Hep B
Reading File
Asking for Preferences
Finding Sources
Searching the Web
hepatitis B pathophysiology overview 2025
Reading File
Finding Sources
Searching Images
hepatitis B virus structure replication lifecycle
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of Hepatitis B virus (HBV) — virology, replication, and pathogenesis.
Hepatitis B: Overview & Pathophysiology
1. The Virus
HBV is a hepadnavirus — a partially double-stranded DNA virus with a unique replication strategy that involves reverse transcription, shared with retroviruses. It is the smallest known human DNA virus.
Three circulating particles appear in infected serum:
| Particle | Size | Description |
|---|---|---|
| Intact virion (Dane particle) | 42 nm | Complete, double-shelled; infectious |
| Spherical subviral particles | 22 nm | Excess HBsAg; non-infectious |
| Tubular filaments | 22 nm diameter | Also HBsAg only; non-infectious |
The small spheres and tubules outnumber complete virions by 100–1000:1. Blood concentrations can reach 500 µg/mL of HBsAg and 10 trillion particles/mL.
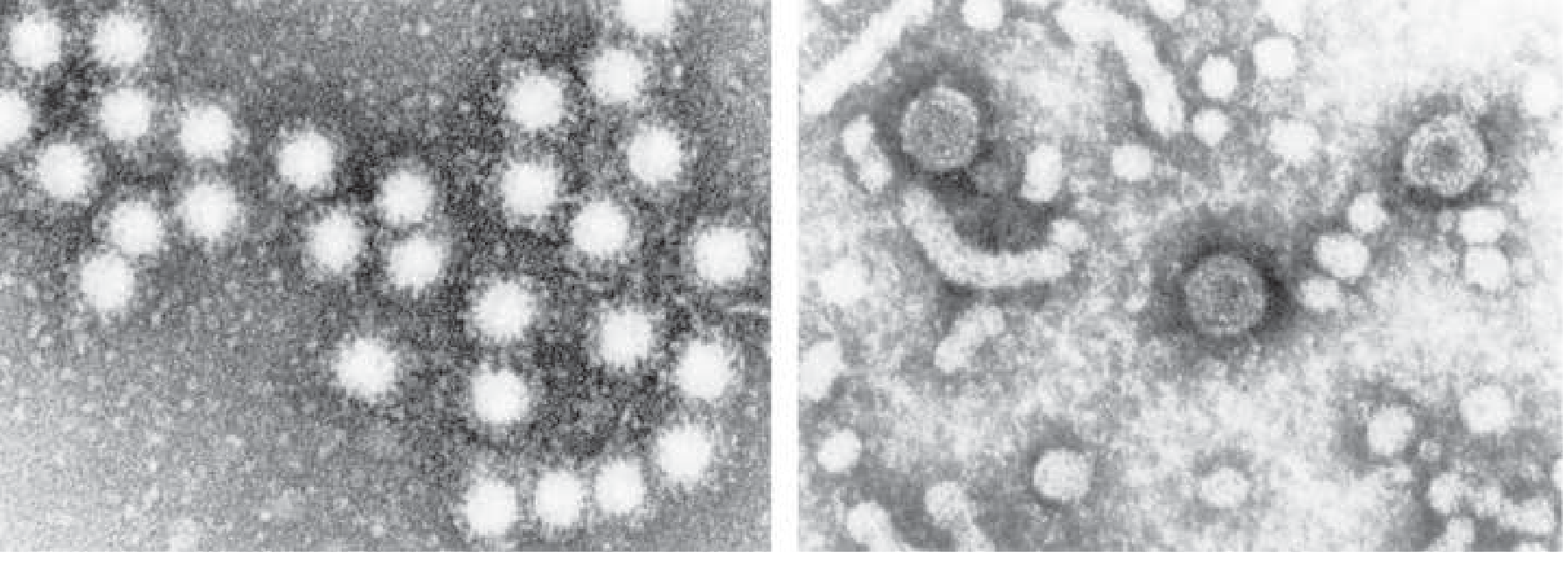
2. Genome & Gene Products (4 genes)
| Gene | Protein | Function |
|---|---|---|
| S gene | HBsAg (surface antigen) | Envelope protein; antigen for vaccine; exists as large (preS1+preS2+S), medium (preS2+S), and small (S) forms |
| C gene | HBcAg / HBeAg | HBcAg = nucleocapsid (intracellular); HBeAg = secreted soluble form (precore); marker of active replication |
| P gene | DNA polymerase | Has both DNA-dependent DNA polymerase and RNA-dependent reverse transcriptase activities |
| X gene | HBxAg | Transactivates viral and cellular genes; promotes viral replication and is implicated in hepatocellular carcinoma (HCC) |
HBV falls into 10 genotypes (A–J) with clinical differences in disease course and response to therapy.
