The internal structure of the kidney. Nephron. Blood vessels of the kidney, venous and lymphatic outflow, innervation
Reading File
Finding Sources
Finding Sources
Searching Images
kidney internal structure nephron anatomy diagram
Finding Sources
Finding Sources
Loading Image
Reading File
Reading File
Finding Sources
Reading File
Reading File
Searching Images
renal blood vessels cortex medulla arcuate interlobular arteries peritubular capillaries
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Finding Sources
Reading File
I now have comprehensive information from multiple authoritative sources. Let me compile the full answer.
Internal Structure of the Kidney, Nephron, Blood Vessels, Venous and Lymphatic Outflow, and Innervation
1. Gross Internal Structure
On a bisected kidney, two main regions are visible:
- Cortex — the granular outer zone (~1 cm thick in humans). Its granularity results from glomerular tufts and densely packed convoluted tubules. The cortex contains the cortical labyrinth (glomeruli, proximal and distal convoluted tubules) and medullary rays (straight portions of tubules and collecting ducts projecting into the cortex from medullary pyramids).
- Medulla — the darker striated inner zone, subdivided into 8–18 conical renal pyramids with bases at the corticomedullary junction and apices forming papillae. Each papilla bears the area cribrosa, perforated by openings of the collecting ducts (ducts of Bellini), through which urine flows into the minor calyces.
Each renal lobe = one pyramid + its surrounding cortex. Cortical tissue dipping between pyramids forms the columns of Bertin. The outer medulla is subdivided into an outer stripe (containing proximal straight tubules and collecting ducts) and an inner stripe (containing thick ascending limbs and vasa recta); the inner medulla contains thin limbs and inner medullary collecting ducts.
The renal sinus is the central space enclosed by renal parenchyma, containing the renal pelvis with major/minor calyces, blood vessels, nerves, and fat.
2. The Nephron
The nephron is the structural and functional unit of the kidney. Each human kidney contains ~1 million nephrons (range ~600,000–1.5 million). A nephron consists of the renal corpuscle and a renal tubule with distinct segments.
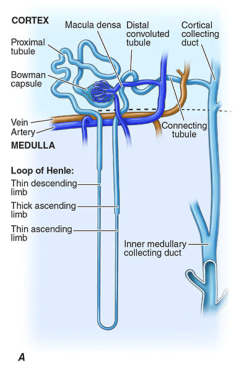
2a. Renal Corpuscle (Malpighian Corpuscle)
Located entirely in the cortex. Consists of:
- Bowman's capsule — a double-walled epithelial cup. The parietal layer is simple squamous epithelium; the visceral layer is composed of podocytes (highly modified visceral epithelial cells).
- Glomerulus — a tuft of fenestrated capillaries (~30–40 loops) invaginated into Bowman's capsule.
Four cell types in the glomerulus:
- Fenestrated endothelial cells — fenestrae 70–100 nm in diameter; form the first filtration barrier.
- Mesangial cells — phagocytic, contractile; provide structural support; produce extracellular matrix; modulate glomerular blood flow.
- Parietal epithelial cells — line Bowman's capsule.
- Podocytes (visceral epithelial cells) — wrap capillaries with primary and secondary foot processes (pedicels). Filtration slits between adjacent foot processes (~30–40 nm wide) are bridged by slit diaphragms composed of nephrin, podocin, and NEPH1 proteins. Podocyte glycocalyx provides charge-selective filtration.
The glomerular filtration barrier = fenestrated endothelium + glomerular basement membrane (GBM, ~320 nm thick in humans) + slit diaphragm. The GBM is negatively charged, restricting anionic macromolecules such as albumin.
The corpuscle has a vascular pole (entry of afferent arteriole, exit of efferent arteriole) and a urinary pole (transition to proximal tubule).
2b. Juxtaglomerular Apparatus (JGA)
Located at the vascular pole, comprising:
- Macula densa — a plaque of specialized, columnar, closely packed cells in the thick ascending limb (TAL) at the vascular pole of the parent glomerulus. Detects NaCl concentration and signals to the JGA.
