pulmonary infiltrate syndrome
Reading File
search_medical_text
pulmonary infiltrate syndrome etiology classification diagnosis
search_medical_text
pulmonary infiltrate management differential diagnosis treatment
search_medical_image
pulmonary infiltrate chest X-ray CT imaging findings
GradeResponse
GradeResponse
GradeResponse
Pulmonary Infiltrate Syndrome
Definition
"Pulmonary infiltrate" is a radiologic term describing material of increased density in the lung parenchyma — fluid, pus, blood, cells, or protein — that partially or completely obliterates air spaces. "Pulmonary infiltrate syndrome" refers to the clinical constellation of respiratory symptoms combined with radiographically visible parenchymal opacities, requiring systematic evaluation to identify the underlying cause.
Classification of Pulmonary Infiltrates
By Pattern
| Pattern | Description | Common Causes |
|---|---|---|
| Consolidation | Air replaced by solid/liquid material; air bronchograms often present | Bacterial pneumonia, pulmonary edema, hemorrhage |
| Ground-glass opacity (GGO) | Hazy increased density without obliterating vessels | Viral pneumonia, early edema, interstitial disease |
| Interstitial | Linear, reticular, or nodular markings along interstitium | ILD, lymphangitic carcinomatosis, sarcoidosis |
| Nodular | Focal rounded opacities | Malignancy, fungal infections, granulomas |
| Miliary | Diffuse small nodules | TB, histoplasmosis, metastases |
By Distribution
| Distribution | Common Association |
|---|---|
| Lobar/segmental | Bacterial (lobar) pneumonia |
| Bilateral perihilar ("bat-wing") | Pulmonary edema, PCP |
| Peripheral/subpleural | Eosinophilic pneumonia, COP |
| Basal predominance | Aspiration, atelectasis, edema |
| Upper lobe predominance | TB reactivation, sarcoidosis |
Etiology
Infectious
- Bacterial: S. pneumoniae, S. aureus, Klebsiella, Legionella, atypicals (Mycoplasma, Chlamydophila)
- Viral: Influenza, SARS-CoV-2, CMV, RSV
- Fungal: Aspergillus, Pneumocystis jirovecii (PCP), Histoplasma, Coccidioides
- Mycobacterial: M. tuberculosis, NTM
Non-Infectious
- Cardiogenic pulmonary edema — hydrostatic fluid leak from elevated PCWP
- ARDS — diffuse alveolar damage from sepsis, trauma, aspiration, etc.
- Eosinophilic syndromes — drugs (NSAIDs, antibiotics), environmental exposures, radiation therapy, idiopathic (AEP, CEP) (Harrison's, p. 7968)
- Organizing pneumonia (COP/BOOP) — idiopathic or secondary to infection/drugs
- Diffuse alveolar hemorrhage (DAH) — vasculitis (GPA, MPA), anticoagulation, bone marrow transplant
- Malignancy — lymphoma, bronchoalveolar cell carcinoma, lymphangitic spread
- Sarcoidosis, hypersensitivity pneumonitis, drug toxicity
Special Context: Acute Chest Syndrome (Sickle Cell Disease)
Chest pain, cough, fever, hypoxia, and pulmonary infiltrate on chest X-ray are the major diagnostic criteria. Etiology is multifactorial: in situ vaso-occlusion, fat/bone marrow emboli, infection, and post-operative hypoventilation — occurring in >50% of sickle cell patients, often repeatedly (Harrison's, p. 2920).
Clinical Presentation
| Feature | Details |
|---|---|
| Symptoms | Cough (productive or dry), fever, dyspnea, pleuritic chest pain, hemoptysis |
| Signs | Tachypnea, tachycardia, dullness to percussion, bronchial breath sounds, crackles, hypoxia |
| Red flags | SpO₂ <90%, bilateral involvement, rapid progression, multi-organ dysfunction |
Diagnostic Approach
Imaging
Chest X-ray is the first-line modality. CT chest (HRCT) offers superior sensitivity and pattern characterization, particularly for subtle or early infiltrates and differentiation of GGO vs. consolidation vs. interstitial patterns.
