expain in detail clinia features causes defn pathopahysioogy investigation mangemnt in proper way iwth diagram approprite in detail
clonus definition features causes pathophysiology clinical examination
clonus investigation management upper motor neuron lesion
clonus ankle knee neurological examination diagram
🧠 CLONUS — Complete Clinical Overview
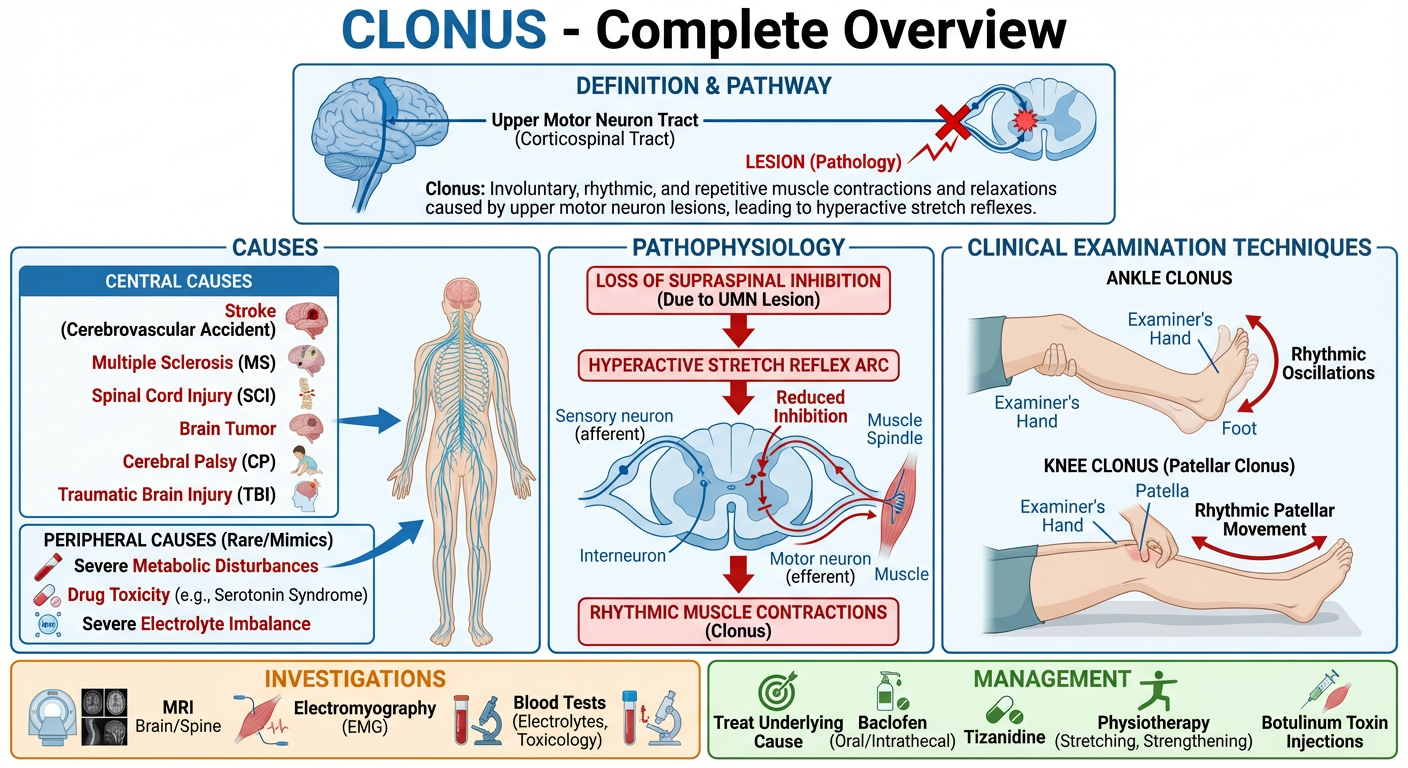
1. DEFINITION
Clinically: sustained tendon stretch → rhythmic oscillations at 5–8 Hz that persist as long as the stretch is maintained.
2. TYPES / FEATURES
| Feature | Description |
|---|---|
| Sustained clonus | Oscillations persist throughout the stretch (pathological) |
| Unsustained clonus | 1–3 beats then stops (may be normal in anxious patients) |
| Ankle clonus | Most common site; tested at ankle by dorsiflexion |
| Knee (patellar) clonus | Tested by sudden downward displacement of the patella |
| Wrist clonus | Less common; wrist extension test |
| Jaw clonus | Rare; sign of bilateral corticobulbar tract lesion |
- Hyperreflexia
- Babinski sign (extensor plantar response)
- Spasticity (velocity-dependent increase in muscle tone)
- Weakness (pyramidal distribution)
- Absent superficial abdominal reflexes
3. CAUSES
🔴 Central Nervous System (UMN Lesions)
| Cause | Notes |
|---|---|
| Stroke (ischemic / hemorrhagic) | Most common cause; contralateral signs |
| Multiple Sclerosis (MS) | Demyelination of corticospinal tracts |
| Traumatic Brain Injury (TBI) | Diffuse axonal injury |
| Brain tumors | Compression of motor cortex / tracts |
| Cerebral Palsy | Perinatal UMN injury |
| Hepatic encephalopathy | Metabolic UMN dysfunction |
| Cause | Notes |
|---|---|
| Spinal cord compression | Cervical spondylotic myelopathy, disc prolapse |
| Transverse myelitis | Inflammatory demyelination |
| Motor Neuron Disease (ALS) | UMN + LMN combination |
| Syringomyelia | Central canal expansion |
| Spinal cord tumors | Intramedullary / extramedullary |
| Subacute Combined Degeneration | Vit B12 deficiency |
| Friedreich's Ataxia | Spinocerebellar degeneration |
🟡 Metabolic / Toxic
- Hypocalcemia (causes neuromuscular hyperexcitability)
- Eclampsia / severe hypomagnesemia
- Serotonin syndrome (drug-induced hyperreflexia + clonus)
- Strychnine poisoning
- Neonatal drug withdrawal
4. PATHOPHYSIOLOGY
NORMAL PHYSIOLOGY:
Muscle Stretch
↓
Ia afferent fibers (muscle spindle)
↓
Alpha motor neuron in anterior horn
↓
Muscle contraction (monosynaptic stretch reflex)
↓
MODULATED by supraspinal corticospinal inhibition
CLONUS MECHANISM (UMN LESION):
Loss of corticospinal inhibitory signals
↓
Alpha motor neuron HYPEREXCITABILITY
↓
Increased muscle spindle (Ia afferent) sensitivity
↓
Stretch reflex threshold LOWERED
↓
Sustained stretch → Ia fires → alpha MN fires → muscle contracts
↓
Muscle shortens → Ia silenced → alpha MN relaxes → muscle relaxes
↓
Relaxation = re-stretching → Ia fires again
↓
RHYTHMIC OSCILLATION (5–8 Hz) = CLONUS
- Loss of supraspinal inhibition — corticospinal tract lesion removes tonic descending inhibition on spinal interneurons
- Increased Ia afferent excitability — lowered threshold of muscle spindle afferents
- Decreased Ib (Golgi tendon organ) inhibitory input — normally limits reflex; impaired in spasticity
- Interneuronal disinhibition — Ia inhibitory interneurons become hyperactive, amplifying reflex
5. CLINICAL EXAMINATION
Ankle Clonus (Most Common)
- Patient supine, knee slightly flexed
- Support the leg under the calf
- Rapidly dorsiflex the foot and maintain the stretch
- Positive: rhythmic beats of plantarflexion-dorsiflexion
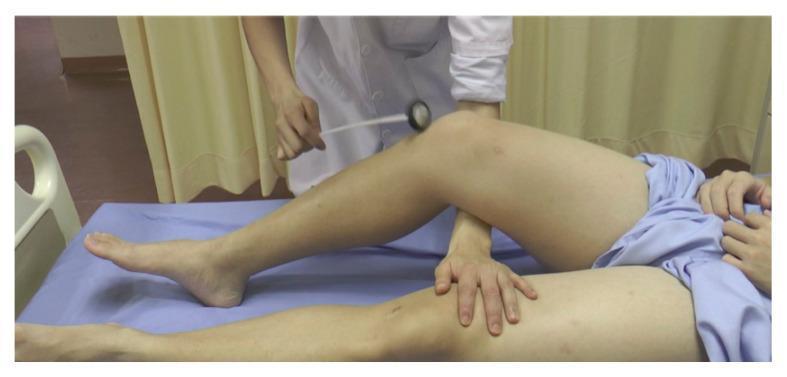
Knee (Patellar) Clonus
- Patient supine, leg extended
- Grasp the patella between thumb and forefinger
- Sharply push it distally and maintain downward pressure
- Positive: rhythmic contractions of quadriceps visible/felt
Grading
| Beats | Interpretation |
|---|---|
| 0 beats | Normal / absent |
| 1–3 beats | Unsustained — possibly normal (anxiety, fatigue) |
| ≥4 sustained | Pathological — UMN lesion |
6. INVESTIGATIONS
First-Line
| Investigation | Purpose |
|---|---|
| MRI Brain | Stroke, MS plaques, tumors, TBI (with gadolinium if needed) |
| MRI Spine | Cord compression, myelitis, syrinx, AVM |
| CBC, ESR, CRP | Inflammatory / infectious etiology |
| Serum B12, folate | Subacute combined degeneration |
| Serum calcium, magnesium | Metabolic causes |
| Blood glucose, LFTs, RFTs | Metabolic encephalopathies |
Second-Line / Specialized
| Investigation | Purpose |
|---|---|
| EMG / Nerve Conduction Studies | Differentiate UMN from LMN; neuromuscular disorders |
| CSF analysis | MS (oligoclonal bands), transverse myelitis, infection |
| Visual Evoked Potentials (VEP) | MS optic nerve involvement |
| CT Brain | Acute setting for hemorrhage (faster than MRI) |
| Urine toxicology | Drug-induced serotonin syndrome, withdrawal |
| Anti-AQP4, Anti-MOG antibodies | Neuromyelitis optica, MOGAD |
Harrison's Principles (p. 751): "Investigations typically begin with spinal MRI, but when UMN signs are associated with drowsiness, confusion, seizures, or other hemispheric signs, brain MRI should also be performed."