- Granular (juxtaglomerular) cells — modified smooth muscle cells in the wall of the afferent arteriole; contain renin granules; release renin in response to decreased stretch, sympathetic stimulation, or macula densa signals.
- Extraglomerular mesangial (Lacis/Goormaghtigh) cells — fill the space between the macula densa and the arterioles; coupled by gap junctions (connexin 40) to both the macula densa and the intraglomerular mesangium, enabling tubuloglomerular feedback.
2c. Proximal Tubule (~14 mm long in humans)
Begins at the urinary pole; located in the cortical labyrinth and medullary ray. Three morphologically distinct segments:
- S1 (pars convoluta, PCT) — tall cells with extensive brush border (microvilli ~1 μm long), abundant mitochondria in basal infoldings, prominent endocytic apparatus. The main site of reabsorption of glucose, amino acids, sodium, bicarbonate, and water.
- S2 — intermediate cell height; transitions from cortical labyrinth into the medullary ray.
- S3 (pars recta, PST) — extends through the outer stripe of the outer medulla; lower cells; still has a brush border but fewer mitochondria.
2d. Thin Limbs of the Loop of Henle
Arise from the pars recta; form the hairpin loop. Four epithelial types (I–IV) distinguish descending and ascending thin limbs. Short-loop nephrons (superficial cortical) have a short thin segment; long-loop nephrons (juxtamedullary) have long thin descending and ascending limbs extending deep into the inner medulla. Thin limbs are highly permeable to water (descending) and salt (ascending), enabling the countercurrent mechanism.
2e. Thick Ascending Limb (TAL)
Arises abruptly from the thin limbs; spans the inner and outer stripes of the outer medulla, enters the cortex, and contacts its own glomerulus at the macula densa. Cells are tall in the medullary TAL (mTAL) with extensive basolateral infoldings and abundant mitochondria, housing Na⁺-K⁺-2Cl⁻ (NKCC2) cotransporters. The TAL is impermeable to water, critical for generating the medullary hyperosmotic gradient. Also has the highest density of sympathetic nerve terminals of any nephron segment.
2f. Distal Tubule
- Distal convoluted tubule (DCT) — extends from the macula densa through the cortex; cells with extensive basolateral infoldings and abundant mitochondria; expresses NaCl cotransporter (NCC).
- Connecting tubule — transition segment leading to the collecting duct system.
2g. Collecting Duct System
- Cortical collecting duct (CCD) — two principal cell types:
- Principal cells (most abundant) — express ENaC (epithelial sodium channel) and aquaporin-2; aldosterone and ADH targets.
- Intercalated cells (type A and B) — regulate acid-base balance via proton-ATPase and anion exchangers (AE1).
- Outer medullary collecting duct (OMCD) — higher proportion of type A intercalated cells.
- Inner medullary collecting duct (IMCD) — the terminal portion, fusing into the ducts of Bellini that open at the area cribrosa. The terminal IMCD cells (IMCD cells) have a distinctive ultrastructure with no central cilium and a prominent glycocalyx.
3. Blood Vessels of the Kidney
The kidneys receive ~20–25% of cardiac output (~1.2–1.3 L/min at rest), despite comprising <0.5% of body weight.
3a. Arterial Supply
The hierarchy of renal arterial supply (Campbell-Walsh Urology):
Renal artery → Segmental artery → Interlobar artery → Arcuate artery → Interlobular artery → Afferent arteriole → Glomerulus → Efferent arteriole
- Renal artery — single (usually), arises from the abdominal aorta; enters at the hilum and divides into anterior and posterior branches.
- Segmental arteries — 5 in total (posterior, apical, upper, middle, lower). These are end arteries — no collateral circulation exists between them, so occlusion of one causes infarction of the corresponding segment.
- Interlobar arteries — travel through the cortical columns of Bertin between the renal pyramids.
- Arcuate arteries — run parallel to the kidney's surface along the corticomedullary junction; give rise to interlobular arteries.
