Classify the anti-retroviral drugs and discuss their mechanisms of action in details
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Searching PubMed
antiretroviral therapy HIV classification mechanisms
I have all the information needed to write a comprehensive answer. Let me compile it now.
Classification and Mechanisms of Action of Antiretroviral Drugs
Antiretroviral therapy (ART) targets the HIV replication cycle at multiple steps. There are five major classes of antiretroviral drugs, plus pharmacokinetic boosters.
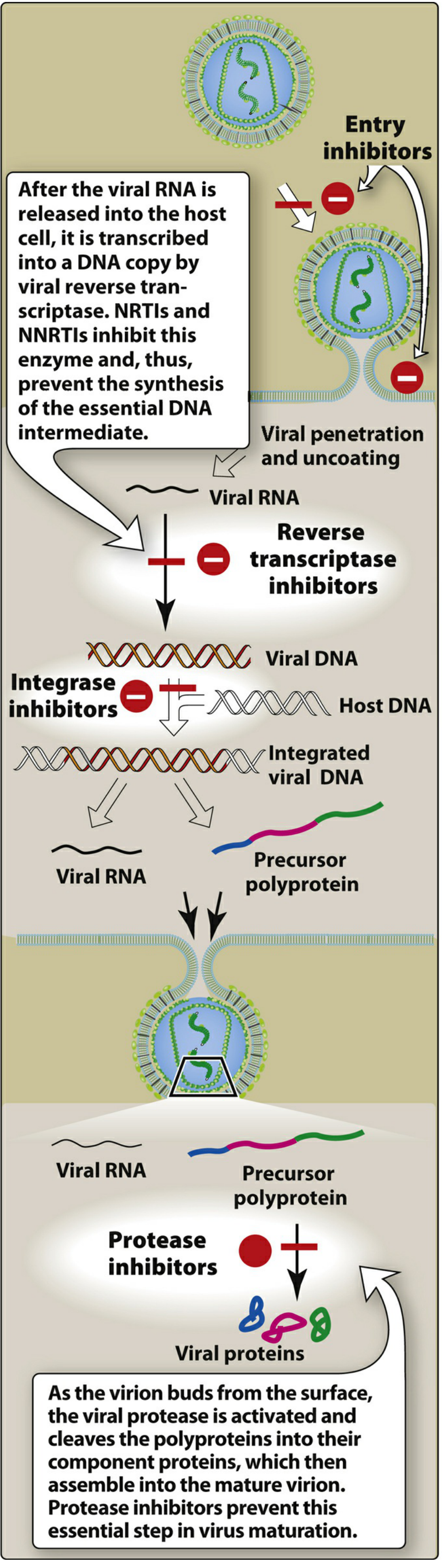
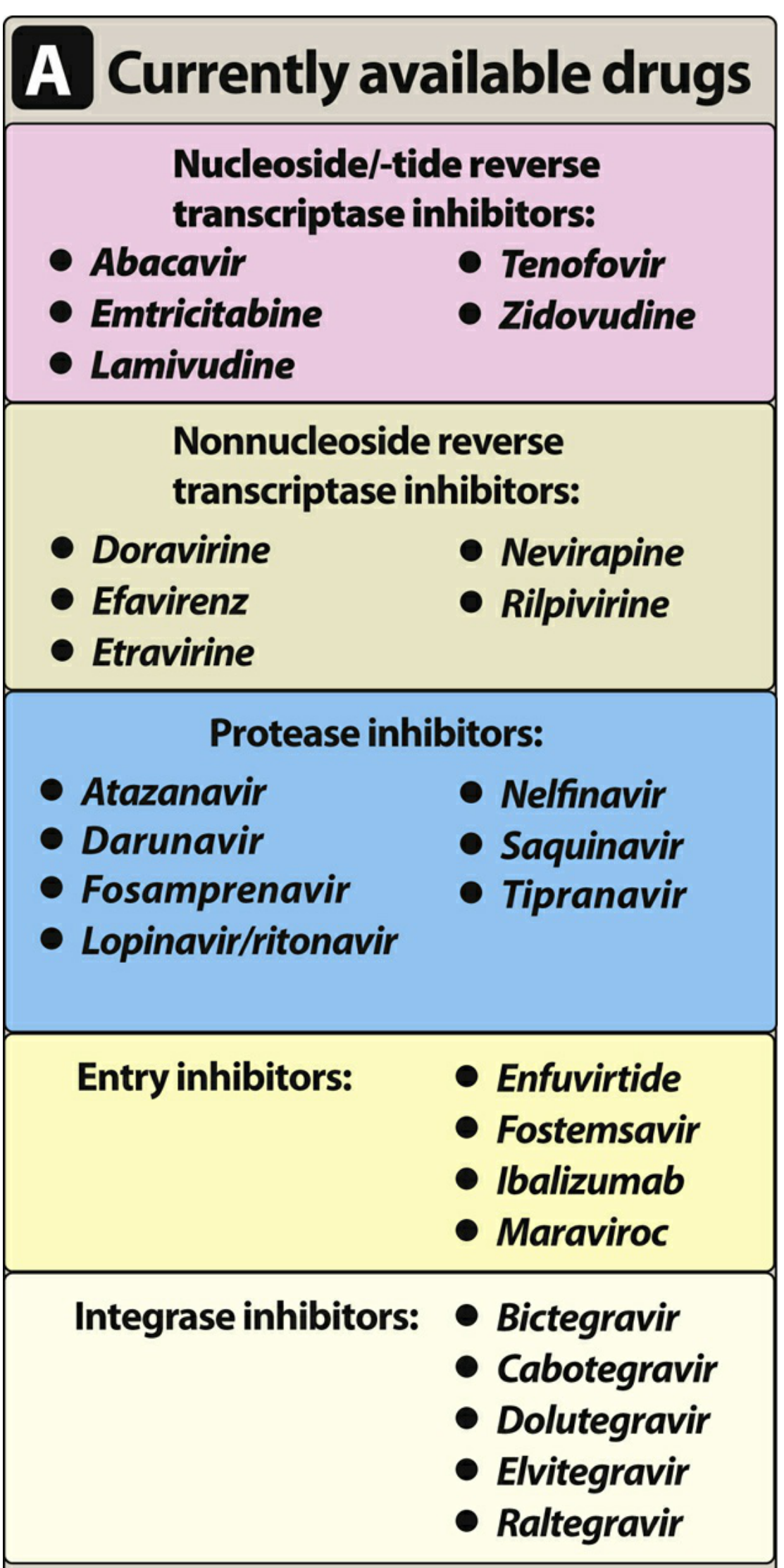
CLASS 1 — Nucleoside/Nucleotide Reverse Transcriptase Inhibitors (NRTIs)
Drugs: Zidovudine (AZT/ZDV), Lamivudine (3TC), Emtricitabine (FTC), Abacavir (ABC), Tenofovir disoproxil fumarate (TDF), Tenofovir alafenamide (TAF), Didanosine (ddI)
Mechanism of Action
NRTIs were the first agents developed to treat HIV. They are analogs of natural nucleosides/nucleotides but all lack a 3′-hydroxyl (–OH) group on the ribose sugar.
Step-by-step:
- NRTIs enter host cells and are phosphorylated by cellular kinases to their active triphosphate form.
- HIV reverse transcriptase (RT) preferentially incorporates the triphosphate NRTI analog into the growing viral DNA chain in place of the natural nucleotide.
- Because the 3′-OH group is absent, the phosphodiester bond required to add the next nucleotide cannot form → obligate chain termination.
