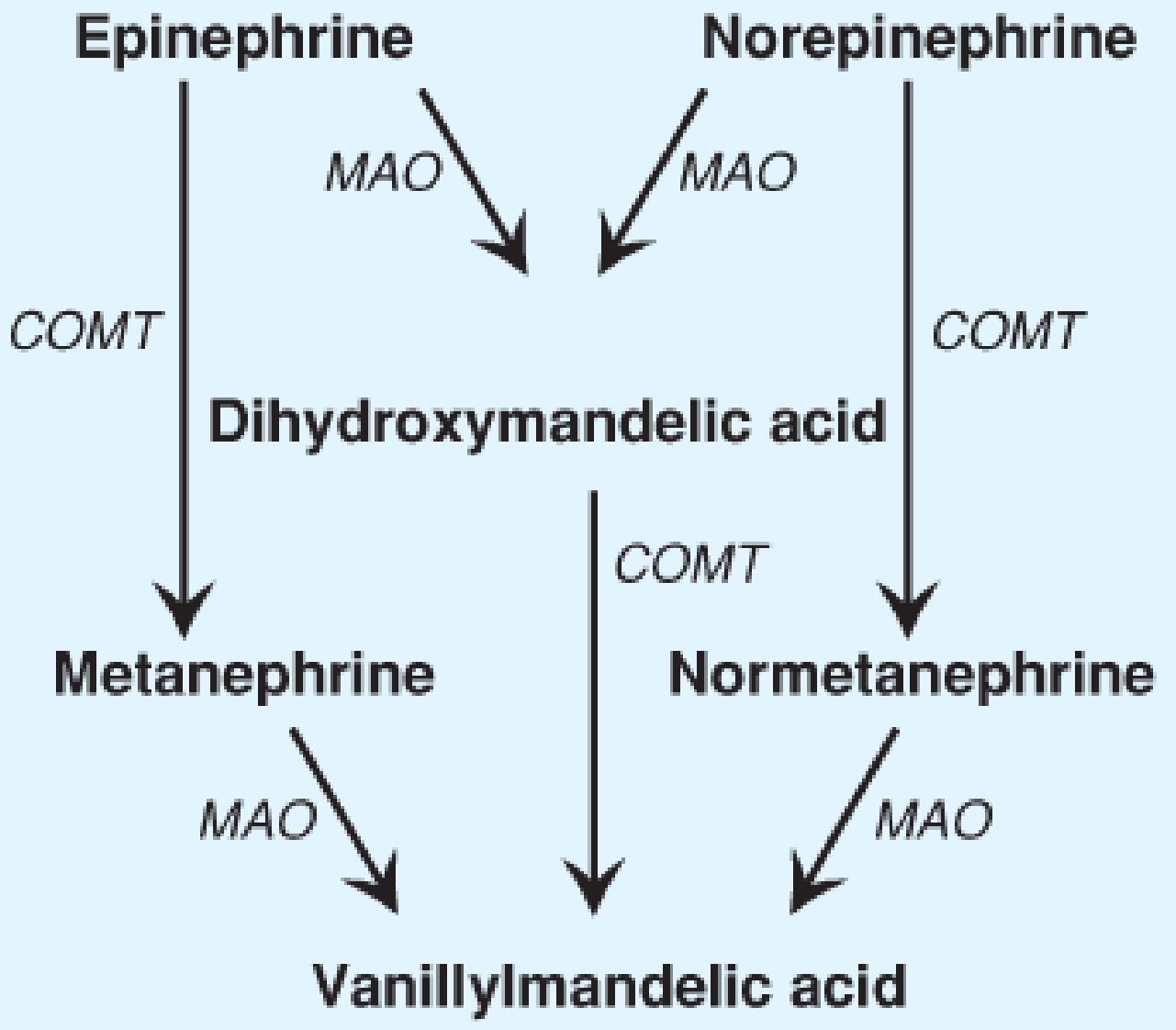
Simplified catabolism diagram (Lippincott):
Comprehensive catabolism pathway diagram (Tietz):
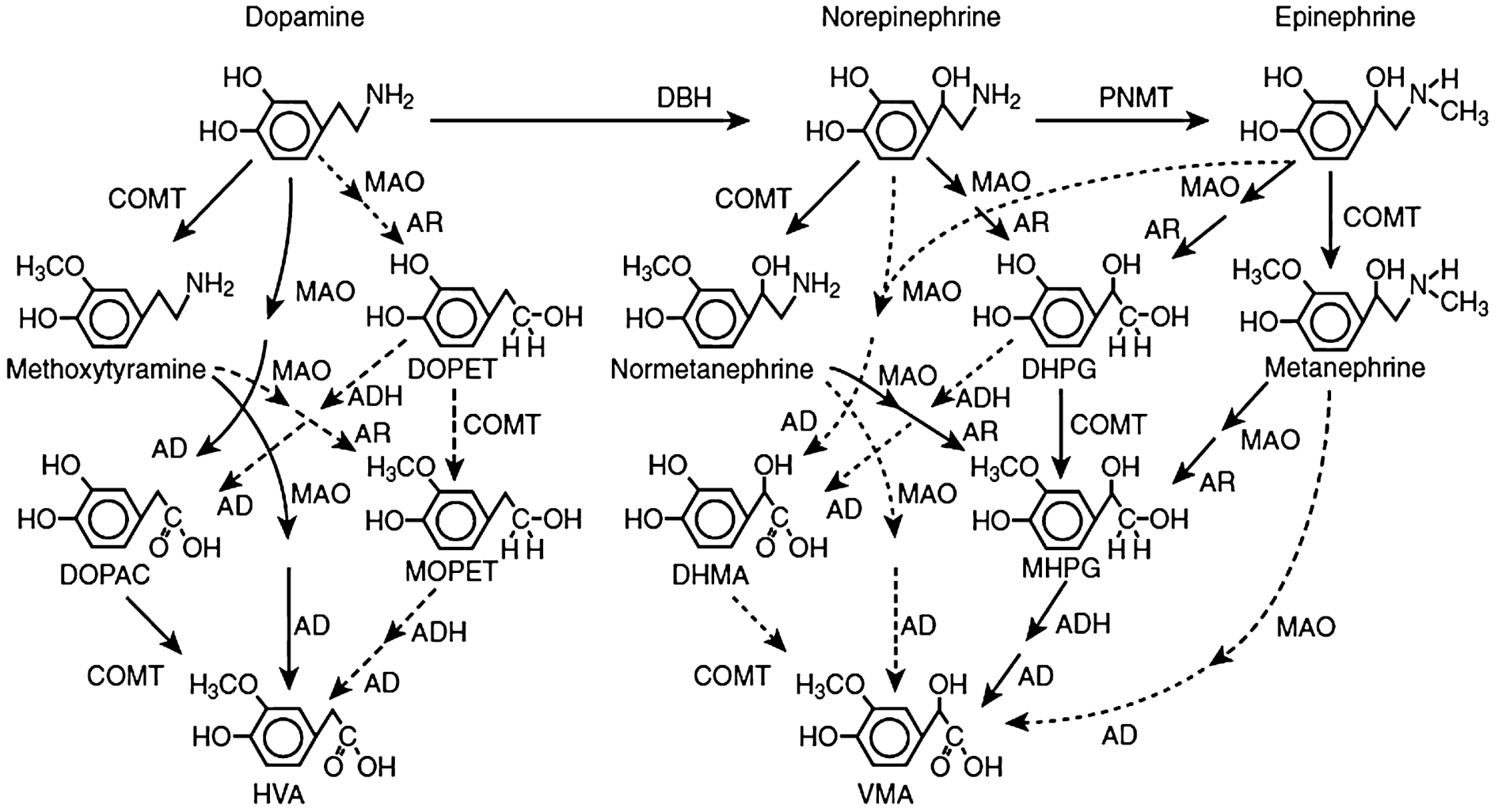
Pathway A — NE/Epi: COMT First, Then MAO
Most important for extraneuronal (circulating) catecholamines
- COMT acts: NE → normetanephrine; Epi → metanephrine (collectively called metanephrines)
- MAO acts on these O-methylated intermediates → reactive aldehydes → converted by aldehyde dehydrogenase → vanillylmandelic acid (VMA)
Pathway B — NE/Epi: MAO First, Then COMT
Most important for neuronal (intraneuronal) catecholamines leaking from storage vesicles
- MAO acts intraneuronally on NE/Epi → aldehyde intermediate → reduced by aldehyde reductase → DHPG (3,4-dihydroxyphenylglycol)
- COMT then O-methylates DHPG → MHPG (3-methoxy-4-hydroxyphenylglycol)
- MHPG can be further oxidized by aldehyde dehydrogenase → VMA
VMA (vanillylmandelic acid) is the major final urinary metabolite of NE and Epi. — Ganong's; Goldman-Cecil
Dopamine Catabolism
Dopamine follows a parallel pathway:
- MAO → DOPAC (3,4-dihydroxyphenylacetic acid) — via aldehyde dehydrogenase
