Cystic fibrosis
Reading File
Asking for Preferences
Finding Sources
Searching PubMed
"cystic fibrosis" AND diagnosis
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
I now have comprehensive, well-sourced information. Here is a thorough overview of cystic fibrosis diagnosis and investigations.
Cystic Fibrosis: Diagnosis & Investigations
Diagnostic Criteria (CFF Consensus Guidelines)
A diagnosis of CF requires both of the following:
Criterion 1 — One or more of:
- Characteristic phenotypic features (sinopulmonary disease, GI/nutritional abnormalities, salt loss, male urogenital abnormalities)
- Family history of CF in a first-degree relative
- Positive newborn screening (NBS) result
Criterion 2 — Evidence of CFTR dysfunction via at least one of:
- Sweat chloride ≥60 mmol/L (diagnostic)
- Sweat chloride in the intermediate range (30–59 mmol/L) plus identification of two disease-causing CFTR mutations
- Sweat chloride in the intermediate range plus demonstration of abnormal nasal epithelial ion transport (nasal potential difference or intestinal current measurement)
(Robbins Pathologic Basis of Disease; CFF Guidelines, Farrell et al. J Pediatr 2017)
Diagnostic Algorithm
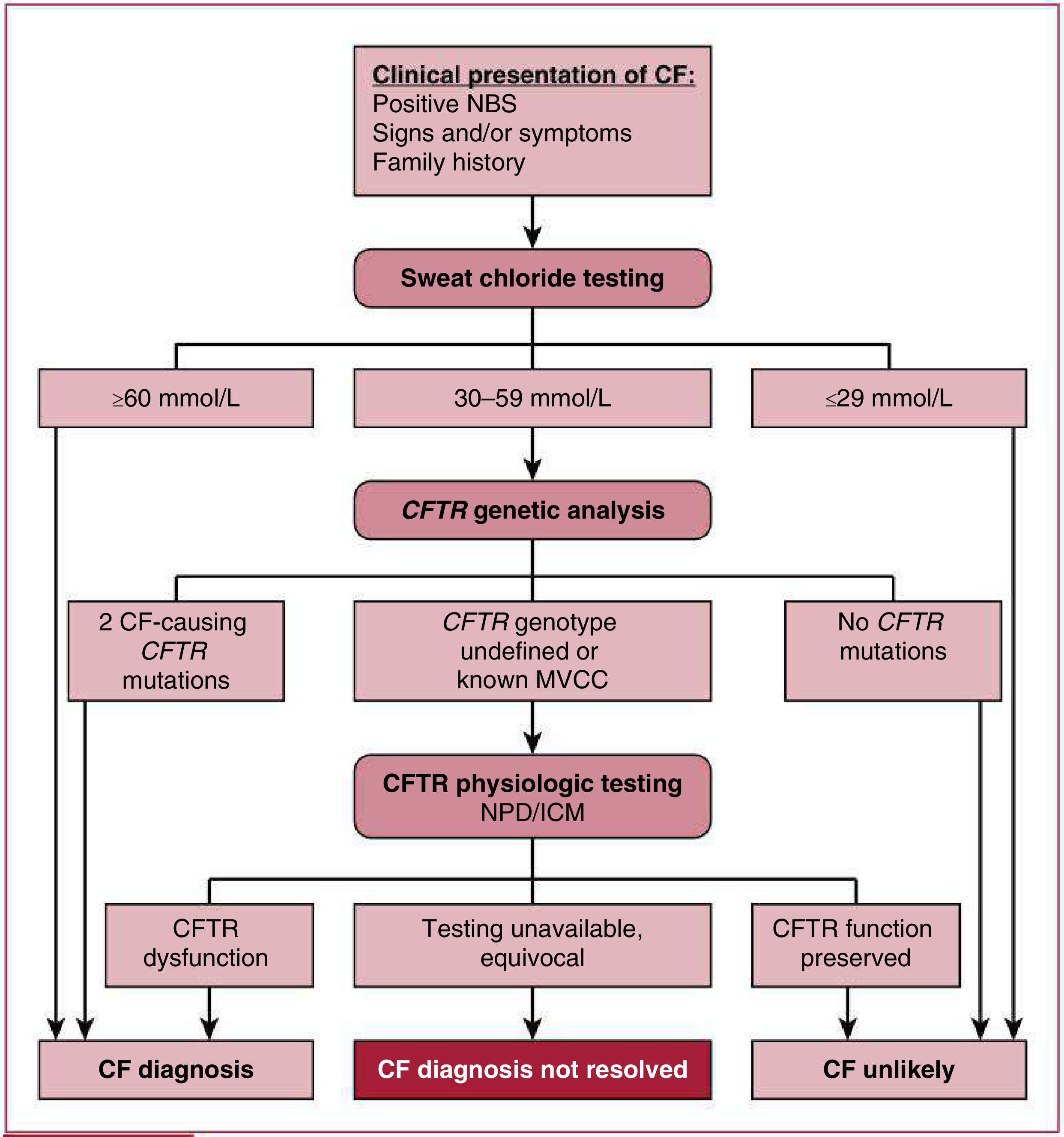
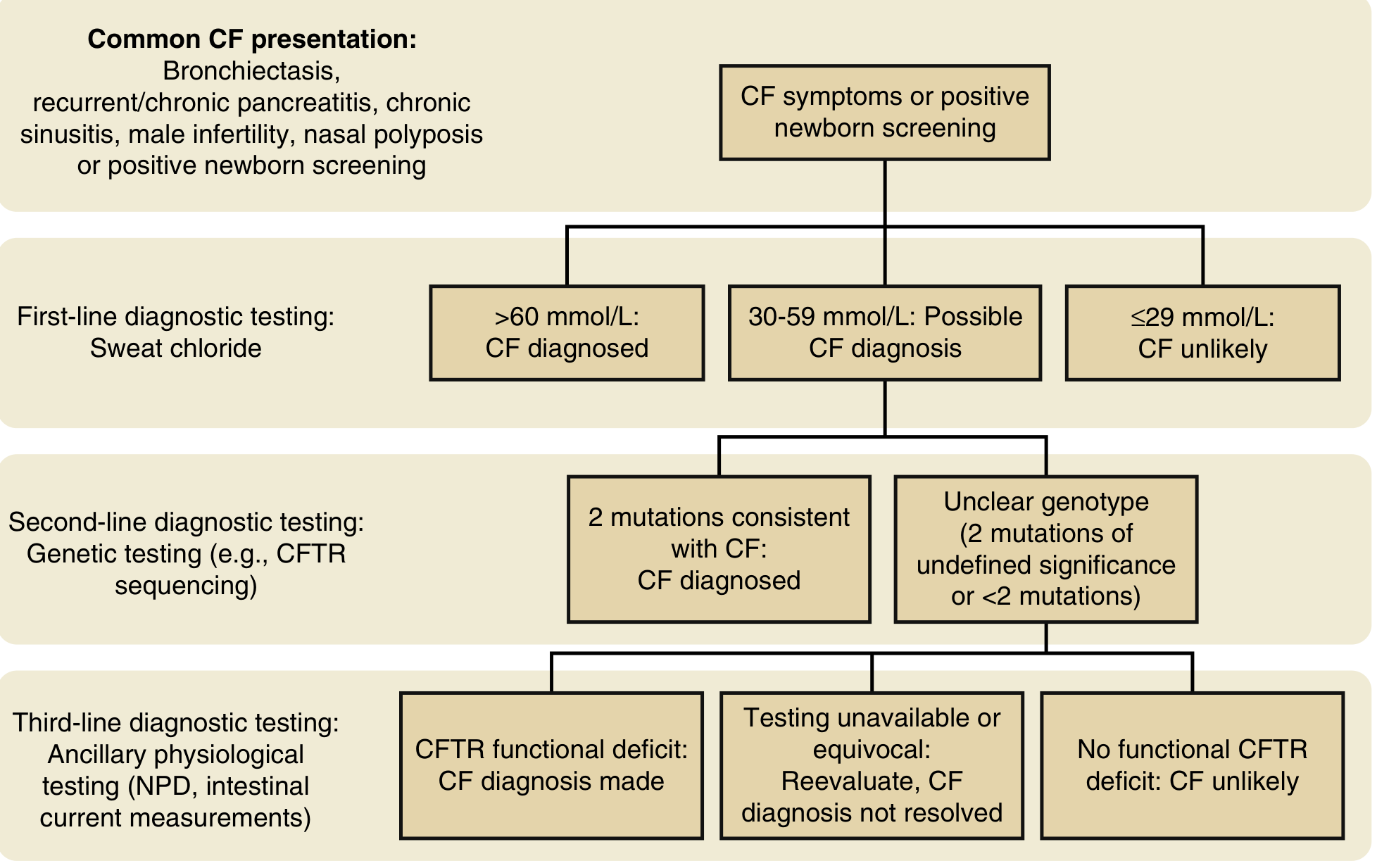
1. Newborn Screening (NBS)
- The most common NBS test measures immunoreactive trypsinogen (IRT) in blood — elevated because pancreatic injury in CF causes trypsinogen to leak into circulation.
- Over 60% of CF patients in the USA are now diagnosed via NBS (median age at diagnosis: 4 months).
- A positive NBS is presumptive only — must be confirmed with sweat chloride testing ± genetic analysis.