- Viral DNA synthesis halts — preventing formation of double-stranded proviral DNA.
"Because the 3′-hydroxyl group is not present, a 3′,5′-phosphodiester bond between an incoming nucleoside triphosphate and the growing DNA chain cannot be formed, and DNA chain elongation is terminated." — Lippincott Illustrated Reviews: Pharmacology
Tenofovir specifics: TDF and TAF are both prodrugs of tenofovir. TAF achieves a 5–7-fold higher intracellular drug concentration in lymphoid cells with lower plasma levels, resulting in less renal toxicity and less bone mineral density loss than TDF.
Key toxicities:
- Class effect: lactic acidosis and hepatomegaly with steatosis (mitochondrial DNA polymerase γ inhibition)
- Abacavir: HLA-B*5701-associated hypersensitivity reaction (~5% of patients) — never rechallenge
- Zidovudine: bone marrow suppression, macrocytic anemia
- Didanosine (rarely used): peripheral neuropathy, pancreatitis, lipoatrophy
- TDF: nephrotoxicity, reduced bone mineral density
CLASS 2 — Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
Drugs: Efavirenz (EFV), Nevirapine (NVP), Rilpivirine (RPV), Etravirine (ETR), Doravirine (DOR)
Mechanism of Action
NNRTIs act on reverse transcriptase but via a completely different mechanism from NRTIs — they are non-competitive allosteric inhibitors:
- NNRTIs bind directly to a hydrophobic pocket on the RT enzyme near (but distinct from) the active catalytic site — they do not require intracellular phosphorylation.