7. MANAGEMENT
🎯 Principle: Treat the Underlying Cause First
| Cause | Specific Treatment |
|---|---|
| Stroke | Thrombolysis (ischemic), neurosurgery (hemorrhagic), rehabilitation |
| MS | DMTs (interferon-β, natalizumab, ocrelizumab) |
| Spinal cord compression | Surgical decompression ± steroids |
| B12 deficiency | IM Hydroxocobalamin supplementation |
| Eclampsia | IV Magnesium sulfate, antihypertensives, delivery |
| Serotonin syndrome | Cyproheptadine, benzodiazepines, supportive care |
💊 Pharmacological Management of Spasticity/Clonus
| Drug | Mechanism | Dose | Notes |
|---|---|---|---|
| Baclofen | GABA-B agonist → presynaptic inhibition | 5–20 mg TDS | First-line for spinal spasticity |
| Tizanidine | α2 agonist → reduces excitatory interneurons | 2–8 mg TDS | Hepatotoxicity monitoring required |
| Diazepam | GABA-A agonist | 2–10 mg TDS | Short-term; sedating |
| Dantrolene | Inhibits Ca²⁺ release from SR | 25–100 mg QDS | Acts peripherally; liver function monitoring |
| Clonazepam | GABA-A agonist | 0.5–2 mg OD | Useful especially for nocturnal clonus |
| Intrathecal Baclofen | Direct spinal GABA-B agonism | Pump-delivered | Severe refractory spasticity |
💉 Interventional
| Intervention | Indication |
|---|---|
| Botulinum Toxin A (Botox) | Focal spasticity/clonus; blocks ACh release at NMJ |
| Intrathecal baclofen pump | Severe generalized spasticity unresponsive to oral therapy |
| Phenol nerve block | Selective motor nerve blockade; longer lasting than Botox |
🏃 Physiotherapy & Rehabilitation
- Stretching exercises — maintain range of motion, reduce contractures
- Splinting / orthoses — ankle-foot orthosis (AFO) prevents equinus deformity
- Hydrotherapy — warm water reduces spasticity
- Functional electrical stimulation (FES) — modulates spinal cord excitability
- TENS — reduces clonus via sensory modulation
- Constraint-induced movement therapy (CIMT) — post-stroke rehabilitation
8. COMPLICATIONS (If Untreated)
- Contractures — fixed joint deformities
- Pressure sores — from immobility
- Falls and injury — from unpredictable limb spasms
- Pain — painful muscle spasms
- Functional disability — impaired gait, ADLs
- Psychological impact — depression, reduced QoL
9. SUMMARY FLOWCHART
Patient presents with rhythmic involuntary limb contractions
↓
Elicit clonus clinically
(ankle/knee/wrist)
↓
Sustained ≥4 beats → PATHOLOGICAL
↓
Look for other UMN signs:
Hyperreflexia + Babinski + Spasticity
↓
Localize the lesion:
Brain (+ cortical signs) vs Spinal cord
↓
┌───────────────────────────────┐
│ MRI Brain / Spine │
│ + Blood investigations │
│ + EMG/NCS if needed │
└───────────────────────────────┘
↓
Treat underlying cause
↓
Symptom control: Baclofen/Tizanidine
↓
Physiotherapy + Rehabilitation
10. KEY DIFFERENTIALS
| Condition | Differentiating Features |
|---|---|
| Tremor | Not stretch-triggered; present at rest or with movement |
| Myoclonus | Sudden, shock-like; not rhythmic oscillation |
| Epileptic seizures | Altered consciousness; EEG changes |
| Asterixis | Metabolic; "negative myoclonus" — brief lapses in posture |
| Normal hyperreflexia | No sustained clonus; no other UMN signs |
Note: If you meant a different "clinia" term (e.g., Cholinergic Crisis, Colitis, Cholera, or another condition), please clarify and I'll provide a full detailed breakdown of that topic instead.
In detail, explain in detail clinical features, causes, definition, and investigation in detail and medical management, surgical management in detail about GBS.
Guillain-Barre syndrome definition pathophysiology etiology causes
Guillain-Barre syndrome clinical features variants AIDP AMAN Miller Fisher
Guillain-Barre syndrome investigations CSF nerve conduction EMG diagnosis criteria
Guillain-Barre syndrome treatment IVIG plasmapheresis management ICU ventilation
Guillain-Barre syndrome nerve demyelination pathology diagram
🧠 GUILLAIN-BARRÉ SYNDROME (GBS) — Complete Clinical Overview
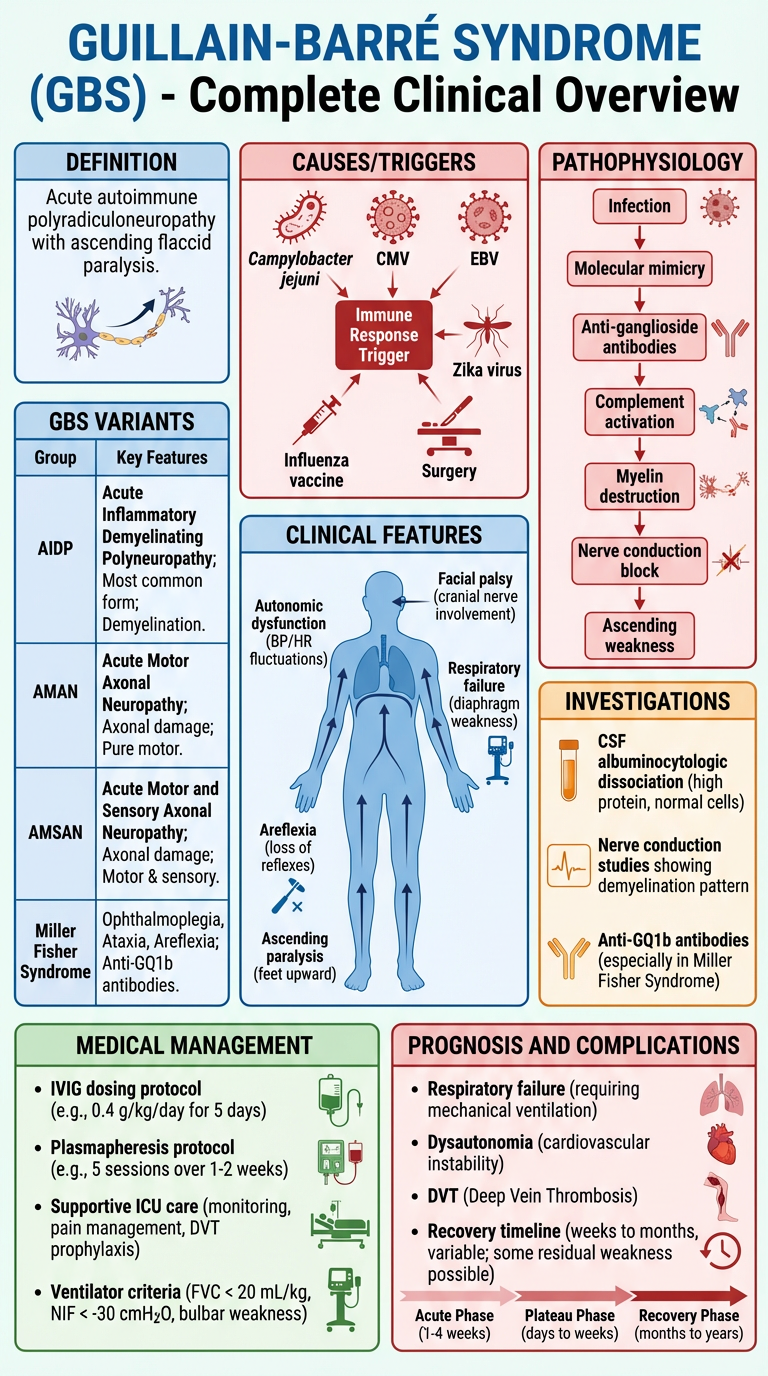
1. DEFINITION
Harrison's Principles of Internal Medicine, 21st Ed. (p. 12718): "GBS is an acute, frequently severe, and fulminant polyradiculoneuropathy that is autoimmune in nature. It occurs year-round at a rate of between 10–20 cases per million annually."
