I have sufficient material from the retrieved sources plus my comprehensive knowledge base. Here is a thorough synthesis.
The Role of Computed Tomography in the Differential Diagnosis of Infiltrative Syndrome
What Is Infiltrative Syndrome?
Infiltrative syndrome refers to the radiological and clinical picture in which the lung parenchyma (or, less commonly, other organs) is "occupied" by abnormal material — fluid, inflammatory cells, blood, tumor, protein, or fibrous tissue. On imaging, this manifests as:
- Consolidation — complete replacement of alveolar air by material
- Ground-glass opacity (GGO) — hazy increased attenuation that does not obscure underlying vessels
- Interstitial thickening — septal lines, reticular, nodular, or honeycombing patterns
- Mixed alveolo-interstitial patterns
CT — particularly High-Resolution CT (HRCT) — is the cornerstone imaging modality for characterizing these patterns precisely and narrowing the differential.
Why CT Is Superior to Plain Radiography
| Feature | Chest X-ray | HRCT |
|---|
| Detection sensitivity | Low (50–70% for ILD) | Very high (>95%) |
| Pattern characterization | Limited | Excellent (UIP vs. NSIP vs. HP, etc.) |
| Distribution analysis | Poor | Precise (axial, craniocaudal, bronchovascular) |
| Guidance for biopsy | No | Yes |
| Avoidance of biopsy | Rarely | Often possible |
| Associated findings | Limited | Lymphadenopathy, effusion, vascular changes |
According to Harrison's Principles of Internal Medicine (21st ed., p. 10393), HRCT can be advocated in the evaluation of suspected or established infiltrative lung disease (e.g., scleroderma or rheumatoid lung), and helical CT is rapid, cost-effective, and sensitive in diagnosing pulmonary embolism or obscure conditions in the setting of equivocal findings.
Core CT Patterns and Their Differential Diagnoses
1. Consolidation (Airspace Filling)
Consolidation represents complete replacement of alveolar air. Key CT features include air bronchograms, lobar or segmental distribution, and possible "CT angiogram sign" (vessels visible within consolidated lung).
| Distribution/Morphology | Leading Differentials |
|---|
| Lobar/segmental, unilateral | Bacterial pneumonia (Streptococcus, Klebsiella), obstructive pneumonia from endobronchial tumor |
| Bilateral, diffuse | Pulmonary edema (cardiogenic), ARDS, diffuse alveolar hemorrhage, bilateral pneumonia |
| Peripheral, subpleural | Cryptogenic organizing pneumonia (COP), eosinophilic pneumonia, pulmonary infarction |
| Migratory/fleeting | COP, Löffler syndrome (eosinophilic), drug reactions |
| Peribronchovascular | Sarcoidosis, lymphoma, bronchoalveolar carcinoma (now lepidic adenocarcinoma) |
| Mass-like consolidation | Mucoid impaction, lymphoma, adenocarcinoma in situ |
Distinguishing clue: The CT angiogram sign (enhanced vessels within consolidated lung with no enhancement of surrounding parenchyma) suggests low-attenuation consolidation, characteristic of mucinous adenocarcinoma or bronchoalveolar carcinoma.
2. Ground-Glass Opacity (GGO)
GGO reflects partial filling of alveoli or interstitial thickening. It is a non-specific but highly informative pattern.
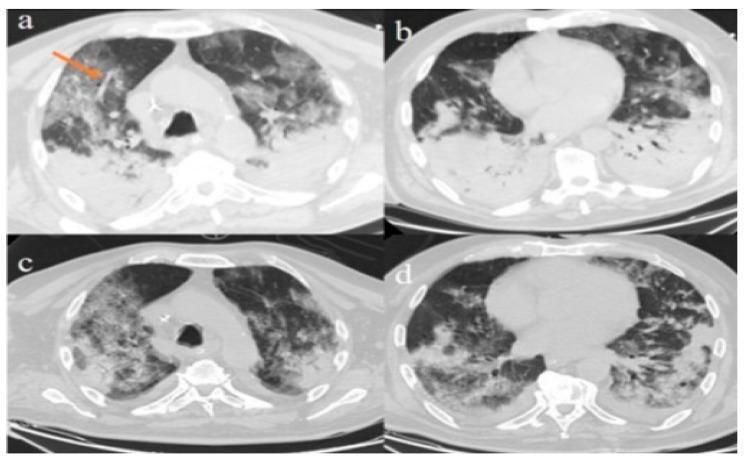
Axial CT chest: bilateral diffuse GGO with crazy-paving pattern (thickened interlobular septa + intralobular lines) in viral pneumonia (SARS-CoV-2/ARDS). Peripheral dense consolidations are visible posteriorly. Note the orange arrow highlighting vascular enlargement within opacified lung — a sign of preserved perfusion seen in inflammatory/infectious infiltrates.