- Interlobular (cortical radial) arteries — ascend radially through the cortex; provide afferent arterioles to each glomerulus.
- Afferent arteriole — high-resistance vessel; forms the glomerular capillary tuft.
- Efferent arteriole — exits the glomerulus; for superficial/cortical nephrons, forms the peritubular capillary network that supplies cortical tubules. For juxtamedullary nephrons, descends into the medulla as vasa recta (arteriolae rectae spuria) — thin-walled, parallel vessels forming hairpin loops alongside the loop of Henle; critical for the countercurrent exchange mechanism maintaining the medullary gradient.
An avascular plane (line of Brodel) lies just posterior to the lateral convex border between anterior and posterior segmental arteries — surgically important for minimizing hemorrhage.
Accessory renal arteries (arising separately from the aorta, usually to the lower pole) occur in 25–28% of individuals.
3b. Venous Outflow
The venous drainage mirrors the arterial supply but with a key difference: veins anastomose freely (unlike the segmental arteries), providing extensive collateral drainage.
The venous pathway (Campbell-Walsh Urology):
Peritubular/glomerular capillaries → Interlobular veins → Arcuate veins → Interlobar veins → Lobar veins → Segmental veins (3–5) → Renal vein → Inferior vena cava (IVC)
- The subcapsular stellate venous plexus connects interlobular veins to perinephric veins, providing an additional collateral route.
- The right renal vein is short (2–4 cm) and drains directly into the IVC.
- The left renal vein is longer (6–10 cm), runs anterior to the aorta, and receives the left suprarenal vein, left gonadal vein (testicular or ovarian), and often a lumbar vein.
- Because venous collaterals are rich, occlusion of a segmental venous branch has little effect on overall venous outflow.
4. Lymphatic Drainage
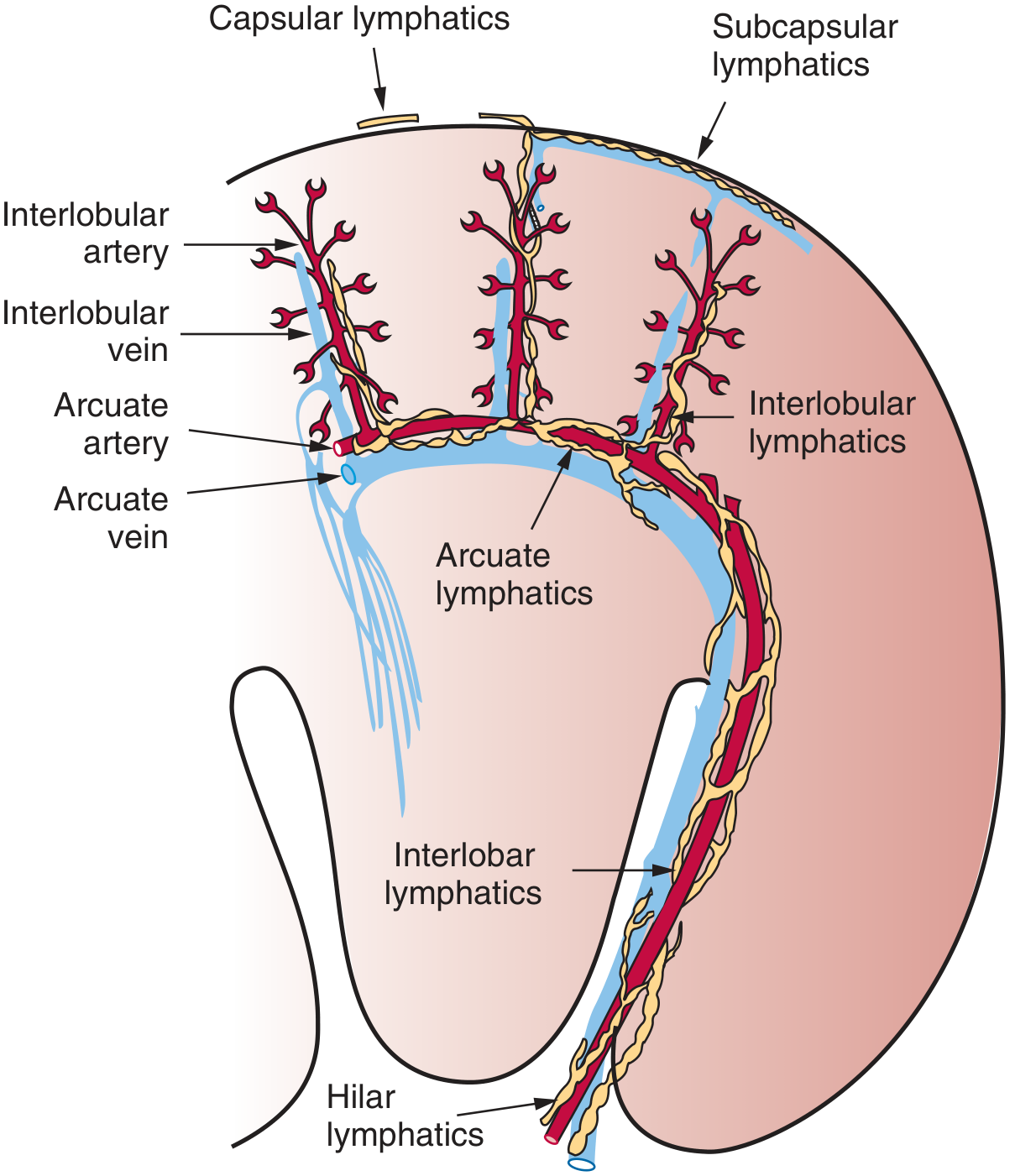
The renal lymphatic system has three components (Brenner & Rector's The Kidney):
4a. Intrarenal Lymphatics
- Lymphatic capillaries cluster primarily in the adventitia surrounding interlobular and arcuate arteries in the cortex.
- They coalesce into interlobular lymphatics → arcuate lymphatics (near the corticomedullary junction) → interlobar lymphatics → hilar lymphatic vessels.