- Incidence: 10–20 per million per year worldwide
- ~5,000–6,000 cases/year in the United States
- Males slightly more affected than females
- Bimodal age distribution: young adults (15–35 yrs) and elderly (50–75 yrs)
- Leading cause of acute flaccid paralysis in post-polio era
2. CAUSES & TRIGGERING FACTORS
🦠 Infectious Triggers
| Organism | Notes |
|---|---|
| Campylobacter jejuni | Most common trigger (~30%); associated with AMAN variant; anti-GM1/GD1a antibodies |
| Cytomegalovirus (CMV) | Second most common; associated with severe sensory involvement |
| Epstein-Barr Virus (EBV) | Mononucleosis-associated GBS |
| Zika Virus | Epidemics in South America, Pacific Islands |
| HIV | GBS can occur at seroconversion |
| SARS-CoV-2 (COVID-19) | Reported association, cranial nerve variants |
| Mycoplasma pneumoniae | Atypical pneumonia preceding GBS |
| Haemophilus influenzae | Upper respiratory tract infection |
| Hepatitis A, B, E | Hepatitis-associated GBS |
💉 Non-Infectious Triggers
| Trigger | Notes |
|---|---|
| Vaccination | Influenza vaccine (rare, ~1–2/million doses); swine flu vaccine 1976 |
| Surgery / Trauma | Post-operative GBS (rare) |
| Pregnancy / Postpartum | Immune dysregulation |
| Bone marrow transplantation | Graft-versus-host immune mechanisms |
| Lymphoma (Hodgkin's) | Paraneoplastic trigger |
| SLE / Autoimmune disease | Background immune dysregulation |
3. PATHOPHYSIOLOGY
Molecular Mimicry — Core Mechanism
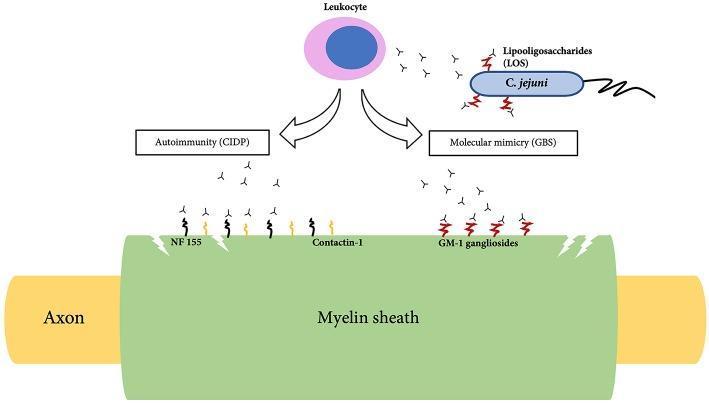
STEP 1: PRECEDING INFECTION
Campylobacter jejuni / Virus infects patient
↓
STEP 2: IMMUNE ACTIVATION
T-cells and B-cells activated against pathogen antigens
↓
STEP 3: MOLECULAR MIMICRY
Pathogen surface antigens (e.g., LOS of C. jejuni)
structurally resemble gangliosides on peripheral nerve myelin
(GM1, GD1a, GQ1b, GT1a)
↓
STEP 4: AUTOANTIBODY PRODUCTION
IgG anti-ganglioside antibodies produced
↓
STEP 5: COMPLEMENT ACTIVATION
Antibodies bind to Nodes of Ranvier / myelin sheath
→ Complement cascade activated (C3b, C5b-9 MAC)
↓
STEP 6: NERVE DAMAGE
① Demyelination → slowed conduction (AIDP)
② Axonal damage → lost conduction (AMAN/AMSAN)
↓
STEP 7: CLINICAL SYNDROME
Ascending weakness, areflexia, sensory loss,
autonomic dysfunction, respiratory failure
Histopathology
- AIDP: Lymphocytic infiltration + macrophage-mediated demyelination of peripheral nerves and roots; segmental demyelination
- AMAN: IgG/complement attack on nodal axolemma at Nodes of Ranvier; axonal degeneration without demyelination
- AMSAN: Same as AMAN but includes sensory axons; severe, poor recovery
4. GBS VARIANTS
| Variant | Full Name | Key Features | Antibody | Prognosis |
|---|---|---|---|---|
| AIDP | Acute Inflammatory Demyelinating Polyneuropathy | Classic ascending weakness; demyelinating NCS | Anti-GM1 (some) | Good |
| AMAN | Acute Motor Axonal Neuropathy | Pure motor; preceded by C. jejuni; common in Asia/China | Anti-GM1, Anti-GD1a | Variable |
| AMSAN | Acute Motor-Sensory Axonal Neuropathy | Motor + sensory axonal loss; severe | Anti-GD1a | Poor |
| MFS | Miller Fisher Syndrome | Triad: ophthalmoplegia + ataxia + areflexia; NO limb weakness | Anti-GQ1b | Excellent |
| PCB | Pharyngeal-Cervical-Brachial | Oropharyngeal + neck + arm weakness; no leg involvement | Anti-GT1a | Good |
| BBE | Bickerstaff's Brainstem Encephalitis | Ophthalmoplegia + ataxia + drowsiness/coma (CNS involvement) | Anti-GQ1b | Good |
| Acute Panautonomic | — | Severe autonomic failure; minimal weakness | — | Variable |
5. CLINICAL FEATURES
🔺 Typical Presentation Timeline
WEEK 1–2: PRODROME
Preceding infection (URTI, gastroenteritis)
↓
DAY 1–7: ONSET
Tingling / paresthesias in toes and fingertips
Mild symmetric limb weakness (distal → proximal)
↓
WEEK 1–4: PROGRESSION PHASE
Ascending flaccid paralysis
Areflexia (loss of tendon reflexes)
Pain (back pain, radicular pain) — often early
↓
WEEK 2–4: PLATEAU PHASE
Maximum weakness reached
~25–30% require mechanical ventilation
↓
WEEKS TO MONTHS: RECOVERY PHASE
Slow recovery from proximal to distal
🔴 Motor Features
- Ascending bilateral symmetric weakness — starts in lower limbs, ascends to trunk, arms, cranial muscles
- Flaccid paralysis — reduced tone (contrast with UMN spasticity)
- Areflexia (hallmark) — absent deep tendon reflexes bilaterally
- Respiratory muscle weakness → may need ventilator (~25–30%)
- Facial diplegia (~50%) — bilateral LMN facial nerve palsy
- Oculomotor palsy — especially in Miller Fisher variant
- Bulbar weakness — dysphagia, dysphonia (risk of aspiration)
- Neck flexor weakness — patient cannot lift head off pillow
🟡 Sensory Features
- Glove-and-stocking paresthesias — tingling, numbness (distal)
- Back pain and radicular pain — often the first symptom
- Proprioceptive loss — unsteady gait
- Pain — often severe, neuropathic in nature; early and prominent feature
- Sensory loss often LESS severe than motor loss
🟠 Autonomic Features (in ~70% of cases)
| Autonomic Feature | Clinical Manifestation |
|---|---|
| Cardiac dysrhythmia | Sinus tachycardia, bradycardia, heart block |
| Blood pressure lability | Hypertension ↔ hypotension |
| Urinary retention | Neurogenic bladder |
| Constipation / ileus | Reduced gut motility |
| Diaphoresis | Excessive sweating |
| Orthostatic hypotension | On standing |
| Autonomic storm | Life-threatening BP/HR swings |
⚠️ Autonomic dysfunction is a major cause of death in GBS — requires continuous cardiac monitoring in ICU.
🔵 Cranial Nerve Features
| Cranial Nerve | Feature |
|---|---|
| CN VII (Facial) | Facial diplegia (bilateral) — most common |
| CN IX/X (Glossopharyngeal/Vagus) | Dysphagia, dysarthria |
| CN III/IV/VI (Oculomotor) | Ophthalmoplegia (Miller Fisher) |
| CN XII (Hypoglossal) | Tongue weakness |
📋 Brighton Collaboration Diagnostic Criteria (2011)
| Level | Criteria |
|---|---|
| Level 1 (most certain) | Bilateral flaccid limb weakness + decreased/absent DTRs in weak limbs + monophasic illness + interval 12 hrs–28 days to nadir + CSF cell count ≤50/μL + CSF protein above normal + NCS consistent with GBS subtype |
| Level 2 | Above except CSF results absent/unavailable |
| Level 3 | Bilateral flaccid limb weakness + decreased/absent DTRs + monophasic illness |
6. INVESTIGATIONS
🔬 Cerebrospinal Fluid (CSF) Analysis — Most Important
| Parameter | Finding in GBS | Significance |
|---|---|---|
| Protein | Elevated (> 0.45 g/L; often 1–10 g/L) | Key finding |
| Cell count | Normal (< 10 cells/μL) | No pleocytosis |
| Glucose | Normal | Differentiates from infection |
| Pattern | Albuminocytologic dissociation | HALLMARK of GBS |
| Timing | Normal in first 1 week in ~50% | Repeat if negative early |
Albuminocytologic dissociation = high protein + normal cell count. This is the pathognomonic CSF finding of GBS.
⚡ Nerve Conduction Studies (NCS) + EMG
| Finding | AIDP (Demyelinating) | AMAN (Axonal) |
|---|---|---|
| Conduction velocity | Reduced (< 70% normal) | Normal or mildly reduced |
| Distal latency | Prolonged | Normal or mildly prolonged |
| F-waves | Absent/prolonged | Present |
| CMAP amplitude | Mildly reduced | Severely reduced |
| Sensory NCS | Abnormal | Normal (pure motor) |
| H-reflex | Absent early | May be absent |
NCS are the most sensitive investigation — abnormal in >85% of cases within 2 weeks.
🩸 Blood Investigations
| Test | Purpose |
|---|---|
| Anti-ganglioside antibodies | Anti-GQ1b (MFS), Anti-GM1/GD1a (AMAN) |
| CBC, ESR, CRP | Baseline; exclude infection |
| LFTs, RFTs | Baseline; monitor drug toxicity |
| Stool culture / PCR | Confirm C. jejuni precipitant |
| HIV serology | Exclude HIV-associated GBS |
| Serum electrolytes | Hyponatremia (SIADH can occur) |
| Campylobacter antibodies | Serological confirmation |
| Anti-nuclear antibodies (ANA) | Exclude SLE |
🖥️ Imaging
| Investigation | Indication | Finding |
|---|---|---|
| MRI Spine with contrast | Exclude cord compression; confirm nerve root enhancement | Gadolinium enhancement of spinal nerve roots / cauda equina |
| MRI Brain | If encephalopathy or cranial nerve involvement (BBE) | Brainstem enhancement in BBE |
| Chest X-Ray | Respiratory monitoring; aspiration pneumonia | Consolidation if aspirated |
| CT Brain | If altered consciousness | Usually normal |
📈 Respiratory Monitoring (Critical)
| Parameter | Action Threshold |
|---|---|
| Forced Vital Capacity (FVC) | < 20 mL/kg → consider elective intubation |
| FVC < 15 mL/kg | Mandatory intubation |
| Negative Inspiratory Force (NIF) | < –25 cmH₂O → consider intubation |
| SpO₂ < 92% | Urgent airway management |
| "20-30-40 Rule" | FVC < 20 mL/kg, MIP < 30 cmH₂O, MEP < 40 cmH₂O → intubate |
7. MEDICAL MANAGEMENT
🚨 Phase 1: Emergency Stabilization & ICU Monitoring
On diagnosis of GBS:
1. ADMIT to monitored bed (HDU/ICU if moderate-severe)
2. RESPIRATORY: serial FVC q4-6h, pulse oximetry
3. CARDIAC: continuous ECG monitoring (autonomic dysfunction)
4. IV ACCESS + baseline bloods
5. URINARY CATHETER if retention
6. DVT PROPHYLAXIS: LMWH + compression stockings
7. NG TUBE if bulbar palsy / swallowing impaired
8. PAIN MANAGEMENT: neuropathic agents
💊 Specific Immunomodulatory Treatment
Option 1: Intravenous Immunoglobulin (IVIG) ✅ FIRST-LINE
| Parameter | Detail |
|---|---|
| Mechanism | Neutralizes autoantibodies; blocks Fc receptors; modulates complement; reduces T-cell activation |
| Dose | 0.4 g/kg/day IV for 5 days (total 2 g/kg) |
| Indication | Unable to walk independently (GBS disability score ≥ 3), or rapidly deteriorating |
| Onset | Improvement begins within 1–2 weeks |
| Advantages | Easy to administer; no special equipment; can be given in non-ICU settings |
| Side effects | Headache, fever, hemolysis, renal failure (sucrose-containing), thrombosis, aseptic meningitis, anaphylaxis (IgA deficiency) |
| Contraindications | IgA deficiency (anaphylaxis risk) — check IgA levels before administration |
Option 2: Plasma Exchange (Plasmapheresis / PE) ✅ EQUALLY EFFECTIVE
| Parameter | Detail |
|---|---|
| Mechanism | Removes circulating autoantibodies (anti-ganglioside IgG), complement, inflammatory mediators |
| Protocol | 5 exchanges over 10–14 days (200–250 mL/kg total); exchange with albumin or FFP |
| Indication | Same as IVIG; preferred if IVIG contraindicated |
| Timing | Most effective if started within 2 weeks of onset |
| Advantages | Proven efficacy; rapid antibody removal |
| Side effects | Hypotension, hypocalcemia (citrate toxicity), line infections, pneumothorax, clotting factor depletion, hemodynamic instability |
| Disadvantages | Requires central venous access; specialized equipment; not available everywhere |
⚠️ IVIG + Plasmapheresis combined is NOT more effective than either alone (Dutch GBS Study Group). Do NOT combine routinely.