- Binding causes a conformational change in the RT enzyme, reducing its catalytic activity.
- The result is impaired synthesis of viral DNA from the RNA template.
Key distinctions from NRTIs:
- Do not need activation (not prodrugs)
- Do not compete with natural nucleotides
- Highly specific for HIV-1 RT (not active against HIV-2 — clinically important)
- Single mutations (e.g., K103N for efavirenz/nevirapine) can confer high-level resistance; etravirine and doravirine have a higher genetic barrier
Key toxicities:
- Efavirenz: CNS effects (vivid dreams, dizziness, depression), teratogenic (avoid in first trimester)
- Nevirapine: hepatotoxicity (especially in women with high CD4 counts), severe rash/Stevens-Johnson syndrome
- Rilpivirine: requires food and adequate gastric acid (avoid with PPIs); less CNS toxicity than efavirenz
- All NNRTIs: rash, CYP3A4 induction/inhibition (major drug interactions)
CLASS 3 — Protease Inhibitors (PIs)
Drugs: Atazanavir (ATV), Darunavir (DRV), Lopinavir (LPV, always boosted with ritonavir), Ritonavir (RTV, used as booster), Saquinavir, Indinavir, Nelfinavir, Fosamprenavir, Tipranavir
Mechanism of Action
PIs act at a late stage of the HIV replication cycle — after viral DNA integration and gene transcription:
- After the integrated provirus is transcribed and translated, HIV produces a long polyprotein precursor (Gag-Pol polyprotein).
- The viral protease enzyme must cleave this polyprotein into individual functional proteins (structural proteins + enzymes) necessary for virion maturation.
- PIs are peptidomimetic compounds that competitively bind the active site of HIV protease, blocking cleavage of the polyprotein.
- The result is release of non-infectious, immature viral particles that cannot establish new infections.
"As the virion buds from the surface, the viral protease is activated and cleaves the polyproteins into their component proteins, which then assemble into the mature virion. Protease inhibitors prevent this essential step in virus maturation." — Lippincott Illustrated Reviews: Pharmacology
Pharmacokinetic boosting: Ritonavir (RTV) and cobicistat are CYP3A4 inhibitors used at sub-therapeutic doses to boost plasma levels of other PIs, allowing lower doses and once-daily dosing. This is why lopinavir is always co-formulated as LPV/r (Kaletra).
Key toxicities:
- Metabolic syndrome: dyslipidemia, insulin resistance, lipodystrophy (fat redistribution)
- Atazanavir: indirect hyperbilirubinemia (benign jaundice, inhibits UGT1A1), nephrolithiasis
- Indinavir: nephrolithiasis, requires high fluid intake
- Class effect: CYP3A4 inhibition → extensive drug interactions
- Tipranavir: hepatotoxicity, rare intracranial hemorrhage
CLASS 4 — Entry Inhibitors
Entry inhibitors block HIV from entering the host CD4+ T cell. This class has three distinct sub-mechanisms:
4a. Fusion Inhibitors
Drug: Enfuvirtide (ENF, T-20)
Mechanism:
- HIV envelope glycoprotein gp120 binds to CD4 on the host cell → conformational changes expose gp41 (transmembrane glycoprotein).
- gp41 undergoes a "hairpin" conformational change, bringing viral and host cell membranes together to fuse.
- Enfuvirtide is a synthetic 36-amino-acid peptide that binds to gp41, blocking the conformational change required for membrane fusion → viral RNA cannot enter the cell.
- Must be given subcutaneously (peptide, not orally bioavailable); injection-site reactions occur in nearly all patients.
- Reserved for treatment-experienced patients with virologic failure.
4b. CCR5 Antagonists (Co-receptor Antagonists)
Drug: Maraviroc (MVC)
Mechanism:
- HIV uses either CCR5 (R5-tropic strains) or CXCR4 (X4-tropic strains) as a co-receptor alongside CD4 for entry.
- Maraviroc binds CCR5 on the host cell, causing an allosteric conformational change that prevents gp120 from binding the co-receptor → entry blocked.
- Requires tropism testing before use (Trofile assay) — effective only against R5-tropic virus; useless against X4-tropic or dual/mixed-tropic strains.