- In humans and most mammals, lymphatic capillaries are rare among tubules and glomeruli; they are predominantly perivascular.
- The lymphatic volume density in the cortex is very small (0.02–0.37% depending on species).
4b. Subcapsular and Capsular Lymphatics
- A network of lymphatics lies in the space between the renal capsule and parenchyma (subcapsular).
- Capsular lymphatics can communicate with intrarenal lymphatics via communicating and perforating channels that transverse the capsule, often associated with interlobular arteries and veins.
4c. Hilar Lymphatic Outflow and Regional Nodes
As lymphatics exit the renal hilum, they join branches from the renal capsule, perinephric tissues, and renal pelvis, then drain to regional lymph nodes:
- Right kidney → right interaortocaval and right paracaval lymph nodes (between common iliac vessels and diaphragm).
- Left kidney → preaortic, paraaortic, and retroaortic lymph nodes. Posterior efferent vessels may drain to retroaortic nodes.
5. Innervation
5a. Sympathetic (Efferent) Innervation
The kidney receives exclusively sympathetic innervation — there are no parasympathetic fibers to the kidney (Ganong's; Medical Physiology).
- Preganglionic fibers originate from the intermediolateral column of the spinal cord between T6 (or T9) and L2 (or T13). Higher centers projecting to this region include the raphe nucleus, rostral ventrolateral medulla (vasomotor center), pontine A5 noradrenergic cell group, and paraventricular nucleus of the hypothalamus.
- Preganglionic fibers exit the cord between T11 and L3/L4 and synapse in both paravertebral and prevertebral ganglia — the latter including the aorticorenal, splanchnic, celiac, and mesenteric ganglia, as well as the renal plexus.
- Postganglionic fibers (vast majority unmyelinated, ~96–99%) form the renal plexus and travel along the renal artery into the kidney.
Distribution within the kidney:
- Fibers run with the renal arteries, lying within the perivascular interstitium, innervating smooth muscle of interlobar, arcuate, and interlobular arteries, and afferent and efferent arterioles (including JGA granular cells).