❌ Corticosteroids are NOT beneficial in GBS — multiple RCTs have shown no benefit; they may even delay recovery. Do NOT use steroids in GBS.
📊 IVIG vs Plasmapheresis Comparison
| Feature | IVIG | Plasmapheresis |
|---|---|---|
| Efficacy | Equivalent | Equivalent |
| Ease of use | ✅ Easier | ❌ Complex |
| Availability | ✅ Widely available | ❌ Specialist centers |
| Cost | Higher | Moderate |
| Access requirement | Peripheral IV | Central venous catheter |
| Pediatric use | ✅ Preferred | Less preferred |
| Hemodynamic stability needed | Less critical | ✅ Required |
💊 Supportive Medical Management
Pain Management
| Drug | Dose | Mechanism |
|---|---|---|
| Gabapentin | 300–900 mg TDS | Ca²⁺ channel α2δ ligand; neuropathic pain |
| Pregabalin | 75–150 mg BD | Same class as gabapentin |
| Carbamazepine | 200–400 mg BD | Na⁺ channel stabilizer; radicular pain |
| IV Morphine / Opioids | PRN | Severe acute pain |
| IV Ketamine | Infusion | Refractory neuropathic pain in ICU |
| Amitriptyline | 10–75 mg nocte | Chronic neuropathic pain (recovery phase) |
Autonomic Dysfunction
| Problem | Management |
|---|---|
| Tachycardia | Short-acting beta-blocker (esmolol IV) — use cautiously |
| Bradycardia / Heart block | Atropine IV; transcutaneous pacing if severe |
| Hypertension | IV labetalol, nitroprusside; avoid overtreatment |
| Hypotension | IV fluids, cautious vasopressors (phenylephrine, norepinephrine) |
| Urinary retention | Urinary catheterization |
| Ileus / constipation | Prokinetics, stool softeners, NG feeding |
Respiratory Support
| Step | Action |
|---|---|
| FVC 20–30 mL/kg | Monitor q4h, BiPAP if hypercapnic |
| FVC < 20 mL/kg | Elective intubation + mechanical ventilation |
| Bulbar palsy present | Early intubation (aspiration risk) |
| Ventilation weaning | Slow, guided by FVC recovery (> 15 mL/kg to extubate) |
| Tracheostomy | If prolonged ventilation expected (> 2–3 weeks) |
DVT / PE Prevention
- LMWH (e.g., enoxaparin 40 mg SC OD) — started early
- Graduated compression stockings
- Intermittent pneumatic compression
- Early mobilization when possible
Nutrition
- NG feeding if dysphagia/bulbar palsy
- High-protein diet to support nerve recovery
- Vitamin B12 monitoring and supplementation
Psychological Support
- GBS is extremely distressing — rapid paralysis with preserved consciousness
- Regular reassurance of expected recovery
- Psychiatric liaison if severe anxiety/depression
- Patient and family education
8. SURGICAL / INTERVENTIONAL MANAGEMENT
🔧 Surgical / Procedural Interventions
1. Tracheostomy
| Indication | Details |
|---|---|
| Prolonged mechanical ventilation (>14–21 days) | Reduces VAP risk, allows weaning trials, improves comfort |
| Severe bulbar palsy | Airway protection |
| Procedure | Percutaneous dilational tracheostomy (PDT) at bedside, or surgical tracheostomy in OR |
| Benefits | Reduced sedation needs, better oral hygiene, easier weaning, patient communication |
2. Central Venous Access (for Plasmapheresis)
- Double-lumen central venous catheter (internal jugular or femoral)
- Required for adequate blood flow during plasma exchange (>150 mL/min)
- Placed under USS guidance in ICU or radiology suite
- Complications: infection, pneumothorax, thrombosis
3. Percutaneous Endoscopic Gastrostomy (PEG)
| Indication | Details |
|---|---|
| Prolonged dysphagia (>4–6 weeks) | Provides long-term nutritional route |
| Persistent bulbar palsy | Prevents repeated NG tube insertion |
| Procedure | Endoscopic + percutaneous placement of gastrostomy tube |
4. Urinary Catheterization (Surgical Cystostomy)
- If prolonged urinary retention and urethral catheter not tolerated
- Suprapubic cystostomy placed surgically or percutaneously
5. Surgical Management of Complications
| Complication | Surgical Intervention |
|---|---|
| Pulmonary embolism | IVC filter (if anticoagulation contraindicated) |
| Pressure sores | Surgical debridement ± skin grafting |
| Joint contractures | Surgical release (rare; rehabilitation-first approach) |
| Corneal ulceration (from facial diplegia) | Lateral tarsorrhaphy or eye patch to protect cornea |
6. Cardiac Pacing
- Temporary transcutaneous/transvenous pacing for severe autonomic bradycardia / complete heart block
- A cardiology/electrophysiology consult required
- Permanent pacemaker rarely needed — autonomic dysfunction typically resolves
9. DISABILITY GRADING SCALE (GBS-DS)
| Grade | Description |
|---|---|
| 0 | Healthy |
| 1 | Minor symptoms; able to run |
| 2 | Able to walk >10 m without aid |
| 3 | Able to walk >10 m WITH aid/support |
| 4 | Bedridden/wheelchair bound |
| 5 | Requires assisted ventilation |
| 6 | Dead |
Treat with IVIG or PE if Grade ≥ 3 (cannot walk independently) or if deteriorating rapidly.
10. COMPLICATIONS
| System | Complication |
|---|---|
| Respiratory | Respiratory failure (25–30%), aspiration pneumonia, pneumothorax |
| Cardiovascular | Arrhythmias, hemodynamic instability, cardiac arrest |
| Musculoskeletal | Contractures, pressure sores, foot drop |
| Thromboembolic | DVT, pulmonary embolism |
| Neurological | Chronic pain, residual weakness, relapse (5%) |
| Psychological | Depression, PTSD, anxiety |
| Nutritional | Malnutrition from dysphagia |
| Urological | UTI from catheterization |
| SIADH | Hyponatremia |
11. PROGNOSIS
| Timeframe | Outcome |
|---|---|
| Plateau | Reached within 2–4 weeks in 90% |
| Recovery begins | 2–4 weeks after plateau |
| Full recovery | ~85% by 6–12 months |
| Residual disability | ~15–20% have some permanent deficit |
| Mortality | ~3–5% (higher in elderly, ventilated patients) |
| Relapse | ~5% (consider CIDP if >3 relapses) |
Poor Prognostic Factors
- Age > 60 years
- Rapid progression (nadir within 1 week)
- Preceding C. jejuni infection
- AMAN/AMSAN variant
- Need for mechanical ventilation
- Low CMAP amplitude on NCS
- Axonal loss on EMG
12. DIFFERENTIALS TO EXCLUDE
| Condition | Key Differentiating Feature |
|---|---|
| CIDP | Chronic (>8 weeks); responds to steroids |
| Myasthenia Gravis | Fatigable weakness; NMJ disorder; reflexes preserved |
| Botulism | Descending paralysis; dilated pupils; toxin exposure |
| Transverse Myelitis | Spinal cord level; UMN signs; bladder early |
| Poliomyelitis | Asymmetric; fever; CSF pleocytosis |
| Tick Paralysis | Ascending weakness; resolves on tick removal |
| Hypermagnesemia | Reflexes lost; history of Mg exposure |
| Critical Illness Polyneuropathy | Ventilated ICU patient; EMG changes |
| Diphtheria | Palatal palsy first; diphtheritic membrane |
| West Nile Virus | Asymmetric; fever; pleocytosis |
13. SUMMARY ALGORITHM
Acute ascending flaccid paralysis + areflexia
↓
Exclude spinal cord compression (MRI spine)
↓
CSF: Albuminocytologic dissociation (↑protein, normal cells)
NCS/EMG: Demyelinating or axonal pattern
↓
DIAGNOSE GBS
↓
ASSESS SEVERITY (GBS Disability Scale)
↓
┌────────────────────────────────────────┐
│ IMMEDIATE ICU/HDU admission │
│ Serial FVC every 4–6 hours │
│ Continuous cardiac monitoring │
│ DVT prophylaxis │
└────────────────────────────────────────┘
↓
SPECIFIC TREATMENT (if Grade ≥3 or deteriorating):
┌─────────────────────┬─────────────────────┐
│ IVIG 0.4 g/kg/day │ Plasmapheresis │
│ × 5 days IV │ 5 exchanges/10 days │
│ (First-line, easier)│ (Equally effective) │
└─────────────────────┴─────────────────────┘
(Do NOT combine; Do NOT use steroids)
↓
SUPPORTIVE:
Pain control | Autonomic monitoring
Nutrition | Physiotherapy | Psychological support
↓
MONITOR FOR RESPIRATORY FAILURE:
FVC < 20 mL/kg → Elective intubation
Bulbar palsy → Early intubation
↓
SURGICAL (if needed):
Tracheostomy | PEG | Central line | Pacing
↓
REHABILITATION:
Physiotherapy | OT | Speech therapy
Graded mobilization | AFO if foot drop
- Harrison's Principles of Internal Medicine, 21st Edition (pp. 12718, 12724)
- Brighton Collaboration GBS Diagnostic Criteria (2011)
- Dutch GBS Study Group — IVIG vs Plasmapheresis RCT
- European Academy of Neurology / Peripheral Nerve Society GBS Guidelines
Explain in detail, explain in detail, causes, classification, clinical features, pathology, then investigations, and medical and surgical management of head injury, everything related to head injury, clinical neuro, with appropriate diagrams.
traumatic brain injury TBI classification causes GCS primary secondary injury mechanisms
head injury pathology epidural hematoma subdural hematoma subarachnoid hemorrhage contusion diffuse axonal injury
traumatic brain injury clinical features investigation CT scan NICE criteria management ICP
head injury surgical management craniotomy decompressive craniectomy intracranial pressure monitoring burr hole
epidural hematoma subdural hematoma CT scan head injury brain
Glasgow Coma Scale head injury assessment neurological
Explain in detail, explain in detail, causes, classification, clinical features, pathology, then investigations, and medical and surgical management of head injury, everything related to head injury, clinical neuro, with appropriate diagrams.
traumatic brain injury TBI classification causes GCS primary secondary injury mechanisms
head injury pathology epidural subdural hematoma contusion diffuse axonal injury skull fracture
epidural hematoma subdural hematoma CT scan traumatic brain injury
Glasgow Coma Scale head injury neurological assessment table
🧠 HEAD INJURY (Traumatic Brain Injury) — Complete Clinical Overview
1. DEFINITION
Bailey & Love's Short Practice of Surgery, 28th Ed. (p. 381): "TBI can be considered as the combination of primary injury sustained on impact, and hence not medically modifiable, and secondary injury developing in the following hours and days. Understanding the importance of intracranial pressure (ICP) and related parameters is key to minimising secondary injury and improving outcomes."