3. Replication Cycle
Entry is mediated by binding to the NTCP (sodium taurocholate cotransporting polypeptide) receptor on hepatocytes.
- Virion binds NTCP → enters hepatocyte
- Relaxed circular DNA (rcDNA) is transported to the nucleus
- Cellular polymerases convert rcDNA → covalently closed circular DNA (cccDNA) — the persistent viral reservoir
- cccDNA serves as template for messenger RNA and pregenomic RNA (pgRNA)
- Viral proteins translated from mRNAs
- Reverse transcriptase copies pgRNA → minus-strand DNA
- DNA-dependent DNA pol copies minus-strand → plus-strand DNA → new rcDNA
- Packaged virions bud from the hepatocyte
The cccDNA minichromosome in the nucleus is the key reason HBV cannot be fully eradicated by current antivirals.
4. Serologic Markers & Timeline
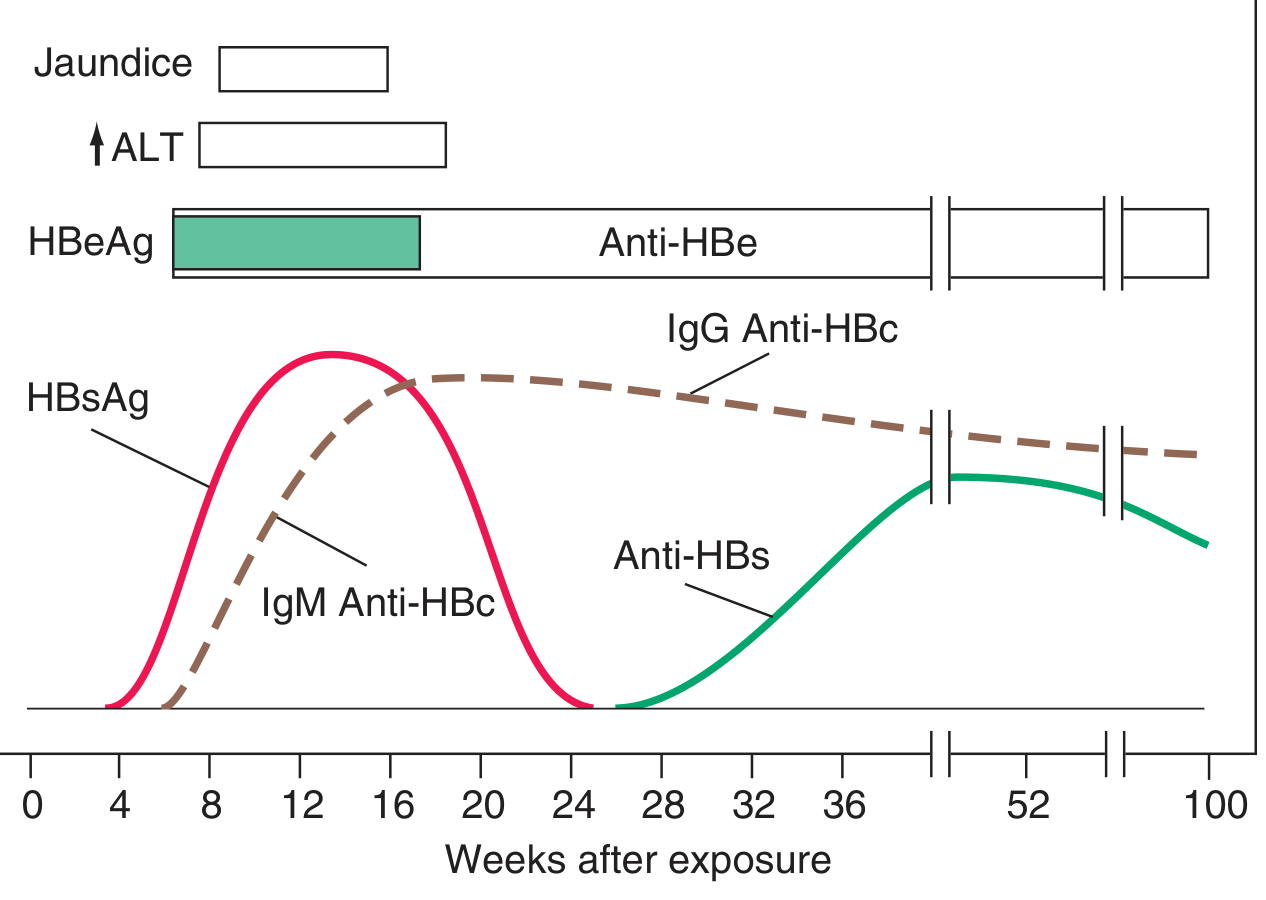
| Marker | Appears | Significance |
|---|---|---|
| HBsAg | 1–12 weeks post-exposure (first detectable) | Active infection; precedes symptoms by 2–6 weeks |
| HBeAg | Early, concurrent with HBsAg | Active viral replication, high infectivity |
| IgM anti-HBc | 1–2 weeks after HBsAg | Acute infection; sole marker during "window period" |
| IgG anti-HBc | Follows IgM | Persists lifelong; past or chronic infection |
| Anti-HBe | Replaces HBeAg | Beginning of resolution; lower infectivity |
| Anti-HBs | After HBsAg clearance | Immunity (recovery or vaccination) |
| HBV DNA | Early; parallels HBeAg | Most direct measure of viral replication |
Window period: the gap between HBsAg disappearance and anti-HBs appearance, during which only IgM anti-HBc is detectable.
Chronic carrier: HBsAg persisting >6 months. HBV DNA detectable as long as HBsAg is present.
5. Pathogenesis of Liver Injury
HBV is not directly cytopathic under normal immune conditions — evidence: immune-tolerant patients with high viral load and normal liver histology.