- Generally well tolerated; hepatotoxicity risk; possible cardiovascular effects.
4c. CD4-Directed Attachment Inhibitors
Drug: Fostemsavir (FTR), Ibalizumab (IBA)
Mechanism:
- Fostemsavir is an attachment inhibitor (prodrug of temsavir) that binds HIV gp120 directly, preventing it from attaching to CD4 — blocking the very first step of viral entry.
- Ibalizumab is a humanized monoclonal antibody that binds to domain 2 of CD4, blocking post-attachment conformational changes needed for co-receptor engagement — given IV every 2 weeks; reserved for heavily treatment-experienced patients with multi-drug resistant HIV.
CLASS 5 — Integrase Strand Transfer Inhibitors (INSTIs)
Drugs: Raltegravir (RAL), Elvitegravir (EVG), Dolutegravir (DTG), Bictegravir (BIC), Cabotegravir (CAB)
Mechanism of Action
INSTIs target the HIV integrase enzyme, which is responsible for inserting the viral double-stranded DNA into the host cell genome:
- Integrase first performs 3′-processing: removes two nucleotides from each end of the viral DNA, creating reactive 3′-OH ends.
- Integrase then catalyzes strand transfer: the processed viral DNA ends attack the host chromosomal DNA, inserting the provirus.
- INSTIs bind the active site of integrase by chelating the two divalent Mg²⁺ metal cations at the catalytic core — this occupies the active site and blocks the strand transfer step specifically.
- Proviral integration is halted → no permanent infection of the cell.
"The active site of the integrase enzyme binds to the host cell DNA and includes two divalent metal cations that serve as chelation targets for the INSTIs." — Lippincott Illustrated Reviews: Pharmacology
Long-acting injectable forms: Cabotegravir IM + rilpivirine IM (given monthly or every 2 months) — first complete long-acting ART regimen. Cabotegravir alone is also approved for HIV pre-exposure prophylaxis (PrEP) as bimonthly injection.
Drug interactions: Oral INSTIs have chelation interactions with antacids, multivitamins, and iron/calcium supplements (significantly reduce absorption) — must be separated by several hours.
Resistance: Raltegravir and elvitegravir share cross-resistance mutations; dolutegravir and bictegravir have a high genetic barrier to resistance and limited cross-resistance with first-generation INSTIs.
Key toxicities: Generally well tolerated — nausea, diarrhea; dolutegravir: modest serum creatinine elevation (inhibits tubular creatinine secretion, not actual GFR change), neural tube defect risk (peri-conception).
PHARMACOKINETIC BOOSTERS (Not Antiretroviral Drugs per se)
| Agent | Mechanism | Use |
|---|---|---|
| Ritonavir (RTV) | Potent CYP3A4 inhibitor | Boosts atazanavir, darunavir, lopinavir, others |
| Cobicistat (COBI) | CYP3A4 inhibitor (no anti-HIV activity) | Boosts elvitegravir, atazanavir, darunavir |
Both agents lack intrinsic anti-HIV activity at boosting doses but raise co-administered drug AUC significantly, enabling lower doses and once-daily administration.
Summary Classification Table
| Class | Abbreviation | Key Drugs | Target | Stage of Cycle Blocked |
|---|---|---|---|---|
| Nucleoside/tide RT inhibitors | NRTIs | Tenofovir, Emtricitabine, Abacavir, Lamivudine, Zidovudine | Reverse transcriptase (competitive, chain termination) | Reverse transcription |
| Non-nucleoside RT inhibitors | NNRTIs | Efavirenz, Rilpivirine, Doravirine, Nevirapine, Etravirine | Reverse transcriptase (allosteric) | Reverse transcription |
| Protease inhibitors | PIs | Darunavir, Atazanavir, Lopinavir/r | Viral protease | Polyprotein cleavage (post-budding) |
| Fusion inhibitors | FIs | Enfuvirtide | gp41 conformational change | Entry (membrane fusion) |
| CCR5 antagonists | — | Maraviroc | CCR5 co-receptor on host cell | Entry (co-receptor binding) |
| Attachment inhibitors | — | Fostemsavir, Ibalizumab | gp120 / CD4 receptor | Entry (attachment) |
| Integrase strand transfer inhibitors | INSTIs | Dolutegravir, Bictegravir, Cabotegravir, Raltegravir | Integrase enzyme | Integration |
| Capsid inhibitors | — | Lenacapavir | HIV-1 capsid protein | Multiple steps |
Standard Initial Regimen (per Harrison's 22e, 2025)
The preferred backbone for most ART-naïve patients is:
- Two NRTIs (tenofovir-based [TDF or TAF] + emtricitabine or lamivudine) PLUS
- An INSTI (dolutegravir or bictegravir preferred) OR
- A boosted PI (darunavir/r or /c) OR
- An NNRTI (rilpivirine or doravirine)
Two-drug regimens (dolutegravir + lamivudine) are also supported for selected patients.