- Sleisenger & Fordtran's GI & Liver Disease
2. Sweat Chloride Testing — The Gold Standard
Performed by quantitative pilocarpine iontophoresis (QPIT):
| Sweat Cl⁻ (mmol/L) | Interpretation |
|---|---|
| ≥60 | CF diagnostic |
| 30–59 | Intermediate — further workup required |
| ≤29 | CF unlikely |
Key points:
- Updated CFF guidance lowered the intermediate range threshold from 40 to 30 mmol/L, improving detection of milder/atypical CF without increasing false positives.
- The test should be performed at a certified CF center using standardized methods — false positives and false negatives occur with malnutrition, certain medications, or inadequate sweat volume.
- Conditions other than CF that raise sweat electrolytes include ectodermal dysplasia, adrenal insufficiency, glycogen storage disease type 1, familial hypoparathyroidism, and mucopolysaccharidoses.
- Harrison's Principles of Internal Medicine 22E; Sleisenger & Fordtran's
Indications for sweat testing include: siblings of CF patients, chronic pulmonary symptoms, bronchiectasis, recurrent respiratory infections, failure to thrive, nasal polyposis, meconium ileus, rectal prolapse, jaundice in early infancy, cirrhosis in childhood, male aspermia/azoospermia, and heat stroke.
3. CFTR Genetic Analysis (Second-Line)
- Triggered by an intermediate sweat chloride (30–59 mmol/L) or when confirmation is needed.
- Full CFTR sequencing (or high-sensitivity panel) is preferred — standard panels testing <40 mutations are insufficient for atypical/mild cases, as they are biased toward common Caucasian mutations and will miss rare partial-function variants.
- Two CF-causing mutations = CF confirmed.
- One or zero mutations with intermediate sweat Cl⁻ → proceed to third-line physiologic testing.
- Genotype information is catalogued in the CFTR-2 repository (cftr2.org), useful for predicting phenotypic severity and guiding mutation-specific therapy.
- Murray & Nadel's Textbook of Respiratory Medicine; Thompson & Thompson Genetics 9e
4. CFTR Physiologic Testing (Third-Line)
Used when sweat Cl⁻ is intermediate and genetic testing is unclear/insufficient:
| Test | Principle | Availability |
|---|---|---|
| Nasal potential difference (NPD) | Measures transepithelial ion transport via nasal epithelium; abnormal in CF | Many CF research centers |
| Intestinal current measurement (ICM) | Measures CFTR activity in rectal mucosal biopsy | Very limited; few centers worldwide |
These tests reflect CFTR functional deficits along a continuum and can distinguish mild from severe phenotypes even beyond genotype information alone.
5. Ancillary Investigations
| Test | Role |
|---|---|
| Fecal elastase-1 | Supports diagnosis by indicating pancreatic exocrine insufficiency; low in pancreatic-insufficient CF |
| Chest X-ray / CT chest | Hyperinflation, bronchiectasis, mucus plugging, air trapping |
| Pulmonary function tests (spirometry) | Obstructive pattern (↓FEV₁, ↓FEV₁/FVC); tracks disease progression |
| Sputum culture | Identifies organisms — S. aureus (early), P. aeruginosa (chronic), B. cepacia (poor prognosis) |
| Abdominal imaging / MRCP | Assesses hepatobiliary disease, pancreatic changes |
| Semen analysis | Obstructive azoospermia in ~95% of males with CF (CBAVD) |
| Liver function tests | Monitor for CF-related liver disease (onset typically around puberty) |
| OGTT / HbA1c | Screen for CF-related diabetes (up to 50% of adults) |
CFTR-Related Spectrum
CF diagnosis exists on a continuum:
- Classic CF (pancreatic-insufficient): most severe; biallelic severe CFTR mutations; sweat Cl⁻ ≥60
- Atypical/nonclassic CF: milder; often one severe + one mild mutation; may have normal/borderline sweat Cl⁻
- CFTR-related metabolic syndrome (CRMS) / CFSPD: positive NBS + intermediate sweat Cl⁻ + <2 CF-causing mutations; not CF but at risk
- CFTR-related disorders (CFTR-RD): isolated manifestations (CBAVD, idiopathic pancreatitis, bronchiectasis) in patients who don't meet full CF criteria but carry CFTR mutations
Important Caveats
- <2% of patients with classic CF phenotype have normal sweat chloride — molecular CFTR analysis is essential in these cases.
- Some patients exhibit CF-like disease without identifiable CFTR mutations — may be due to mutations in the epithelial sodium channel (ENaC) gene or non-coding CFTR regions.
- Rare CFTR mutations not on standard panels are enriched in patients with atypical/adult-onset CF — always use full sequencing in this population.
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease; Harrison's Principles of Internal Medicine 22E; Murray & Nadel's Textbook of Respiratory Medicine; Sleisenger & Fordtran's GI & Liver Disease; Thompson & Thompson Genetics & Genomics in Medicine 9e; Harriet Lane Handbook 23e
This is a shared conversation. Sign in to Orris to start your own chat.