Give me everything about the opiod poisoning
Reading File
Finding Sources
Searching PubMed
opioid poisoning overdose management
Searching the Web
opioid poisoning toxicology clinical management 2025
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Loading Image
Reading File
Reading File
Finding Sources
I now have comprehensive information from multiple authoritative textbooks. Let me compile the complete answer.
Opioid Poisoning — Comprehensive Overview
1. Definition & Epidemiology
Opioid poisoning (overdose) is a life-threatening medical emergency resulting from excessive stimulation of opioid receptors, most critically causing respiratory depression leading to hypoxic death. It occurs with illicit opioids (heroin, fentanyl, nitazenes), prescribed opioids (morphine, oxycodone, methadone, buprenorphine), and synthetic analogues.
- In 2020, opioids were involved in 75% of all drug overdose deaths in the United States; 82.3% of opioid-involved deaths involved synthetic opioids (primarily fentanyl).
- Three epidemic "waves" have been described: (1) prescription opioids from the 1990s; (2) heroin from 2010; (3) synthetic opioids (fentanyl and analogues) from 2013 onward.
- Fentanyl is 50–100× more potent than morphine and 10× more potent than heroin.
- Street heroin purity in the U.S. has risen from ~4% to 45–75%, increasing the risk of unintentional overdose.
— Goodman & Gilman's The Pharmacological Basis of Therapeutics, p. 561; Harrison's Principles of Internal Medicine 22E (2025), p. 3734
2. Receptor Pharmacology & Pathophysiology
Opioids act on five classes of receptors: μ (mu), κ (kappa), δ (delta), σ (sigma), and the nociceptin receptor. The μ-receptor is primarily responsible for analgesia, euphoria, and — critically — respiratory depression and miosis.
Key mechanisms of toxicity:
- Respiratory depression: Direct narcotic depression of brainstem respiratory centers (medullary respiratory nuclei), causing shallow, slow respirations (2–8/min), periodic breathing, or complete apnea. This is the primary mechanism of death.
- CNS depression: Stupor, coma, unresponsiveness to stimulation.
- Miosis (pinpoint pupils): Mediated by the Edinger-Westphal nucleus; in late severe anoxia, pupils paradoxically dilate and become non-reactive.
- Cardiovascular depression: Bradycardia, hypotension (only dramatic with severe anoxia); cardiac arrhythmias may be related to anoxia or adulterants (e.g., quinine in heroin).
- Pulmonary edema: A frequent complication, particularly with heroin overdose (pathogenesis not fully understood).
- Hypothermia: Opiates suppress thermoregulation; however, by the time patients reach hospital, aspiration pneumonia may have caused normal or elevated temperature.