Sources: Lippincott Illustrated Reviews: Pharmacology, 8th ed. | Harrison's Principles of Internal Medicine, 22e (2025) | Goldman-Cecil Medicine, International Edition
For mbbs 2nd yr refer kdt
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
I have all the content needed. Here is the complete answer structured in the style and depth appropriate for MBBS 2nd year, drawing from Katzung's Basic & Clinical Pharmacology and Goodman & Gilman (the international equivalents of KDT in our library — KDT itself is not in the digital collection, but these cover the same syllabus content at the same depth).
Note: KD Tripathi's Essentials of Medical Pharmacology is not available in this library's digital collection. The following answer is compiled from Katzung's Basic & Clinical Pharmacology (16th ed.) and Goodman & Gilman's Pharmacological Basis of Therapeutics — both cover antiretrovirals at exactly the same depth and format as KDT for MBBS 2nd year.
Antiretroviral Drugs — Classification & Mechanism of Action
As per Katzung / Goodman & Gilman (MBBS 2nd Year)
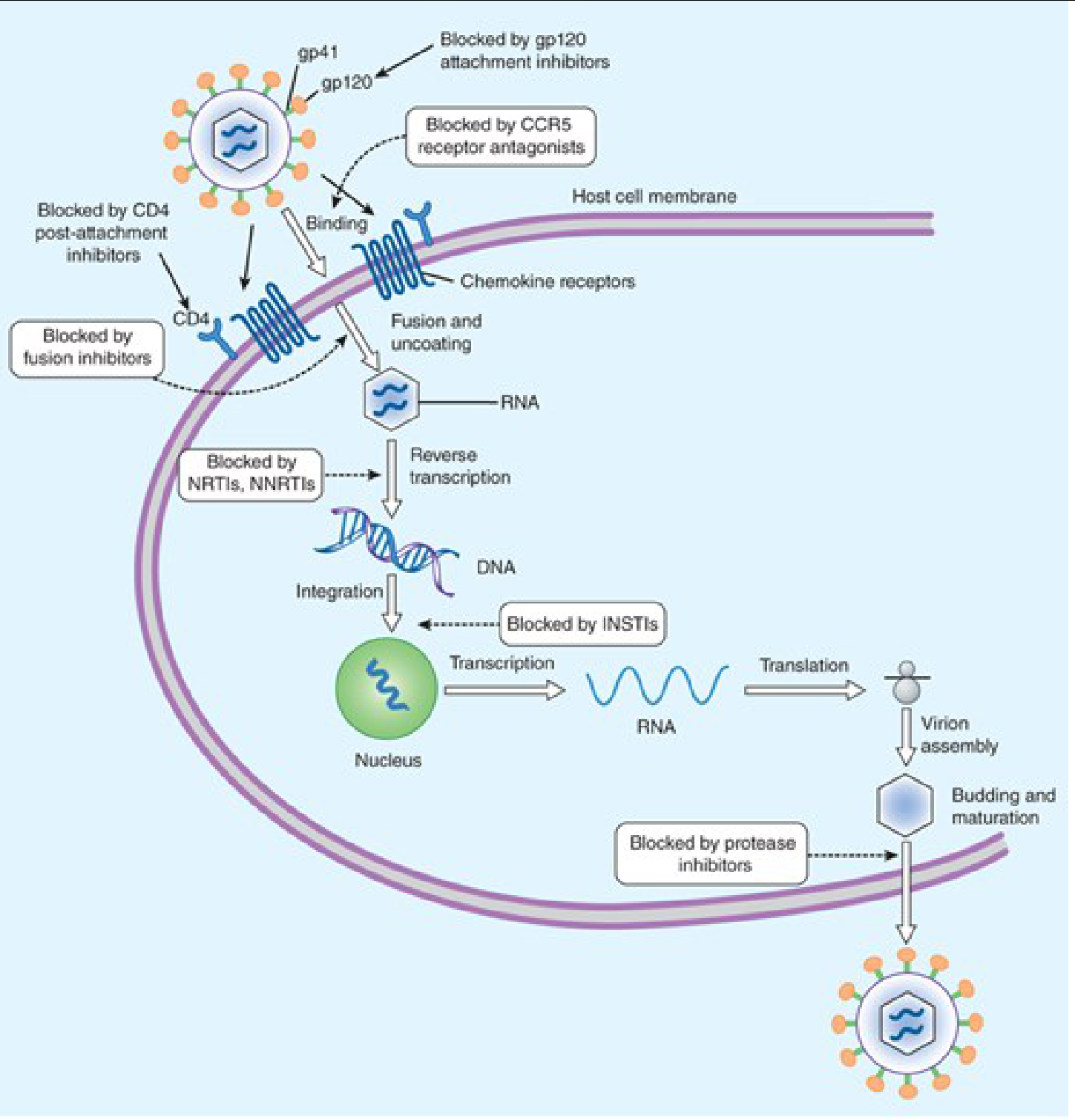
CLASSIFICATION
Antiretroviral drugs are classified based on which step of the HIV replication cycle they inhibit:
| # | Class | Abbreviation | Examples |
|---|---|---|---|
| 1 | Nucleoside/Nucleotide Reverse Transcriptase Inhibitors | NRTIs | Zidovudine, Lamivudine, Tenofovir, Abacavir, Emtricitabine |
| 2 | Non-Nucleoside Reverse Transcriptase Inhibitors | NNRTIs | Nevirapine, Efavirenz, Rilpivirine, Etravirine, Doravirine |
| 3 | Protease Inhibitors | PIs | Atazanavir, Darunavir, Lopinavir/r, Ritonavir, Saquinavir |
| 4 | Integrase Strand Transfer Inhibitors | INSTIs | Raltegravir, Dolutegravir, Bictegravir, Elvitegravir, Cabotegravir |
| 5 | Entry Inhibitors | — | Enfuvirtide (fusion), Maraviroc (CCR5), Fostemsavir (attachment), Ibalizumab |
| 6 | Capsid Inhibitor | — | Lenacapavir |
| — | Pharmacokinetic Boosters | — | Ritonavir (low dose), Cobicistat |
1. NUCLEOSIDE/NUCLEOTIDE REVERSE TRANSCRIPTASE INHIBITORS (NRTIs)