- Leading cause of death and disability in persons aged 1–45 years
- Worldwide incidence: ~69 million TBIs per year
- Males affected 2–3× more than females
- Bimodal peak: young adults (15–24 yrs) and elderly (>65 yrs)
- Road traffic accidents are the most common cause globally
2. CAUSES
| Category | Specific Causes |
|---|---|
| Road Traffic Accidents (RTA) | Most common globally; drivers, passengers, pedestrians, cyclists |
| Falls | Most common in elderly and children; domestic, occupational |
| Assault / Violence | Direct blows, interpersonal violence; shaken baby syndrome |
| Sports injuries | Boxing, rugby, cycling, equestrian, contact sports |
| Blast / Explosion | Military combat, improvised explosive devices (IEDs) |
| Penetrating injuries | Gunshot wounds, stab wounds, impalement |
| Industrial / Occupational | Falling objects, machinery accidents |
| Birth trauma | Forceps delivery, vacuum extraction |
3. CLASSIFICATION
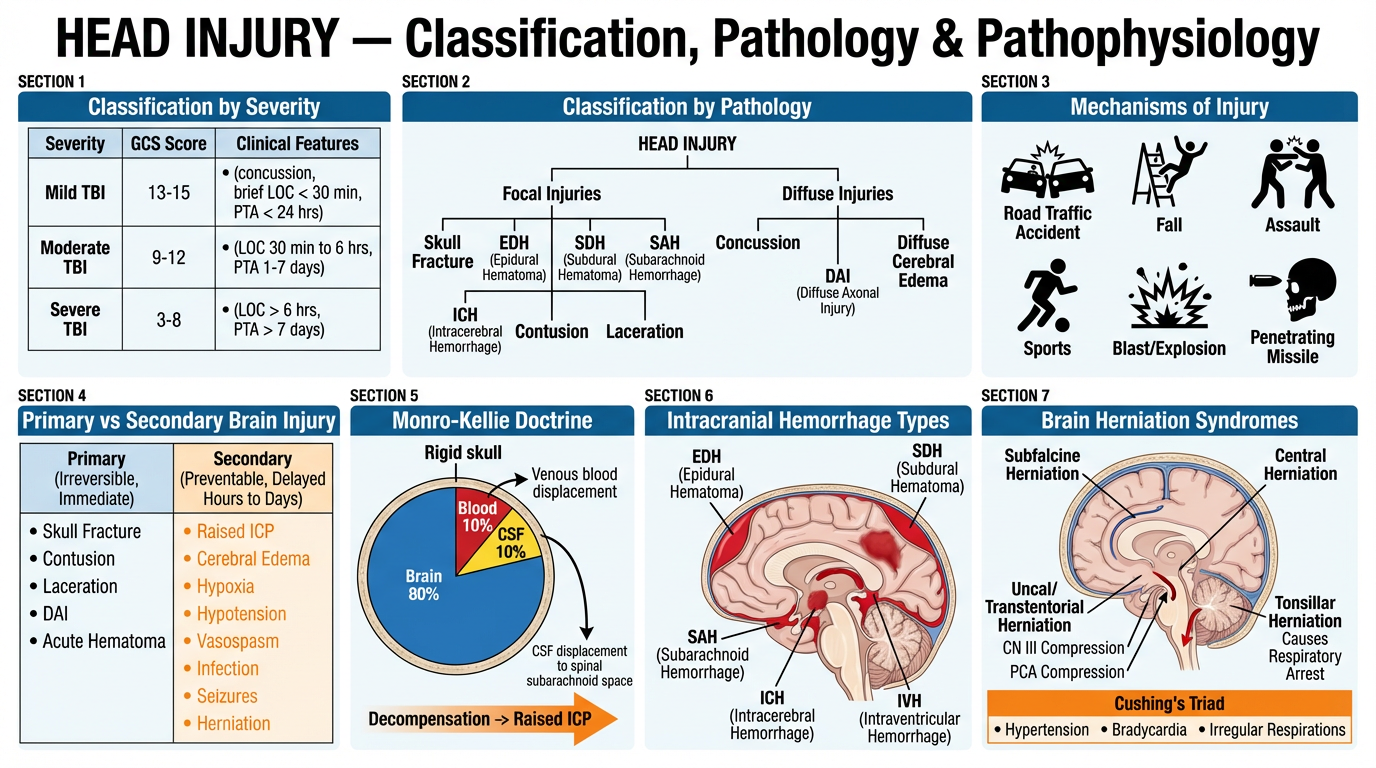
3A. By Severity — Glasgow Coma Scale (GCS)
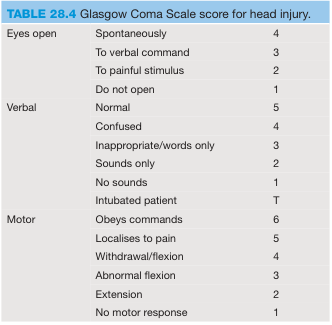
| Component | Response | Score |
|---|---|---|
| Eye Opening (E) | Spontaneously | 4 |
| To verbal command | 3 | |
| To painful stimulus | 2 | |
| No response | 1 | |
| Verbal (V) | Normal/oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Sounds only | 2 | |
| No sounds | 1 | |
| Intubated | T | |
| Motor (M) | Obeys commands | 6 |
| Localises to pain | 5 | |
| Withdrawal/flexion | 4 | |
| Abnormal flexion (decorticate) | 3 | |
| Extension (decerebrate) | 2 | |
| No motor response | 1 |
| Severity | GCS Score | LOC Duration | PTA Duration |
|---|---|---|---|
| Mild TBI | 13–15 | < 30 minutes | < 24 hours |
| Moderate TBI | 9–12 | 30 min – 6 hours | 1–7 days |
| Severe TBI | 3–8 | > 6 hours | > 7 days |
GCS ≤ 8 = comatose → requires airway protection (intubation)
3B. By Structural Nature
| Type | Description |
|---|---|
| Open (Compound) | Breach in scalp + skull (dura may be torn); infection risk |
| Closed | No breach of dura; most common |
| Penetrating | Foreign body enters cranial cavity (bullet, knife) |
| Depressed fracture | Bone fragments pushed inward ≥ thickness of skull |
3C. By Pathological Type
| Category | Subtypes |
|---|---|
| Focal Injuries | Skull fracture, EDH, SDH, SAH, ICH, cerebral contusion, laceration |
| Diffuse Injuries | Concussion, diffuse axonal injury (DAI), diffuse cerebral edema |
3D. By Mechanism
| Mechanism | Injury Pattern |
|---|---|
| Acceleration-deceleration | Coup-contrecoup contusions, SDH, DAI |
| Direct impact (contact) | Skull fracture, EDH, local contusion |
| Rotational / angular | DAI (white matter shearing) |
| Penetrating | Laceration, intracerebral hemorrhage along tract |
| Blast wave | Diffuse axonal injury, contusion |
4. PATHOLOGY
4A. Primary vs Secondary Brain Injury
PRIMARY BRAIN INJURY (Immediate — NOT reversible)
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
Occurs at moment of impact
• Skull fracture
• Cerebral contusion/laceration
• Diffuse axonal injury (DAI)
• Acute intracranial hemorrhage (EDH, SDH, SAH, ICH)
• Direct neuronal death
SECONDARY BRAIN INJURY (Hours to days — PREVENTABLE)
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
Systemic: Hypoxia (SpO₂ <90%), Hypotension (SBP <90 mmHg),
Hyperthermia, Hypoglycemia, Hypo/hypernatremia
Intracranial: Raised ICP, Cerebral edema, Vasospasm,
Excitotoxicity (glutamate), Free radical damage,
Herniation, Seizures, Infection
Key principle: Primary injury cannot be reversed; all treatment targets secondary injury prevention.