- Fentanyl-specific: Can produce respiratory muscle rigidity and laryngospasm ("wooden chest" syndrome), complicating bag-mask ventilation.
— Plum and Posner's Diagnosis and Treatment of Stupor and Coma, p. 468; Adams and Victor's Principles of Neurology, p. 1202; Kaplan & Sadock's Comprehensive Textbook of Psychiatry, p. 4249
3. Clinical Presentation
The Classic Opioid Toxidrome (Triad)
| Feature | Finding |
|---|---|
| Consciousness | Stupor → coma, unresponsive to stimulation |
| Pupils | Pinpoint, reactive (miosis) |
| Respirations | Slow (2–8/min), shallow, or apneic |
Additional Signs
- Skin: Cold, clammy, cyanosis
- Temperature: Hypothermia
- Heart rate: Bradycardia; blood pressure decreased (dramatically only with severe anoxia)
- Neurological: Slurred speech, impaired attention, flaccid muscle tone
- GI: Nausea, vomiting, constipation (in milder intoxication)
- Pulmonary: Froth at mouth, X-ray evidence of pulmonary edema, aspiration pneumonia
- Late/severe: Pupils become large and non-reactive (from hypoxia, not the opioid itself); cardiovascular collapse
Mild Intoxication Signs
Anorexia, nausea, vomiting, constipation, loss of sexual interest
DSM-5 Criteria for Opioid Intoxication
- Behavioral/psychological changes (initial euphoria → apathy, dysphoria, psychomotor agitation or retardation, impaired judgment)
- Pupillary constriction
- Drowsiness or coma
- Slurred speech
- Impairment in attention or memory
— Adams and Victor's Principles of Neurology, p. 1202; Kaplan & Sadock's Comprehensive Textbook of Psychiatry, p. 4249
4. Populations at Increased Risk
- Children: Increased susceptibility; small doses may be toxic
- Adults with: myxedema, Addison disease, chronic liver disease, pneumonia
- Persons who have lost tolerance after a period of abstinence (formerly well-tolerated dose may be fatal on re-use)
- Polydrug users (opioids + benzodiazepines — major fatal combination)
- Users of illicit opioids with variable or unknown purity
— Adams and Victor's Principles of Neurology, p. 1202
5. Specific Drug Considerations
| Opioid | Special Concern |
|---|---|
| Fentanyl / analogues | 50–100× potency; may require naloxone doses ≥10 mg; wooden chest syndrome |
| Methadone | Long half-life; naloxone drip for 24–72 h; QTc prolongation; overdose common with early prescribing |
| Buprenorphine | Partial agonist — nearly impossible to overdose alone; however, combined with benzodiazepines causes serious toxicity; high naloxone doses needed (≥10 mg) |
| Meperidine | Can cause seizures (unlike most opioids); accumulation of normeperidine metabolite |
| Tramadol | Seizures at high doses |