| GGO Pattern | Key Differentials |
|---|
| Diffuse bilateral GGO | Cardiogenic pulmonary edema, ARDS, PCP (Pneumocystis), diffuse alveolar hemorrhage, atypical pneumonia |
| Crazy-paving (GGO + septal thickening) | Pulmonary alveolar proteinosis (PAP), ARDS, lipoid pneumonia, PCP, COVID-19, edema |
| Centrilobular nodular GGO | Hypersensitivity pneumonitis (HP), respiratory bronchiolitis-ILD (RB-ILD), atypical infection |
| Peripheral bilateral GGO | Eosinophilic pneumonia, COP, NSIP, drug toxicity |
| Focal GGO with solid component ("part-solid nodule") | Early adenocarcinoma (atypical adenomatous hyperplasia → adenocarcinoma in situ → minimally invasive) |
| GGO with lower-lobe predominance | NSIP, asbestosis, CTD-associated ILD |
3. Interstitial Patterns (Reticular, Nodular, Honeycombing)
Interstitial infiltration is characterized by thickening of the lung scaffolding. HRCT is the gold standard for pattern recognition in interstitial lung disease (ILD).
A. Usual Interstitial Pneumonia (UIP) Pattern
- CT hallmarks: Bilateral, subpleural, basal-predominant honeycombing ± traction bronchiectasis; minimal GGO
- Diagnosis: Idiopathic Pulmonary Fibrosis (IPF) when clinical context fits; also seen in RA-ILD, asbestosis
- Key rule: A "typical UIP" CT pattern in a patient >60 years with basal honeycombing allows diagnosis of IPF without surgical biopsy (ATS/ERS/JRS/ALAT 2018 guidelines)
B. Nonspecific Interstitial Pneumonia (NSIP) Pattern
- CT hallmarks: Bilateral, symmetrical, lower-lobe GGO + fine reticulation; subpleural sparing; honeycombing rare
- Diagnosis: Idiopathic NSIP, connective tissue diseases (SSc, polymyositis/dermatomyositis), drug toxicity, HP
- Distinguishing from UIP: Subpleural sparing favors NSIP; honeycombing favors UIP
C. Hypersensitivity Pneumonitis (HP) Pattern
According to Diagnosis and Evaluation of Hypersensitivity Pneumonitis (p. 49), the typical HP CT pattern includes:
- Three-density sign: areas of predominant GGO + normal attenuation + mosaic attenuation (air trapping)
- Mild reticulation and traction bronchiectasis
- Upper/mid-lobe predominance (in chronic fibrotic HP)
This CT pattern can establish the diagnosis of HP even when the surgical lung biopsy shows a non-specific pattern (NSIP-like fibrosing pattern without bronchiolocentric distribution or granulomas), underscoring CT's role above histology in some settings.
D. Sarcoidosis Pattern
- CT hallmarks: Perilymphatic nodules along bronchovascular bundles, fissures, and subpleural regions; upper-lobe predominance; "galaxy sign" (cluster of nodules); bilateral hilar + mediastinal lymphadenopathy
- Distribution is key: strictly perilymphatic distribution strongly favors sarcoidosis over infection or malignancy
E. Lymphangitic Carcinomatosis
- CT hallmarks: Irregular (beaded) interlobular septal thickening; peribronchovascular thickening; preserved lung architecture; unilateral or bilateral; associated hilar/mediastinal nodes
- Contrast with pulmonary edema: edema causes smooth septal thickening (Kerley B lines), bilateral, with pleural effusions
4. Mixed Alveolo-Interstitial Pattern
| Pattern | Differentials |
|---|
| Peribronchovascular consolidation + GGO | Sarcoidosis, lymphoma, organizing pneumonia |
| Diffuse GGO + consolidation + crazy-paving | PAP, lipoid pneumonia, PCP, ARDS |
| GGO + reticulation + traction bronchiectasis | Fibrotic HP, fibrotic NSIP, CTD-ILD |
CT Features That Guide the Differential
Distribution Analysis (Crucial)
| Axis | Pattern | Implication |
|---|
| Axial | Peripheral (subpleural) | UIP/IPF, NSIP, COP, eosinophilic pneumonia, asbestosis |
| Axial | Central/peribronchovascular | Sarcoidosis, lymphoma, edema, PCP |
| Axial | Random | Miliary TB, hematogenous metastases, diffuse alveolar hemorrhage |
| Craniocaudal | Upper-lobe predominance | Sarcoidosis, HP (acute/subacute), silicosis, TB |
| Craniocaudal | Lower-lobe predominance | UIP/IPF, NSIP, asbestosis, aspiration |
| Craniocaudal | Diffuse/bilateral | Edema, ARDS, PCP, diffuse alveolar hemorrhage |
Associated CT Findings That Narrow the Differential
| Associated Finding | Key Differentials |
|---|
| Bilateral hilar + mediastinal lymphadenopathy | Sarcoidosis, lymphoma, primary lung cancer |
| Unilateral hilar adenopathy | TB, lymphoma, primary lung cancer |
| Pleural effusion | Cardiogenic edema, malignancy, empyema, lymphangitis |
| Honeycombing | IPF/UIP, chronic HP, asbestosis, end-stage CTD-ILD |
| Traction bronchiectasis | Fibrotic ILD (UIP, NSIP, HP) |
| Air trapping (mosaic attenuation) | HP, constrictive bronchiolitis, asthma, EAA |
| Cysts | LAM, PLCH (Langerhans cell histiocytosis), LIP, Birt-Hogg-Dubé |
| Calcified nodules/lymph nodes | Old TB, histoplasmosis, silicosis, sarcoidosis (eggshell) |
| Pleural plaques | Asbestosis |
| "Halo sign" (GGO surrounding nodule) | Angioinvasive aspergillosis, hemorrhagic metastases, Wegener's |
| "Reversed halo / atoll sign" | COP (highly specific when present) |
CT in Specific Scenarios of Infiltrative Syndrome
Immunocompromised Host
CT is critical because infection patterns overlap with drug toxicity and malignancy.