4B. Monro-Kellie Doctrine
- Brain parenchyma: 80% (~1200 mL)
- Blood (arterial + venous): 10% (~150 mL)
- CSF: 10% (~150 mL)
- CSF displaced to spinal subarachnoid space
- Venous blood displaced to jugular veins
- Once exhausted → ICP rises exponentially
Normal ICP: 7–15 mmHg; Treatment threshold: >20–22 mmHg
CPP = MAP − ICP
Target CPP: 60–70 mmHg
4C. Specific Pathological Entities
1. 💀 Skull Fractures
| Type | Description | Significance |
|---|---|---|
| Linear | Simple crack, no displacement | Low risk; marker for underlying hematoma |
| Depressed | Bone pushed inward ≥ skull thickness | Brain compression; surgical elevation needed |
| Comminuted | Multiple fragments | Often needs surgical repair |
| Open (Compound) | Overlying scalp laceration | Infection risk; needs surgical debridement |
| Basal skull fracture | Involves skull base | CSF leak, CN palsies, vascular injury |
| Diastatic | Fracture widens a suture | Common in children |
| Sign | Location | Significance |
|---|---|---|
| Raccoon eyes | Bilateral periorbital bruising | Anterior cranial fossa fracture |
| Battle's sign | Mastoid ecchymosis | Middle cranial fossa / petrous bone fracture |
| CSF rhinorrhoea | CSF from nose | Anterior fossa — cribriform plate fracture |
| CSF otorrhoea | CSF from ear | Middle fossa — petrous temporal bone |
| Hemotympanum | Blood behind tympanic membrane | Middle fossa fracture |
| CN VII/VIII palsy | Facial weakness/deafness | Petrous temporal bone fracture |
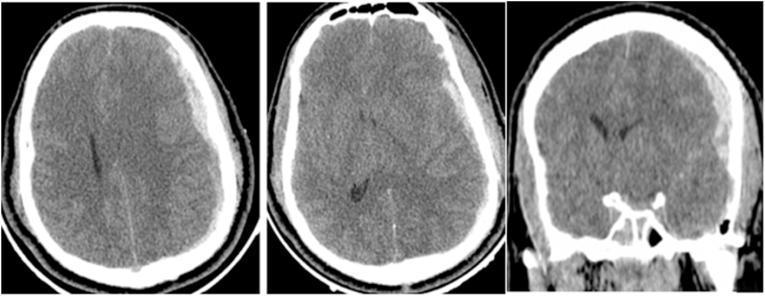
2. 🔴 Extradural (Epidural) Hematoma (EDH)
| Feature | Detail |
|---|---|
| Definition | Arterial bleed between skull and dura |
| Source | Middle meningeal artery (85%); dural venous sinus |
| Cause | Temporal bone fracture tearing MMA |
| CT appearance | Biconvex (lenticular) hyperdense collection; does NOT cross suture lines |
| Classic history | Trauma → lucid interval (minutes to hours) → rapid deterioration |
| Lucid interval | Due to initial concussion recovery, then progressive hematoma expansion |
| Location | Temporal/temporoparietal (most common) |
| Signs | Ipsilateral fixed dilated pupil (CN III compression), contralateral hemiplegia, Cushing's triad |
| Prognosis | Excellent if operated early — "talk and die" if missed |
3. 🟣 Subdural Hematoma (SDH)
| Type | Acute | Subacute | Chronic |
|---|---|---|---|
| Timing | < 72 hours | 3–21 days | > 21 days |
| Bleed source | Bridging cortical veins | Bridging veins | Bridging veins (slow ooze) |
| CT | Hyperdense crescent | Mixed density | Hypodense crescent |
| Cause | High-velocity trauma | Moderate trauma | Minor/trivial trauma (elderly, alcoholics) |
| Risk factors | Young adults | Any age | Elderly, anticoagulated, alcoholics, atrophy |
| Features | Rapid coma | Progressive decline | Fluctuating drowsiness, headache, dementia-like |
| Prognosis | Poor (brain injury underneath) | Moderate | Good if evacuated |
4. 🟡 Subarachnoid Hemorrhage (SAH) — Traumatic
| Feature | Detail |
|---|---|
| Source | Cortical vessel or contusion bleeding into subarachnoid space |
| CT | Hyperdensity in sulci, sylvian fissure, basal cisterns |
| Symptoms | "Worst headache of life," meningism, photophobia |
| Complication | Vasospasm (days 4–14), hydrocephalus |
| Differentiates from aneurysmal SAH | Traumatic SAH follows known mechanism; aneurysmal is spontaneous |
5. 🔵 Cerebral Contusion
| Feature | Detail |
|---|---|
| Definition | Bruising of brain parenchyma; heterogeneous injury |
| Mechanism | Direct impact (coup) or contrecoup at opposite pole |
| Common sites | Frontal and temporal poles (impact against bony prominences) |
| CT | Mixed hypo/hyperdense ("salt and pepper" pattern); petechial hemorrhages |
| Evolution | Can expand over 24–72 hours ("blossoming contusion") → re-image |
| Symptoms | Depends on location; frontal = personality/executive; temporal = memory |
6. ⚡ Diffuse Axonal Injury (DAI)
| Feature | Detail |
|---|---|
| Definition | Widespread tearing of axons due to rotational/shearing forces |
| Mechanism | Rotational acceleration-deceleration; no direct impact needed |
| Pathology | Axonal retraction balls; Wallerian degeneration; microhemorrhages |
| Sites | Grey-white matter junction, corpus callosum, brainstem, cerebellum |
| CT | Often normal or small petechial hemorrhages at grey-white junction |
| MRI | More sensitive — T2/FLAIR/SWI shows multiple punctate lesions |
| Clinical | Immediate, prolonged coma without mass lesion; worst prognosis |
| Grading | Grade 1: Lobar; Grade 2: Corpus callosum; Grade 3: Brainstem |
7. 💥 Concussion
| Feature | Detail |
|---|---|
| Definition | Transient functional disturbance of brain; no structural damage |
| LOC | Brief or absent (< 30 min) |
| CT/MRI | Normal |
| Symptoms | Headache, dizziness, confusion, amnesia, nausea, light/noise sensitivity |
| Post-concussion syndrome | Symptoms persist > 3 months |
| Return to play protocol | Stepwise graded return; no same-day return |
| CTE | Chronic Traumatic Encephalopathy — repeated concussions → tau protein accumulation |
5. CLINICAL FEATURES
5A. Initial Assessment — ATLS ABCDE Approach
A — Airway: Secure airway; C-spine immobilisation
B — Breathing: Ensure adequate ventilation; SpO₂ >95%
C — Circulation: IV access x2; BP maintenance; hemorrhage control
D — Disability: GCS, pupils, limb movement
E — Exposure: Full examination; log roll
5B. Symptoms by Severity
| Mild TBI | Moderate TBI | Severe TBI |
|---|---|---|
| Headache | Headache + vomiting | Coma (GCS ≤ 8) |
| Brief LOC or none | LOC 30 min–6 hrs | Prolonged unresponsiveness |
| Confusion, disorientation | Confusion, agitation | Decerebrate/decorticate posturing |
| Amnesia (retrograde/anterograde) | Focal neurological deficits | Absent brainstem reflexes |
| Nausea ± vomiting | Drowsiness progressing | Apneic; requiring ventilation |
| Dizziness, balance problems | Slurred speech | Fixed dilated pupils |
5C. Localizing Signs
| Finding | Significance |
|---|---|
| Fixed dilated ipsilateral pupil | Uncal herniation → CN III compression |
| Bilateral fixed dilated pupils | Bilateral herniation / brainstem failure |
| Contralateral hemiplegia | Cortical/capsular compression |
| Decorticate posturing (arms flexed, legs extended) | Diencephalon dysfunction |
| Decerebrate posturing (all extended) | Midbrain/pons dysfunction |
| Cushing's Triad | Raised ICP: ↑BP + ↓HR + irregular breathing |
| Papilloedema | Chronic raised ICP |
| Raccoon eyes / Battle's sign | Basal skull fracture |
| CN VI palsy | False localizing sign of raised ICP |
5D. Herniation Syndromes