| Heroin | Pulmonary edema common; purity variable |
| Propoxyphene | Can cause seizures |
| Nitazenes | Ultra-potent novel synthetic opioids; high naloxone requirements |
— Harrison's Principles of Internal Medicine 22E, p. 3734; Maudsley Prescribing Guidelines, p. 523
6. Diagnosis
Primarily clinical — do not wait for laboratory results before treating.
- Recognise the opioid toxidrome: coma + pinpoint pupils + respiratory depression
- Toxicology screen (blood/urine): can confirm but delays management; standard screens may miss fentanyl analogues and novel synthetics
- Response to naloxone is both diagnostic and therapeutic (failure to respond should prompt consideration of other diagnoses)
- Consider co-ingestions: benzodiazepines (most common deadly combination), alcohol, other CNS depressants
- ECG: arrhythmias, QTc prolongation (especially methadone)
7. Management
Emergency Algorithm
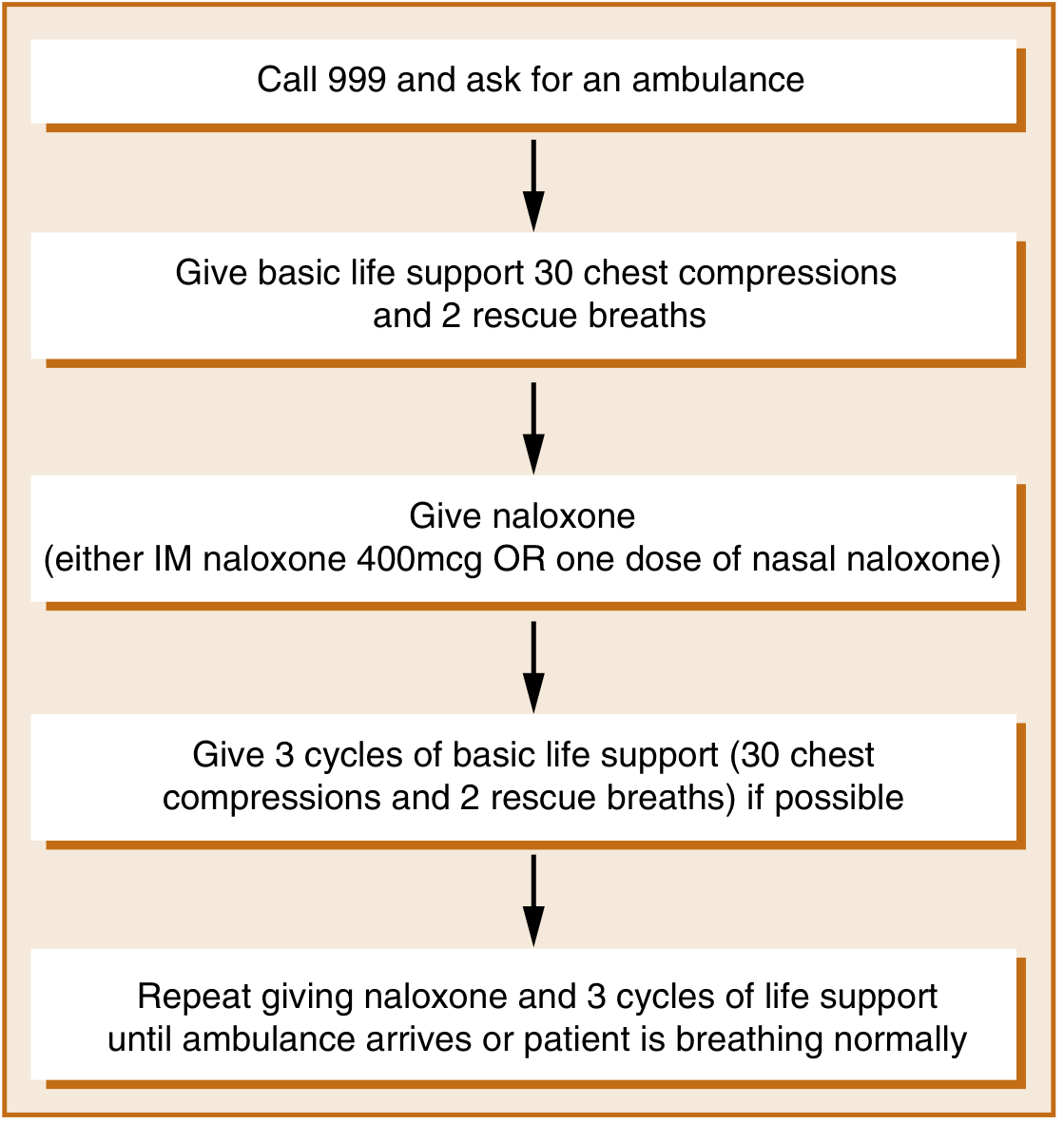
Figure: Flowchart for naloxone administration (adapted from WHO 2014). — Maudsley Prescribing Guidelines, 15th Ed., p. 523
Step-by-Step Management (Harrison's Table 467-2)
- Establish and protect the airway — intubation and mechanical ventilation may be necessary
- Assist ventilation — bag-mask ventilation; note that fentanyl may cause wooden chest syndrome requiring succinylcholine for intubation
- Activated charcoal — for recent large oral ingestions
- Naloxone (antidote — see below)
- Monitor vital signs, oxygen saturation, respiratory rate, GCS
- IV access, fluids — support blood pressure
- Gastric lavage — may be useful for oral ingestion (even hours later, because opioid-induced ileus delays gastric emptying)
- Treat co-ingestions — flumazenil 0.2 mg/min for benzodiazepines (caution: may precipitate seizures, ↑ICP)
Naloxone (Key Antidote)
| Route | Dose | Notes |
|---|---|---|
| IV | 0.4–2.0 mg | Onset ~1–2 min; preferred |
| IM | 400 mcg | Used by laypersons/pre-hospital |
| Intranasal | 2–4 mg | Equivalent onset to 400 mcg IM; longer-lasting peak; preferred for laypersons |
| Endotracheal | 0.4–2.0 mg | If no IV access |
Repeat dosing: Repeat every 2–3 min as needed. For fentanyl/buprenorphine/nitazenes, total doses of 10 mg or more may be required.
Infusion: Once response achieved, a continuous IV naloxone drip is often required because naloxone's half-life (~60–90 min) is shorter than most opioids. Give ½ to ⅔ of the effective initial reversal dose per hour and monitor closely for 24–72 hours depending on opioid type (e.g., morphine vs. methadone).
Goal: Reverse respiratory depression — not to achieve full consciousness. Avoid excessive naloxone in dependent patients, as it will precipitate acute withdrawal.