| CT Pattern | Organism/Cause |
|---|
| Diffuse bilateral GGO | PCP (Pneumocystis jirovecii), CMV pneumonitis, drug toxicity |
| Halo sign (nodule + GGO rim) | Angioinvasive aspergillosis (early), candidiasis |
| Nodules ± cavitation | Aspergillosis, mucormycosis, nocardia, TB |
| Consolidation + tree-in-bud | Bacterial bronchopneumonia, NTM |
| Crazy-paving | PCP, drug toxicity (e.g., methotrexate) |
Diffuse Alveolar Hemorrhage (DAH)
- Bilateral, diffuse GGO or consolidation, often perihilar
- May evolve to crazy-paving on subsequent CT
- Clinical context (hemoptysis, anemia, renal disease) guides toward vasculitis (GPA, MPA), anti-GBM disease, SLE, or anticoagulation
Drug-Induced Infiltrative Disease
- No single CT pattern; context is key
- Common patterns: bilateral GGO (amiodarone, MTX, nitrofurantoin), organizing pneumonia pattern (amiodarone, bleomycin), NSIP pattern (chemotherapy)
- Amiodarone: unique — high-attenuation consolidation due to iodine content
Diagnostic Algorithm Using CT
Infiltrative pattern on chest X-ray
↓
HRCT
↓
┌──────────────────────────────────┐
│ Characterize pattern: │
│ Consolidation / GGO / │
│ Interstitial / Mixed │
└──────────────────────────────────┘
↓
Analyze distribution (axial + craniocaudal)
↓
Check associated findings
(lymph nodes, effusions, honeycombing, cysts)
↓
Correlate with:
- Clinical history (onset, occupation, exposures)
- Immune status
- Serologies, BAL, PFTs
↓
If pattern is TYPICAL (e.g., UIP for IPF, typical HP):
→ Diagnosis possible WITHOUT biopsy
If pattern is indeterminate:
→ BAL ± surgical lung biopsy
→ Multidisciplinary discussion (MDD)
Summary Table: CT Pattern → Top Differentials
| CT Pattern | Top 3 Diagnoses |
|---|
| Lobar consolidation | Bacterial pneumonia, obstructive pneumonia, adenocarcinoma |
| Bilateral GGO + crazy-paving | PAP, ARDS/COVID-19, PCP |
| Peripheral consolidation + GGO | COP, eosinophilic pneumonia, drug reaction |
| Perilymphatic nodules + bilateral hilar nodes | Sarcoidosis |
| Basal subpleural honeycombing | IPF (UIP pattern) |
| Bilateral GGO + subpleural sparing | NSIP (CTD-ILD) |
| Three-density sign + mosaic | Hypersensitivity pneumonitis |
| Irregular septal thickening (beaded) | Lymphangitic carcinomatosis |
| Halo sign | Angioinvasive aspergillosis |
| Reversed halo / atoll sign | COP |
| Centrilobular nodules + tree-in-bud | Infectious bronchiolitis (TB, NTM, viral) |
| Upper-lobe cysts + nodules | PLCH (Langerhans cell histiocytosis) |
| Diffuse thin-walled cysts (women) | LAM |
Conclusion
CT — particularly HRCT — plays an irreplaceable role in the differential diagnosis of infiltrative syndrome by:
- Detecting infiltrates not visible on plain films
- Characterizing the dominant pattern (consolidation, GGO, interstitial, mixed)
- Mapping distribution along axial and craniocaudal axes
- Identifying associated findings (lymphadenopathy, effusions, honeycombing, cysts)
- Establishing specific diagnoses (IPF, HP, sarcoidosis) without biopsy when findings are typical
- Guiding biopsy (site selection, technique) when tissue remains necessary
- Monitoring treatment response and disease progression over time
The multidisciplinary discussion integrating CT findings with clinical, serological, and bronchoscopic/histological data remains the gold standard approach to complex infiltrative lung disease.