Drugs: Zidovudine (AZT), Lamivudine (3TC), Emtricitabine (FTC), Abacavir (ABC), Tenofovir (TDF/TAF), Didanosine (ddI — rarely used now)
Mechanism of Action
- NRTIs are structural analogs of natural nucleosides (building blocks of DNA) but with one critical difference: they lack the 3′-hydroxyl (–OH) group on the ribose sugar.
- After entering the cell, they are phosphorylated by host cell kinases to their active triphosphate form.
- HIV reverse transcriptase (RT) incorporates the triphosphate NRTI into the growing viral DNA chain in preference to the natural nucleotide.
- Because there is no 3′-OH group, the next nucleotide cannot be added → obligate chain termination → viral DNA synthesis stops.
- NRTIs have a lower affinity for host DNA polymerases than for HIV RT, giving selectivity. However, mitochondrial DNA polymerase γ is susceptible → basis of mitochondrial toxicities.
"Phosphorylated to active form to prevent infection of susceptible cells; do not eradicate virus from cells with integrated proviral DNA; active against HIV-1, HIV-2, and in some cases HBV." — Goodman & Gilman
Tenofovir (special note): Available as two prodrugs:
- TDF (tenofovir disoproxil fumarate) — older, more renal/bone toxicity
- TAF (tenofovir alafenamide) — newer, achieves 5–7× higher intracellular levels at lower plasma concentrations → safer kidneys and bones
Key Toxicities
| Drug | Important ADR |
|---|---|
| Zidovudine | Bone marrow suppression (anemia, neutropenia), myopathy |
| Abacavir | Hypersensitivity syndrome (HLA-B*5701 — must screen; never rechallenge — potentially fatal) |
| Tenofovir (TDF) | Nephrotoxicity, Fanconi syndrome, decreased bone mineral density |
| Didanosine | Peripheral neuropathy, pancreatitis |
| All NRTIs (class) | Lactic acidosis + hepatic steatosis (mitochondrial toxicity) |
2. NON-NUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITORS (NNRTIs)
Drugs: Nevirapine (NVP), Efavirenz (EFV), Rilpivirine (RPV), Etravirine (ETR), Doravirine (DOR)
Mechanism of Action
- NNRTIs do NOT need intracellular phosphorylation (unlike NRTIs).