| Type | Structures | Features |
|---|---|---|
| Subfalcine (cingulate) | Cingulate gyrus under falx | Leg weakness; ACA compression |
| Uncal (transtentorial) | Uncus through tentorium | CN III palsy, ipsilateral pupil dilation, contralateral hemiplegia, coma |
| Central (transtentorial) | Central diencephalon | Bilateral pupil changes, decorticate → decerebrate posturing |
| Tonsillar | Tonsils through foramen magnum | Respiratory arrest, neck stiffness, sudden death |
| Upward (transtentorial) | Cerebellum upward | Rare; posterior fossa mass |
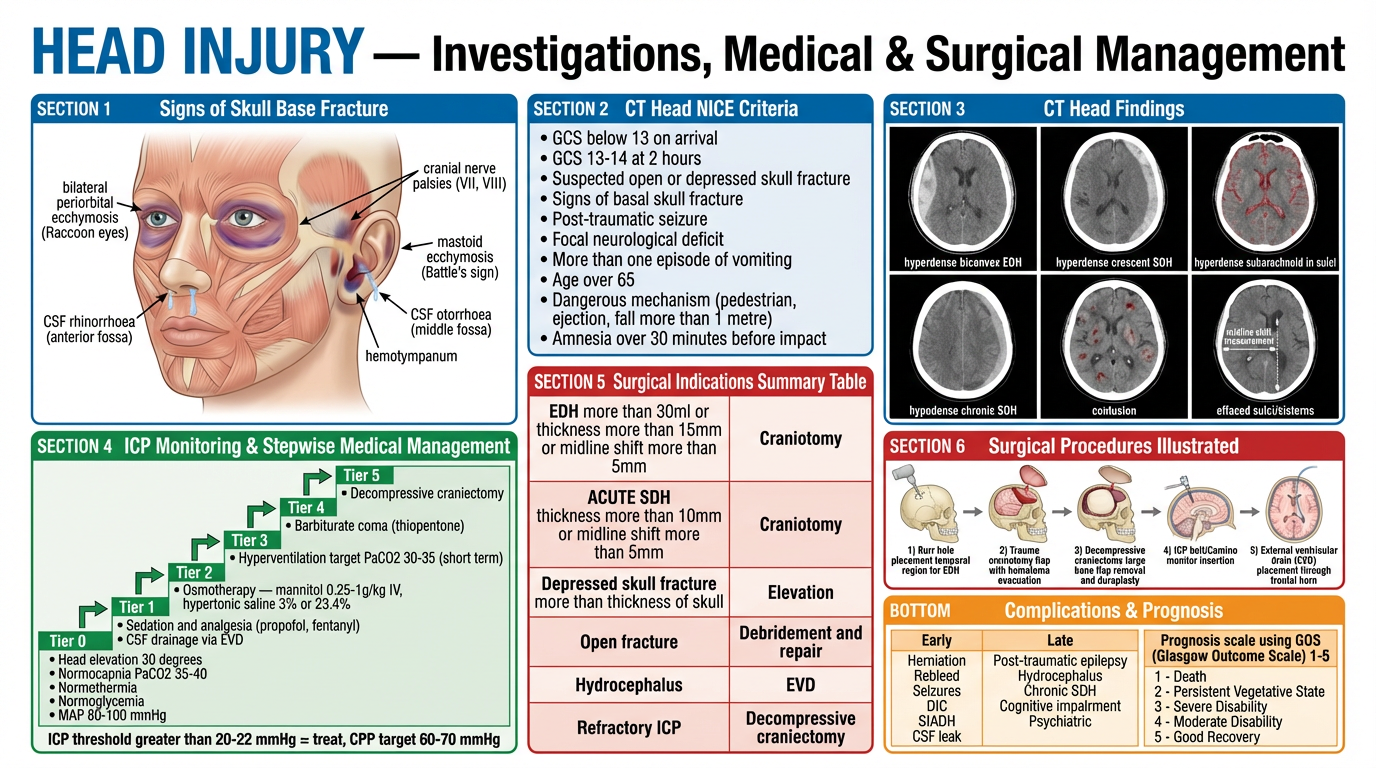
6. INVESTIGATIONS
6A. CT Head — NICE Criteria (Immediate CT within 1 hour)
| Criterion | Category |
|---|---|
| GCS < 13 on arrival | Consciousness |
| GCS 13–14 at 2 hours post-injury | Consciousness |
| Suspected open or depressed skull fracture | Structural |
| Any sign of basal skull fracture (Raccoon eyes, Battle's, CSF leak, hemotympanum) | Structural |
| Post-traumatic seizure | Neurological |
| Focal neurological deficit | Neurological |
| > 1 episode of vomiting | Neurological |
| Age > 65 years | Patient factor |
| Coagulopathy (warfarin, antiplatelet, bleeding disorder) | Patient factor |
| Dangerous mechanism (RTA pedestrian/cyclist, ejection from vehicle, fall > 1m or 5 stairs, high-speed impact) | Mechanism |
| Retrograde amnesia > 30 minutes before impact | Amnesia |
6B. CT Head Findings
| Finding | Appearance | Diagnosis |
|---|---|---|
| Biconvex hyperdensity | Does not cross sutures | Epidural hematoma (EDH) |
| Crescent hyperdensity | Crosses sutures, follows brain contour | Acute subdural hematoma |
| Crescent hypodensity | Crosses sutures | Chronic subdural hematoma |
| Mixed crescent density | Hyper + hypodense | Subacute SDH or rebleed into chronic SDH |
| Sulcal/cisternal hyperdensity | Basal cisterns, sylvian fissure | Subarachnoid hemorrhage |
| "Salt and pepper" petechiae | Frontal/temporal poles | Cerebral contusion |
| Grey-white junction hemorrhages | Corpus callosum, brainstem | Diffuse axonal injury |
| Midline shift | >5 mm = significant | Mass effect; surgical threshold |
| Effaced basal cisterns | Loss of perimesencephalic CSF spaces | Severe raised ICP; herniation imminent |
| Bone windows | Fracture line | Skull fracture |
6C. MRI Brain
| Indication | Advantage |
|---|---|
| Normal CT but persistent neurological deficit | Detects DAI, small contusions, posterior fossa injury |
| Subacute/chronic injury assessment | Better soft tissue contrast |
| Suspected non-accidental injury (NAI) in children | Detects subdural hygroma, retinal hemorrhages on ophthalmology |
| SWI (susceptibility-weighted imaging) | Best for detecting DAI microhemorrhages |
| DWI (diffusion-weighted imaging) | Detects early ischemic change |
6D. Blood Investigations
| Test | Purpose |
|---|---|
| FBC | Anemia, thrombocytopenia |
| Coagulation screen (PT, APTT, INR) | Coagulopathy; guide reversal agents |
| Blood glucose | Hypoglycemia mimics/worsens TBI |
| U&E, serum osmolality | Baseline; guide mannitol/hypertonic saline use |
| ABG | PaCO₂ monitoring; oxygenation |
| Group and save / crossmatch | Pre-operative preparation |
| Serum ethanol, toxicology | Exclude intoxication confounding GCS |
| Serum Na⁺ | SIADH / DI monitoring |
| Serum S100B protein | Biomarker; raised in significant TBI; can help triage mild TBI |
6E. Other Investigations
| Investigation | Indication |
|---|---|
| C-spine CT | All significant head injuries (must exclude concurrent C-spine fracture) |
| Chest/Abdo CT (trauma CT) | Polytrauma; exclude thoracic/abdominal injury |
| EEG | Post-traumatic seizures; non-convulsive status epilepticus |
| ICP monitoring | Severe TBI with GCS ≤ 8; abnormal CT |
| Transcranial Doppler (TCD) | Non-invasive assessment of cerebral blood flow velocity; vasospasm |
| Ophthalmology | Retinal hemorrhages (NAI), papilloedema |
| Cervical spine X-ray | If CT not immediately available |
7. MEDICAL MANAGEMENT
7A. Pre-hospital / Emergency Department
1. AIRWAY — Jaw thrust (not head tilt in trauma); C-spine collar
GCS ≤ 8 → RSI intubation (rapid sequence intubation)
Target SpO₂ > 95%; PaO₂ > 13 kPa
2. BREATHING — Controlled ventilation
Target PaCO₂ 35–40 mmHg (normocapnia)
Avoid hypocapnia (causes vasoconstriction → ischemia)
Avoid hypercapnia (causes vasodilation → raises ICP)
3. CIRCULATION — Two large-bore IVs
Target SBP > 100 mmHg (age 50-69) or > 110 mmHg (age 15-49, >70)
Avoid hypotension (SBP < 90 → doubles mortality)
Avoid hyponatraemia — use 0.9% NaCl not 5% dextrose
Reverse coagulopathy: Vit K, FFP, platelets, TXA
4. DISABILITY — GCS every 30 min; pupil assessment
Glucose control: target 6–10 mmol/L
5. ENVIRONMENT — Normothermia (temp > 38°C worsens outcome)
7B. ICU Management — Stepwise ICP Control
TIER 0 — Basic Neuroprotective Measures (All Severe TBI)
| Measure | Detail |
|---|---|
| Head elevation | 30° head-up, midline position (facilitates venous drainage) |
| Sedation and analgesia | Propofol 1–4 mg/kg/hr IV; Fentanyl/Morphine PRN (reduces agitation and ICP spikes) |
| Normocapnia | PaCO₂ 35–40 mmHg on ventilator |
| Normoxia | PaO₂ > 13 kPa; avoid hyperoxia |
| Normothermia | Paracetamol, cooling blankets; target 36–37°C |
| Normoglycaemia | Glucose 6–10 mmol/L; avoid hypo- and hyperglycemia |
| Seizure prophylaxis | Levetiracetam 500–1000 mg BD for 7 days (or phenytoin); prevents post-traumatic seizures |
| DVT prophylaxis | Sequential compression devices; LMWH after 48–72 hrs if no active bleed |
| Nutrition | Early enteral feeding (NG/NJ) within 24–48 hrs |
TIER 1 — First-Line ICP Reduction
| Measure | Detail |
|---|---|
| CSF drainage (EVD) | External ventricular drain — drains CSF to reduce ICP; monitors ICP directly |
| Increased sedation | Propofol infusion or midazolam for refractory agitation |
| Neuromuscular blockade | Atracurium/vecuronium — eliminates ICP spikes from coughing/suctioning |
TIER 2 — Osmotherapy
| Agent | Dose | Mechanism | Monitoring |
|---|---|---|---|
| Mannitol 20% | 0.25–1 g/kg IV bolus over 15–20 min | Osmotic gradient draws water from edematous brain into vasculature | Serum osmolality < 320 mOsm/kg; avoid if hypovolemic |
| Hypertonic Saline 3% | 250 mL IV over 20 min | Raises serum Na⁺ → draws water out of brain | Target Na⁺ 145–155 mmol/L; serum osmolality < 360 |
| Hypertonic Saline 23.4% | 30–60 mL IV bolus (central line) | Rapid, potent; used for acute herniation | Central line required; Na⁺ monitoring |
Hypertonic saline increasingly preferred over mannitol in hypotensive patients (mannitol causes diuresis → hypotension).