- Nerve terminals make contact with basement membranes of all tubule types, with the highest density in the TAL, followed by DCT, proximal tubule, and collecting duct.
- Nerve varicosities contain norepinephrine (dense-cored vesicles) and tyrosine hydroxylase (rate-limiting enzyme for norepinephrine synthesis).
Functional effects:
- Constriction of afferent arterioles → ↓ GFR, ↓ urine output.
- Constriction of efferent arterioles → ↑ filtration fraction.
- Stimulation of renin secretion from JGA granular cells.
- Modulation of tubular reabsorption (Na⁺ and water).
- Loss of sympathetic innervation → increased urinary output.
- Persistently elevated renal sympathetic nerve activity contributes to resistant hypertension; this is the basis for catheter-based renal denervation therapy.
Notably, transplanted kidneys (which are denervated) function normally, demonstrating that extrinsic innervation is not required for basic renal function.
5b. Afferent (Sensory) Innervation
Afferent fibers constitute a minority (~20–25%) of renal nerve fibers and are predominantly unmyelinated (~75–80%):
- They principally innervate the renal pelvis but also the interlobar, arcuate, and interlobular arteries, and afferent arterioles.
- Unlike efferent fibers (which express tyrosine hydroxylase), afferent fibers express calcitonin gene-related peptide (CGRP).
- Baroreceptors in interlobular arteries and afferent arterioles respond to increased perfusion pressure.
- Chemoreceptors in the renal pelvis respond to renal ischemia and abnormal interstitial ion composition (elevated K⁺ and H⁺).
- A renorenal reflex exists: increased ureteral pressure in one kidney decreases efferent sympathetic activity to the contralateral kidney, promoting natriuresis.
- Nociceptive afferents mediating pain travel in thoracic and upper lumbar dorsal roots.
Sources: Brenner and Rector's The Kidney, 11th Ed. | Campbell-Walsh-Wein Urology | Medical Physiology (Boron & Boulpaep) | Ganong's Review of Medical Physiology, 26th Ed. | Histology: A Text and Atlas (Ross & Pawlina)
I need in anatomy basis for my anatomy exam
Asking for Preferences
Here is a complete anatomy exam prep package on the kidney — detailed notes, bullet summaries, and exam Q&A all in one.
🫘 KIDNEY — Anatomy Exam Prep
PART 1 — DETAILED NOTES
Gross Anatomy: Position & External Features
- Paired retroperitoneal organs, T12–L3; right kidney is slightly lower (displaced by liver)
- Each weighs 125–170 g (male), 115–155 g (female); ~11–12 cm long
- Fibrous capsule covers the surface; perirenal fat surrounds it; all enclosed in Gerota's fascia
- Hilum (medial concave surface): entry/exit point for renal artery, renal vein, lymphatics, nerve plexus, and the ureter
- Renal sinus: space enclosed by parenchyma at the hilum; contains renal pelvis, major/minor calyces, vessels, fat
Internal Structure
| Layer | Key Contents |
|---|---|
| Cortex (~1 cm thick) | Glomeruli, proximal & distal convoluted tubules, cortical collecting ducts |
| Medulla | 8–18 renal pyramids; contains loops of Henle, collecting ducts, vasa recta |
| Columns of Bertin | Cortical tissue dipping between pyramids |
| Medullary rays | Straight tubule bundles projecting from medulla into cortex |
| Renal papilla | Apex of each pyramid; bears the area cribrosa (openings of ducts of Bellini) |
| Minor calyx | Cups around 1 papilla |
| Major calyx | 2–3 minor calyces → 1 major calyx |
| Renal pelvis | Unites 2–3 major calyces → narrows into ureter |
Renal lobe = 1 pyramid + overlying cortex + half of surrounding columns
The Nephron — Functional Unit (~1 million per kidney)
Two types by location:
| Feature | Cortical nephron | Juxtamedullary nephron |
|---|---|---|
| Glomerulus | Superficial cortex | Deep cortex near medulla |
| Loop of Henle | Short loop | Long loop (deep into inner medulla) |
| Efferent arteriole | → peritubular capillaries | → vasa recta |
| Function | Filtration | Urine concentration |
Nephron Segments (in order):