- They bind directly and non-competitively to a hydrophobic allosteric pocket on HIV-1 reverse transcriptase (near but distinct from the active catalytic site).
- This binding causes a conformational change in the enzyme → reduces RT's catalytic activity → viral RNA cannot be reverse-transcribed into DNA.
- Important distinction: NNRTIs are HIV-1 specific — they are NOT active against HIV-2 (which lacks the corresponding binding pocket). This is a key exam point.
- They do not compete with natural nucleotides.
Key Toxicities
| Drug | Important ADR |
|---|---|
| Nevirapine | Rash (common, usually resolves); rarely Stevens-Johnson syndrome; potentially fatal hepatitis (especially in women with CD4 >250 or men with CD4 >400) |
| Efavirenz | CNS toxicity (vivid dreams, dizziness, confusion — usually resolves in 2–4 weeks); teratogenic (avoid in first trimester) |
| Rilpivirine | Must be taken with food; avoid PPIs (need acidic pH); QTc prolongation risk |
| All NNRTIs | CYP3A4 inducers/inhibitors → extensive drug interactions; rash |
Resistance: A single mutation (e.g., K103N) causes high-level resistance to nevirapine and efavirenz — low genetic barrier. Etravirine has a higher genetic barrier (needs multiple mutations).
3. PROTEASE INHIBITORS (PIs)
Drugs: Atazanavir (ATV), Darunavir (DRV), Lopinavir/ritonavir (LPV/r), Saquinavir, Indinavir, Nelfinavir, Tipranavir, Ritonavir (used as booster)
Mechanism of Action
- PIs act at a late post-transcriptional stage of the HIV life cycle.
- HIV first transcribes and translates its genome into a long Gag-Pol polyprotein precursor — a non-functional, immature protein chain.
- The viral protease enzyme must cleave this polyprotein into individual structural proteins and enzymes (e.g., reverse transcriptase, integrase, capsid) → required for virion maturation.
- PIs are peptidomimetic compounds that competitively bind the active site of HIV protease → block polyprotein cleavage → immature, non-infectious viral particles are released.
- Result: virions bud but cannot infect new cells.
Pharmacokinetic Boosting
- Most PIs are metabolized by CYP3A4.
- Ritonavir (at low/booster dose) and cobicistat are potent CYP3A4 inhibitors used to raise plasma levels of co-administered PIs → less frequent dosing, more sustained levels.
- e.g., Lopinavir is always given as LPV/r (+ ritonavir 100 mg); Darunavir + ritonavir (DRV/r) or darunavir + cobicistat.
Key Toxicities
| Toxicity | Drug(s) |
|---|---|
| Metabolic syndrome (dyslipidemia, insulin resistance, lipodystrophy) | Class effect |
| Indirect hyperbilirubinemia / jaundice (UGT1A1 inhibition) | Atazanavir (benign, reversible) |
| Nephrolithiasis | Atazanavir, Indinavir (must maintain high fluid intake) |
| GI intolerance (nausea, diarrhea) | Class effect |
| Hepatotoxicity | Tipranavir, Ritonavir |
| Extensive drug interactions (CYP3A4 inhibition) | All PIs |
4. INTEGRASE STRAND TRANSFER INHIBITORS (INSTIs)
Drugs: Raltegravir (RAL), Elvitegravir (EVG), Dolutegravir (DTG), Bictegravir (BIC), Cabotegravir (CAB)
Mechanism of Action
- After reverse transcription, HIV double-stranded DNA must be inserted into the host cell's chromosomal DNA — a step catalyzed by viral integrase.
- Integrase carries two Mg²⁺ metal ions at its active site, essential for catalytic activity.
- INSTIs chelate these divalent metal cations at the integrase active site → block the strand transfer step (insertion of viral DNA into host DNA) → no provirus formation → no permanent infection.
- The cell is not permanently infected, and viral gene expression cannot occur.