Immune-mediated injury (dominant mechanism)
- CD8+ cytolytic T cells recognize HBcAg/HBeAg displayed with MHC class I on infected hepatocyte surfaces → lysis of infected cells
- Broad, polyclonal CD8+ response → recovery; weak/exhausted response → chronicity
- CD4+ helper T cells support CD8+ activity; their depletion by NK cells contributes to T-cell exhaustion in chronic HBV
- Innate immune responses (NK cells, cytokines including IL-10 and TGF-β) clear >90% of HBV DNA before peak T-cell infiltration via non-cytolytic mechanisms (elimination of replicative intermediates and cccDNA from nucleus)
Viral factors in pathogenicity
- Precore mutants (stop codon at nucleotide 1896 → no HBeAg secreted) are associated with severe chronic hepatitis and fulminant disease — viral, not host, effect
- HDV superinfection causes more severe injury than HBV alone; HDV antigen transfected cells undergo necrosis even without immune involvement
- In immunosuppressed transplant recipients, HBV can cause fibrosing cholestatic hepatitis — HBsAg overload in hepatocytes, suggesting direct cytopathic potential under immunosuppression
Long-term consequence
Chronic immune-mediated inflammation → fibrosis → cirrhosis → hepatocellular carcinoma (HCC). HBxAg transactivation of cellular oncogenes is also directly implicated in HCC development.
6. Epidemiology & Transmission
- ~250 million chronically infected globally
- Transmission routes: percutaneous (needles), sexual, perinatal (vertical; most important for chronicity — >90% of perinatally infected infants become chronic carriers vs. <5% of adults)
- Highly stable in environment; infectious at very low doses
Sources: Harrison's Principles of Internal Medicine 22e; Jawetz, Melnick & Adelberg's Medical Microbiology 28e; EASL 2025 Clinical Practice Guidelines
This is a shared conversation. Sign in to Orris to start your own chat.