1. Renal Corpuscle (cortex only)
- Bowman's capsule: parietal (squamous) + visceral layer (podocytes)
- Glomerulus: ~40 capillary loops
- Filtration barrier: fenestrated endothelium → GBM → slit diaphragm (nephrin/podocin)
- Vascular pole: afferent arteriole in, efferent arteriole out
- Urinary pole: filtered fluid enters proximal tubule
2. Proximal Tubule (cortex; ~14 mm)
- PCT (pars convoluta) — in cortical labyrinth
- PST (pars recta) — descends into outer stripe of outer medulla
- Cells: tall columnar, brush border (microvilli), abundant mitochondria
- Reabsorbs ~65–70% of filtered Na⁺, water, glucose, amino acids, HCO₃⁻
3. Loop of Henle
- Thin descending limb — permeable to water; passes into medulla
- Thin ascending limb — permeable to Na⁺/Cl⁻; impermeable to water
- Thick ascending limb (TAL) — active NaCl reabsorption (NKCC2 cotransporter); impermeable to water → dilutes tubular fluid; macula densa is the terminal TAL
4. Distal Convoluted Tubule (DCT) (cortex)
- NaCl reabsorption (NCC cotransporter); fine-tuning of Ca²⁺ under PTH
5. Collecting Duct System
- Cortical → Outer medullary → Inner medullary collecting duct
- Principal cells: aldosterone → ↑ ENaC → Na⁺ reabsorption; ADH → ↑ AQP2 → water reabsorption
- Intercalated cells (type A & B): acid-base regulation (H⁺-ATPase, AE1)
- Terminal ducts = Ducts of Bellini → open at area cribrosa
Juxtaglomerular Apparatus (JGA) — EXAM FAVOURITE
| Component | Location | Function |
|---|---|---|
| Macula densa | TAL at vascular pole | Senses tubular NaCl; triggers tubuloglomerular feedback |
| Granular (JG) cells | Afferent arteriole wall | Produce and release renin |
| Extraglomerular mesangial cells (Lacis cells) | Between macula densa & arterioles | Signal transduction via gap junctions (connexin 40) |
Renin release triggers: ↓ BP (stretch), ↓ NaCl at macula densa, sympathetic stimulation (β₁)
PART 2 — BULLET SUMMARIES
Blood Vessels — Arterial Pathway
Renal artery → Segmental (5) → Interlobar → Arcuate → Interlobular → Afferent arteriole → Glomerulus → Efferent arteriole → Peritubular capillaries / Vasa recta
- Segmental arteries are END ARTERIES — no anastomoses; occlusion = segmental infarct
- Line of Brödel: avascular plane between anterior and posterior segments — surgical incision site
- Accessory renal arteries from aorta present in 25–28% of people
- Kidneys receive ~20–25% of cardiac output (~1.2 L/min)
Blood Vessels — Venous Pathway
Peritubular capillaries → Interlobular veins → Arcuate veins → Interlobar veins → Segmental veins (3–5) → Renal vein → IVC
- Veins freely anastomose (unlike arteries) → collateral drainage
- Subcapsular stellate venous plexus connects interlobular veins to perinephric veins
- Right renal vein: 2–4 cm, drains straight to IVC
- Left renal vein: 6–10 cm, receives left suprarenal + left gonadal vein (± lumbar vein)
Lymphatic Drainage
Interlobular lymphatics → Arcuate lymphatics → Interlobar lymphatics → Hilar lymphatics → Regional nodes
- Lymphatics follow the arteries (perivascular adventitia)
- Three components: capsular, subcapsular, intrarenal
- Right kidney → interaortocaval + paracaval nodes
- Left kidney → preaortic, paraaortic, retroaortic nodes
Innervation
- Sympathetic ONLY — no parasympathetic fibers
- Preganglionic: T6–L2 (spinal cord intermediolateral column)
- Synapse in celiac, aorticorenal, splanchnic, mesenteric ganglia + renal plexus
- Postganglionic fibers travel along the renal artery into the kidney
- Innervate: afferent + efferent arterioles, JGA granular cells, all tubule types (highest density in TAL)
- Neurotransmitter: norepinephrine (dense-cored vesicles at nerve varicosities)
- Effects: vasoconstriction, ↑ renin, ↑ tubular Na⁺ reabsorption
- Afferent (sensory) fibers: renal pelvis, arterioles → express CGRP; mediate pain (thoracic/lumbar dorsal roots), renorenal reflex
- Denervated transplanted kidneys function normally → extrinsic innervation not essential
PART 3 — EXAM Q&A
Q1: What are the boundaries of the renal sinus?