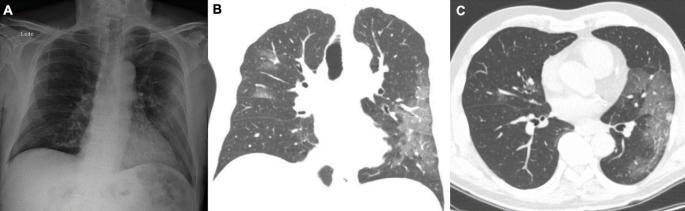
Laboratory
- CBC: leukocytosis → infection; eosinophilia → drug/parasitic/eosinophilic syndrome
- CRP, ESR, procalcitonin — inflammatory/infectious burden
- Blood cultures (before antibiotics), sputum Gram stain & culture
- Urinary antigens: Legionella, S. pneumoniae
- Respiratory viral PCR panel
- Serology: fungal (galactomannan, beta-D-glucan), HIV
- BNP/NT-proBNP — cardiogenic edema
- BAL — essential when diagnosis is unclear (culture, cytology, hemosiderin-laden macrophages for DAH)
- Bronchoscopy ± biopsy — tissue diagnosis when malignancy or unusual infection suspected
Key Differentiating Clues
| Clinical Context | Consider |
|---|---|
| Immunocompromised host | PCP, CMV, invasive fungal, TB reactivation |
| Drug history (NSAIDs, nitrofurantoin, amiodarone) | Drug-induced pulmonary toxicity or eosinophilic pneumonia |
| Recent hospitalization/aspiration risk | Aspiration pneumonia/pneumonitis |
| Heart failure history + bilateral basal infiltrates | Cardiogenic edema |
| Hemoptysis + peripheral infiltrates | DAH, vasculitis |
| Eosinophilia + peripheral infiltrates | Eosinophilic pneumonia, Löffler syndrome |
| Travel history | Endemic mycoses, tropical parasites |
Management
General Principles
- Stabilize — supplemental O₂ for SpO₂ <95%; avoid overhydration; high-flow O₂ or NIV/intubation for severe hypoxia
- Identify and treat the cause — specific therapy depends on etiology (see below)
- Empiric therapy when diagnosis is pending in unstable patients
Infectious
- CAP: Beta-lactam + macrolide (or respiratory fluoroquinolone) per IDSA/ATS guidelines; cover atypicals
- HAP/VAP: Broad-spectrum anti-pseudomonal + MRSA coverage
- PCP: High-dose TMP-SMX; add prednisone if PaO₂ <70 mmHg
- Invasive aspergillosis: Voriconazole first-line
Non-Infectious
- Cardiogenic edema: Diuresis, afterload reduction, treat underlying cardiac cause
- ARDS: Lung-protective ventilation (low tidal volumes 6 mL/kg IBW), prone positioning, conservative fluid strategy
- Eosinophilic pneumonia / drug-induced: Remove the offending agent (mainstay of treatment — Harrison's, p. 7968); systemic corticosteroids for moderate-severe cases
- COP/BOOP: Prednisone 0.75–1 mg/kg/day, taper over 6–12 months
- DAH: High-dose methylprednisolone; treat underlying vasculitis; plasmapheresis for anti-GBM or ANCA-associated
- Acute Chest Syndrome (SCD): Antibiotics, supplemental O₂, judicious hydration, avoid excessive opioids; exchange transfusion for severe/multi-lobe disease (Harrison's, p. 2920)
Complications
- Respiratory failure requiring mechanical ventilation
- Sepsis and multi-organ failure
- Empyema / lung abscess (cavitation)
- Fibrosis and chronic lung disease (post-ARDS, post-organizing pneumonia)
- Recurrence (especially in eosinophilic syndromes without corticosteroid maintenance)
Summary Algorithm
Pulmonary infiltrate on imaging
↓
Clinical context + pattern + distribution
↓
Infectious? → Cultures, PCR, serology, empiric ABx
↓
Non-infectious?
├── Cardiogenic → BNP, echo, diuresis
├── Eosinophilic → CBC diff, drug history, steroids
├── Hemorrhagic → BAL (bloody lavage doesn't clear), ANCA/anti-GBM
├── Organizing pneumonia → CT pattern, BAL, biopsy → steroids
└── Malignant → CT/PET, bronchoscopy, biopsy
The term "pulmonary infiltrate syndrome" is fundamentally a radiologic trigger for a clinical diagnostic workup rather than a single disease entity. The breadth of the differential demands systematic pattern analysis, clinical context, and targeted testing before committing to a diagnosis.
This is a shared conversation. Sign in to Orris to start your own chat.