Nalmefene (Newer Alternative)
- Higher potency, plasma half-life ~11 hours (vs. 60–90 min for naloxone)
- Recently approved for overdose treatment
- No clear advantage in emergency practice over naloxone per current evidence
— Harrison's 22E, p. 3734; Adams and Victor's Neurology, p. 1202; Maudsley Prescribing Guidelines, p. 523–524
8. Complications
| Complication | Mechanism |
|---|---|
| Respiratory arrest / death | Direct brainstem depression — primary cause of death |
| Hypoxic-ischemic encephalopathy | Sustained apnea → cerebral hypoxia |
| Pulmonary edema | Especially with heroin overdose |
| Aspiration pneumonia | Loss of airway protective reflexes |
| Posthypoxic leukoencephalopathy | Begins ~1 week post-hypoxic episode; cognitive impairment, blindness, movement disorder; diffuse white matter T2 signal on MRI — also seen with "chasing the dragon" (heroin inhalation) |
| Rhabdomyolysis | Muscle injury from prolonged immobility |
| Seizures | Uncommon; with meperidine, tramadol, propoxyphene, or polydrug use; NOT a typical opioid feature |
| Cardiac arrhythmias | Anoxia, electrolyte disturbance, adulterants |
| Precipitated withdrawal | From excess naloxone in dependent users |
— Plum and Posner, p. 468; Adams and Victor, p. 1202
9. Opioid Withdrawal
Withdrawal is physiologically the opposite of intoxication (not life-threatening in healthy adults, but intensely distressing).
Symptoms: Anxiety, irritability, restlessness, "achy" legs and back, lacrimation, rhinorrhea, yawning, piloerection ("gooseflesh"), sweating, sneezing, diarrhea, vomiting, insomnia, tachycardia, hypertension, mydriasis. Seizures and delirium are not features of opioid withdrawal (their presence suggests another cause, e.g., alcohol withdrawal).
Precipitated withdrawal (from naloxone in a dependent patient): Often dramatic, with severe hypertension, tachycardia, and agitation — can be difficult to treat. Use the minimum naloxone dose needed.
Detoxification Options
- Methadone: Administer 10 mg orally for objective withdrawal signs every 2–4 h for 24 h → this total is the "stabilization dose" (not to exceed 40 mg); then reduce by 5 mg/day
- Buprenorphine: Initiate after objective withdrawal signs appear (COWS score); taper over days
- α2-Adrenergic agonists (clonidine): 0.1–0.3 mg PO q1h (up to 4 doses); reduces autonomic withdrawal symptoms off-label
10. Longer-Term Treatment (Opioid Use Disorder)
Three FDA-approved medications:
| Drug | Class | Mechanism | Notes |
|---|---|---|---|
| Methadone | Full μ-agonist | Long-acting; cross-tolerance; once daily | Only dispensed via licensed OTPs; ↓overdose & all-cause mortality; 60–80 mg/day more effective |
| Buprenorphine | Partial μ-agonist | Ceiling effect limits overdose risk; long-acting | Office-based prescribing available; combined with naloxone (Suboxone) to deter injection |
| Naltrexone | μ-antagonist | Blocks opioid effects; no abuse potential | Only after full detox; risk of overdose if lapse (tolerance lost) |
Opioid Substitution Treatment (OST) goals: reduce withdrawal symptoms, eliminate non-prescribed drug use, stabilize lifestyle, reduce injecting harm.
11. Prevention
- Take-home naloxone: Patients discharged on >50 morphine milligram equivalents/day should receive naloxone prescription; friends and family trained in administration
- Intranasal naloxone kits (2 mg) are accessible for laypersons
- Warning patients about loss of tolerance after any period of abstinence
- Prescription drug monitoring programs
- Harm reduction: fentanyl test strips, supervised consumption
— Washington Manual of Medical Therapeutics; Maudsley Prescribing Guidelines, p. 524
Recent Evidence Note
A 2026 systematic review (PMID: 41817965) examined supratherapeutic loperamide toxicity — relevant because loperamide (an opioid used for diarrhea) is increasingly abused at massive doses for opioid-like CNS effects, causing cardiac toxicity (QRS widening, arrhythmias) in addition to the standard opioid toxidrome. This represents an emerging variant of opioid poisoning with distinct cardiac management considerations.
Sources: Harrison's Principles of Internal Medicine 22E (2025); Adams and Victor's Principles of Neurology 12th Ed.; Goodman & Gilman's Pharmacological Basis of Therapeutics; Plum and Posner's Diagnosis and Treatment of Stupor and Coma; Kaplan & Sadock's Comprehensive Textbook of Psychiatry; Maudsley Prescribing Guidelines in Psychiatry 15th Ed.; Goldman-Cecil Medicine; Washington Manual of Medical Therapeutics
This is a shared conversation. Sign in to Orris to start your own chat.