Renal parenchyma on all sides except where it opens at the hilum. It contains: renal pelvis, calyces, blood vessels, lymphatics, nerves, and fat.
Q2: What is the avascular plane of the kidney and its clinical significance?
The line of Brödel — lies just posterior to the lateral convex border between the anterior and posterior segmental arteries. Surgical incisions here (e.g., nephrolithotomy, percutaneous access) minimize hemorrhage.
Q3: Why does occlusion of a segmental renal artery cause infarction?
Segmental arteries are end arteries with no anastomoses between them. Occlusion has no collateral bypass → wedge-shaped infarct.
Q4: Why doesn't occlusion of a segmental renal vein cause venous infarction?
Renal veins freely anastomose via multiple collaterals (arcuate venous collars, stellate subcapsular plexus), so occlusion of one segmental vein has minimal effect.
Q5: Where does the left renal vein drain and what does it receive?
Drains into the IVC (left side, longer at 6–10 cm). Receives: left suprarenal vein, left gonadal vein (testicular/ovarian), and often a lumbar vein.
Q6: Trace the lymphatic drainage of the right kidney.
Perivascular intrarenal lymphatics → interlobular → arcuate → interlobar → hilar lymphatics → right interaortocaval and paracaval lymph nodes.
Q7: What is the full structural composition of the glomerular filtration barrier?
Three layers (inside out): 1) Fenestrated capillary endothelium (pores 70–100 nm); 2) Glomerular basement membrane (GBM, negatively charged, ~320 nm thick); 3) Slit diaphragm between podocyte foot processes (nephrin, podocin, NEPH1).
Q8: What are the three components of the JGA and their functions?
- Macula densa — NaCl sensor in TAL; 2) Granular cells in afferent arteriole — secrete renin; 3) Extraglomerular mesangial (Lacis) cells — signal transduction.
Q9: What is the innervation of the kidney — origin, pathway, targets, and neurotransmitter?
Origin: T6–L2 (intermediolateral column). Synapse: celiac/aorticorenal/mesenteric ganglia + renal plexus. Travel along the renal artery. Targets: afferent/efferent arterioles, JGA, all tubule types (TAL >> DCT > PT > CD). Neurotransmitter: norepinephrine. No parasympathetic supply.
Q10: What is the difference between cortical and juxtamedullary nephrons?
Cortical: superficial glomerulus, short loop of Henle, efferent → peritubular capillaries. Juxtamedullary: deep glomerulus, long loop extending into inner medulla, efferent → vasa recta → essential for urine concentration by countercurrent exchange.
Q11: What do principal cells vs. intercalated cells of the collecting duct do?
Principal cells: Na⁺ reabsorption via ENaC (aldosterone-regulated), water reabsorption via AQP2 (ADH-regulated). Intercalated cells: acid-base balance — type A secrete H⁺; type B secrete HCO₃⁻.
Q12: What happens to renal function after sympathetic denervation (e.g., transplant)?
Renal function remains normal — extrinsic sympathetic innervation is not required for basic kidney function. Loss of sympathetic input → mildly increased urinary output.
Good luck on your exam! Let me know if you want to go deeper on any specific section — glomerular histology, tubular physiology, or the collecting duct system, for example.
This is a shared conversation. Sign in to Orris to start your own chat.