Long-acting INSTIs:
- Cabotegravir IM + rilpivirine IM = first long-acting complete ART regimen (given monthly or every 2 months)
- Cabotegravir IM alone = approved for PrEP (pre-exposure prophylaxis)
Key Toxicities
- Generally well tolerated — nausea, diarrhea most common
- Chelation interaction with antacids, iron, calcium, magnesium supplements (reduce oral INSTI absorption significantly — separate doses)
- Dolutegravir: mild creatinine elevation (inhibits tubular secretion, not true nephrotoxicity); neural tube defect risk around conception
- Elvitegravir: requires cobicistat boosting; cross-resistance with raltegravir
5. ENTRY INHIBITORS
5a. Fusion Inhibitor
Drug: Enfuvirtide (ENF, T-20)
Mechanism:
- HIV surface glycoprotein gp120 binds to CD4 on the host cell → exposes gp41 (transmembrane protein).
- gp41 undergoes a hairpin conformational change that brings the viral and host cell membranes close together → membrane fusion → viral RNA enters the cell.
- Enfuvirtide is a 36-amino-acid synthetic peptide that binds to gp41, preventing its conformational change → fusion blocked.
- Must be given subcutaneously (is a peptide, not orally bioavailable).
- Used only in treatment-experienced patients with multidrug-resistant HIV.
- ADR: injection-site reactions (pain, erythema, nodules) — nearly universal.
5b. CCR5 Antagonist (Co-receptor Antagonist)
Drug: Maraviroc (MVC)
Mechanism:
- HIV requires a co-receptor (CCR5 or CXCR4) in addition to CD4 for entry.
- Maraviroc binds CCR5 on the host cell → allosteric conformational change → gp120 cannot bind CCR5 → viral entry blocked.
- Tropism testing is mandatory before use (effective only against R5-tropic virus, not X4-tropic).
- ADR: hepatotoxicity (with allergic reaction), possible cardiovascular risk, upper respiratory tract infections.
5c. gp120 Attachment Inhibitor
Drug: Fostemsavir (FTR)
- Prodrug of temsavir; binds gp120 directly → prevents attachment to CD4.
- Used in heavily treatment-experienced adults with multidrug-resistant HIV.
5d. CD4 Post-attachment Inhibitor
Drug: Ibalizumab (IBA)
- Humanized monoclonal antibody that binds domain 2 of CD4 → blocks post-attachment conformational changes needed for co-receptor engagement → entry inhibited.
- Given IV every 2 weeks; reserved for multidrug-resistant HIV.
6. CAPSID INHIBITOR (Newest Class)
Drug: Lenacapavir
- Binds HIV-1 capsid protein → disrupts multiple stages of the viral life cycle (nuclear import, assembly, uncoating).
- Given twice-yearly subcutaneous injection — longest-acting antiretroviral available.
- Used for multidrug-resistant HIV in combination with other agents; also under investigation for PrEP.
PHARMACOKINETIC BOOSTERS
| Agent | Mechanism | Purpose |
|---|---|---|
| Ritonavir (low dose, 100 mg) | Potent CYP3A4 inhibitor | Boosts PIs (atazanavir, darunavir, lopinavir) |
| Cobicistat (150 mg) | CYP3A4 inhibitor (no anti-HIV activity) | Boosts elvitegravir, atazanavir, darunavir, TAF |
STANDARD FIRST-LINE ART REGIMEN (Preferred)
2 NRTIs + 1 INSTI (preferred)
OR 1 NNRTI
OR 1 boosted PI
Most preferred: Bictegravir/TAF/emtricitabine (single tablet once daily) or Dolutegravir + TDF/3TC
HIGH-YIELD EXAM POINTS (MBBS 2nd Year)
| Point | Detail |
|---|---|
| First antiretroviral ever approved | Zidovudine (1987) |
| NRTIs mechanism in one line | Chain terminators (no 3′-OH → DNA chain stops) |
| NNRTIs active against | HIV-1 only (not HIV-2) |
| PIs act at which stage | Late stage — after viral budding (polyprotein cleavage) |
| Before starting abacavir | Test HLA-B*5701 |
| Before starting maraviroc | Tropism testing (CCR5 vs CXCR4) |
| Efavirenz main CNS ADR | Vivid dreams, dizziness (usually resolves) |
| Atazanavir unique ADR | Benign indirect hyperbilirubinemia |
| INSTIs chelation interaction | Antacids, iron, calcium → separate by 2+ hours |
| "Booster" drugs | Ritonavir & Cobicistat (CYP3A4 inhibitors) |
Sources: Katzung's Basic & Clinical Pharmacology, 16th ed. | Goodman & Gilman's The Pharmacological Basis of Therapeutics, 14th ed.
Classify general anesthetics. Describe the pharmacology of intravenous anesthetics
Reading File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.