- COMT acts on dopamine → methoxytyramine
- Sequential action of both enzymes ultimately yields homovanillic acid (HVA) as the major terminal metabolite of dopamine
3.4 Final Excretion
- Major urinary metabolites: VMA (NE, Epi), HVA (dopamine), MHPG (neuronal NE)
- Metanephrines (metanephrine and normetanephrine) are also measurable in plasma and urine
- Further conjugation with sulfate (SULT1A3) or glucuronic acid in the liver before renal excretion
- Because metanephrines have relatively stable plasma levels (unlike the pulsatile NE/Epi), plasma free metanephrines are the preferred screening test for pheochromocytoma — Sabiston Textbook of Surgery
Summary of Major Metabolites
| Catecholamine | Neuronal Pathway (MAO first) | Extraneuronal Pathway (COMT first) | Final Major Metabolite |
|---|---|---|---|
| Norepinephrine | NE → DHPG → MHPG → VMA | NE → Normetanephrine → VMA | VMA |
| Epinephrine | Epi → DHPG → MHPG → VMA | Epi → Metanephrine → VMA | VMA |
| Dopamine | DA → DOPAC → HVA | DA → Methoxytyramine → HVA | HVA |
Part IV: Pharmacological and Clinical Correlations
| Drug/Condition | Mechanism | Effect |
|---|---|---|
| Metyrosine | TH inhibitor | Reduces catecholamine synthesis (used in pheo) |
| Carbidopa | AADC inhibitor (peripheral) | Prevents peripheral L-DOPA conversion |
| Reserpine | VMAT blocker | Depletes catecholamine stores → antihypertensive |
| Cocaine, TCAs | NET/DAT blocker | Prolongs catecholamine action |
| MAOIs | MAO inhibition | ↑ NE, DA, 5-HT — antidepressant |
| Entacapone/Tolcapone | COMT inhibitor | Prolongs L-DOPA action in Parkinson |
| Pheochromocytoma | Excess NE/Epi secretion | Diagnosed by plasma/urine metanephrines and VMA |
| Parkinson disease | Dopamine deficiency (substantia nigra) | Treated with L-DOPA + carbidopa |
Sources: Harper's Illustrated Biochemistry, 32nd Ed | Ganong's Review of Medical Physiology, 26th Ed | Goldman-Cecil Medicine | Sabiston Textbook of Surgery | Kaplan & Sadock's Comprehensive Textbook of Psychiatry | Tietz Textbook of Laboratory Medicine, 7th Ed | Lippincott's Illustrated Reviews: Biochemistry, 8th Ed
This is a shared conversation. Sign in to Orris to start your own chat.