TIER 3 — Hyperventilation
| Measure | Detail |
|---|---|
| Targeted hyperventilation | Reduce PaCO₂ to 30–35 mmHg |
| Mechanism | CO₂ reduction → cerebral vasoconstriction → reduced CBV → ICP falls |
| Duration | Short-term only (< 2 hours) — reduces ischemia risk |
| Indication | Acute herniation while awaiting surgery |
| Monitoring | Jugular venous O₂ saturation (SjO₂) target 55–75%; brain tissue O₂ (PbtO₂) > 15 mmHg |
TIER 4 — Barbiturate Coma (Refractory ICP)
| Drug | Dose | Mechanism |
|---|---|---|
| Thiopentone (Thiopental) | Load 3–5 mg/kg, then infusion 3–5 mg/kg/hr | Reduces CMRO₂ (cerebral metabolic rate for O₂); suppresses EEG burst suppression pattern |
| Pentobarbital | Load 10 mg/kg over 30 min, then 1–4 mg/kg/hr | Same mechanism |
| Monitoring | Continuous EEG for burst suppression; ICP monitor; vasopressors for hypotension | |
| Risks | Hypotension, immunosuppression, prolonged sedation, hepatic/renal toxicity |
TIER 5 — Temperature Control (Therapeutic Hypothermia)
| Measure | Detail |
|---|---|
| Targeted Temperature Management (TTM) | Cooling to 35–36°C (mild hypothermia) |
| Mechanism | Reduces CMRO₂, excitotoxicity, inflammation |
| Evidence | Modest benefit in refractory ICP; avoid fever (>38°C doubles mortality) |
| Rewarming | Slow, 0.1–0.2°C/hour to avoid rebound ICP |
7C. Specific Drug Treatments
| Drug | Indication | Dose |
|---|---|---|
| Tranexamic Acid (TXA) | Within 3 hours of injury; reduces hemorrhagic progression | 1g IV over 10 min, then 1g over 8 hrs |
| Levetiracetam | Post-traumatic seizure prophylaxis (7 days) | 500–1000 mg BD IV/PO |
| Dexamethasone | ❌ CONTRAINDICATED in TBI — CRASH trial showed increased mortality | — |
| Vitamin K / FFP | Warfarin reversal before surgery | Vit K 5–10 mg IV; FFP 15 mL/kg |
| Idarucizumab | Dabigatran reversal | 5g IV |
| Andexanet alfa | Factor Xa inhibitor (rivaroxaban/apixaban) reversal | Weight-based protocol |
| Platelet transfusion | Antiplatelet reversal or thrombocytopenia | As per hematology guidance |
| Nimodipine | Traumatic SAH with vasospasm | 60 mg PO q4h for 21 days |
| Vasopressors | Maintain MAP 80–100 mmHg | Norepinephrine first-line |
8. SURGICAL MANAGEMENT
8A. Surgical Indications
| Pathology | Indication for Surgery | Operation |
|---|---|---|
| Epidural Hematoma (EDH) | Volume > 30 mL OR thickness > 15 mm OR midline shift > 5 mm OR GCS deterioration | Emergency craniotomy + hematoma evacuation |
| Acute Subdural Hematoma | Thickness > 10 mm OR midline shift > 5 mm OR GCS drop ≥ 2 points OR ICP > 20 mmHg | Emergency craniotomy ± decompressive craniectomy |
| Chronic Subdural Hematoma | Symptomatic (headache, confusion, focal deficits) | Burr hole craniotomy + drainage |
| Depressed Skull Fracture | Depression > full thickness of skull OR open (compound) fracture | Elevation + debridement ± dural repair |
| Open Skull Fracture | Any open fracture with dural breach | Debridement + dural repair + antibiotics |
| Intracerebral Hematoma | GCS deterioration OR progressive edema OR volume > 50 mL superficial | Craniotomy + hematoma evacuation |
| Posterior Fossa Hematoma | Any symptomatic posterior fossa hemorrhage (rapid brainstem compression) | Urgent suboccipital craniotomy |
| Hydrocephalus | Acute (post-traumatic) OR progressive ventricular enlargement | External ventricular drain (EVD) / VP shunt |
| Refractory raised ICP | ICP > 20–25 mmHg refractory to all medical measures | Decompressive craniectomy |
8B. Surgical Procedures in Detail
1. 🔧 Burr Hole Craniotomy
1. Position patient supine or lateral; shave and prep scalp
2. Linear scalp incision over temporal region (for EDH)
3. Drill burr hole with Hudson brace or high-speed drill
— Temporal burr hole: 2 cm anterior and superior to tragus
4. Dura cauterized and incised
5. Clot evacuated by suction and irrigation
6. Hemostasis; drain placed if needed
7. Closure in layers
2. 🏥 Trauma Craniotomy (Hematoma Evacuation)
1. General anesthesia; head pinned in Mayfield clamp
2. Horseshoe/trauma flap scalp incision
— Temporoparietal "trauma flap" for temporal EDH/SDH
3. Burr holes placed at corners of planned bone flap
4. Craniotome used to connect burr holes — bone flap elevated
5. Dura opened (cruciate or semicircular incision)
6. Hematoma evacuated:
— EDH: Liquid + clot suctioned; bleeding vessel coagulated
— SDH: Clot irrigated out with copious warm saline
7. Hemostasis with bipolar diathermy + Surgicel
8. Dura closed (primarily or with patch graft)
9. Bone flap replaced and fixed with titanium plates/screws
10. Wound closure in layers + drain
3. 🧠 Decompressive Craniectomy
1. Large frontotemporo-parietal skin flap
2. Large bone flap (≥12 cm diameter) removed
— Creates space for swollen brain to expand outward
3. DURAPLASTY: Dura opened widely + patch graft (pericranium,
synthetic dura) sewn in to expand dural volume by 20-25%
— Critical step: dura must be expanded, not just skin
4. Bone flap stored: cryopreserved (–80°C) OR abdominal
subcutaneous pocket (to maintain viability)
5. Staged CRANIOPLASTY later (6–12 weeks) when brain swelling
resolved: bone flap replaced or titanium mesh prosthesis
- Sinking skin flap syndrome (paradoxical herniation through defect)
- Hygroma formation
- Bone flap resorption
- CSF leak
- Infection
4. 📡 Intracranial Pressure (ICP) Monitoring
- Severe TBI (GCS 3–8) + abnormal CT scan
- Severe TBI + normal CT + ≥2 of: age >40, SBP <90, motor posturing
| Type | Placement | Accuracy | Features |
|---|---|---|---|
| Intraventricular EVD (Gold standard) | Frontal horn of lateral ventricle | Best | Allows therapeutic CSF drainage; can be recalibrated |
| Intraparenchymal bolt (Camino/Codman) | Brain parenchyma | Good | Simple insertion; cannot drain CSF; drifts |
| Subdural/Epidural | Below/above dura | Less accurate | Less invasive |
- A waves (plateau waves): ICP 50–100 mmHg for 5–20 min → critical; impending herniation
- B waves: ICP 20–50 mmHg for 0.5–2 min → pathological; raised ICP
- C waves: Minor oscillations; physiological
5. 💧 External Ventricular Drain (EVD)
1. Kocher's point: 1 cm anterior to coronal suture,
2.5 cm lateral to midline (mid-pupillary line)
2. Burr hole at Kocher's point
3. Dura punctured; catheter directed perpendicular to brain
surface, aimed at medial canthus of ipsilateral eye
and ipsilateral tragus (toward foramen of Monro)
4. Catheter advanced 5–7 cm → CSF flows
5. Tunneled subcutaneously; connected to closed drainage system
6. Zeroed at level of foramen of Monro (tragus)
6. 🦴 Elevation of Depressed Skull Fracture
- Depression > full skull thickness
- Open (compound) fracture
- Underlying hematoma
- Cosmetic deformity
- Dural breach / pneumocephalus
1. Elliptical scalp incision around fracture
2. Burr hole placed adjacent (away from fracture)
3. Fragments carefully elevated using dissector/periosteal elevator
4. Dura inspected; repaired if lacerated
5. Thorough debridement of contaminated wounds
6. Irrigation with antibiotic solution
7. Bone fragments replaced and fixed if non-contaminated
8. Open/contaminated fractures: fragments discarded;
cranioplasty deferred 6 months
9. IV antibiotics: Co-amoxiclav 1.2g TDS x 5 days
9. COMPLICATIONS
Early Complications (Hours–Days)
| Complication | Management |
|---|---|
| Transtentorial herniation | Emergency surgery ± mannitol |
| Rebleed / hematoma expansion | Repeat CT; surgical re-evacuation |
| Post-traumatic seizures | IV lorazepam; AED prophylaxis |
| SIADH (hyponatremia) | Fluid restriction; hypertonic saline if severe |
| Diabetes insipidus (DI) | IV desmopressin; monitor Na⁺ |
| Neurogenic pulmonary edema | Ventilatory support; diuretics |
| Coagulopathy (DIC) | FFP, cryoprecipitate, platelets |
| CSF leak / meningitis | Prophylactic antibiotics controversial; surgical repair if persistent |
Late Complications (Weeks–Months)
| Complication | Notes |
|---|---|
| Post-traumatic epilepsy | Up to 25% with severe TBI; long-term AEDs |
| Chronic subdural hematoma | Re-accumulation; requires re-drainage |
| Normal pressure hydrocephalus | Ventriculomegaly post-TBI; VP shunt |
| Cognitive impairment | Memory, attention, executive function deficits |
| Psychiatric disorders | Depression, PTSD, personality change |
| Vegetative state / Minimally conscious state | Specialist rehabilitation |
| Chronic Traumatic Encephalopathy (CTE) | Repeated TBI; tau protein accumulation; dementia, parkinsonism |
| Pituitary dysfunction | Hypopituitarism in 25–50% severe TBI |
10. PROGNOSIS — Glasgow Outcome Scale (GOS)
| Score | Category | Description |
|---|---|---|
| GOS 5 | Good recovery | Minor deficits; normal life |
| GOS 4 | Moderate disability | Disabled but independent |
| GOS 3 | Severe disability | Dependent on others for daily care |
| GOS 2 | Vegetative state | No meaningful awareness |
| GOS 1 | Death | — |
- GCS ≤ 3 on admission (especially motor score = 1)
- Bilateral fixed dilated pupils
- Age > 60 years
- Hypotension + hypoxia on admission
- CT: effaced basal cisterns, bilateral hemorrhages, SAH
- DAI Grade 3 (brainstem involvement)
- Prolonged ICP > 40 mmHg
11. SUMMARY MANAGEMENT ALGORITHM
TRAUMA CALL — Head Injury Patient
↓
ATLS: Airway (C-spine) → Breathing → Circulation
↓
GCS Assessment + Pupil Exam
↓
┌─────────────────────────────────┐
│ GCS 13-15 (Mild) │
│ Observe; CT if NICE criteria │
│ Discharge if CT normal + well │
└─────────────────────────────────┘
↓
┌─────────────────────────────────┐
│ GCS 9-12 (Moderate) │
│ CT HEAD + C-spine │
│ Admit to HDU; serial GCS │
│ Neurosurgical referral │
└─────────────────────────────────┘
↓
┌─────────────────────────────────┐
│ GCS ≤ 8 (Severe) │
│ RSI Intubation │
│ CT HEAD immediately │
│ Neurosurgical ICU │
│ ICP monitoring │
└─────────────────────────────────┘
↓
CT shows mass lesion + mass effect?
↓ ↓
YES NO
↓ ↓
EMERGENCY Medical ICP
SURGERY Management
Craniotomy/ (Tiered protocol)
Burr hole Tier 0→1→2→3→4
↓ ↓
ICP refractory to all measures?
↓
DECOMPRESSIVE CRANIECTOMY
↓
ICU: Neuroprotection + Rehabilitation
- Bailey & Love's Short Practice of Surgery, 28th Ed. (pp. 381, 384)
- Harrison's Principles of Internal Medicine, 21st Ed. (p. 12563)
- Brain Trauma Foundation (BTF) Guidelines for Severe TBI, 4th Ed. (2016)
- NICE Head Injury Guidelines CG176 (2014, updated 2023)
- CRASH-3 Trial — Tranexamic acid in TBI (Lancet 2019)
- DECRA Trial — Decompressive craniectomy (NEJM 2011)
- RESCUEicp Trial (NEJM 2016)
3. Explain Transient ischemic attack. Describe the acute management of stroke 4. List the causes of stroke. Compare ischemic &hemoragic stroke. 5. Define Stroke. List the risk factors and early warning signs of stroke. Add a note on acute management of stroke 6. Enumerate the risk factors for cerebrovascular accidents. Discuss the management of haemorrhagic stroke. 7. Define and classify stroke. Describe Transient ischemic stroke and its clinical significance in deatil strustured answer
stroke definition classification ischemic hemorrhagic types causes risk factors
transient ischemic attack TIA definition ABCD2 score clinical features management significance
acute ischemic stroke management thrombolysis tPA alteplase thrombectomy time window
hemorrhagic stroke intracerebral hemorrhage subarachnoid management blood pressure surgery
ischemic stroke CT MRI brain infarction hemorrhagic stroke comparison
stroke FAST warning signs cerebrovascular accident brain anatomy territories
🧠 STROKE & TIA — Complete Structured Clinical Answer
Covering Questions 3, 4, 5, 6 & 7
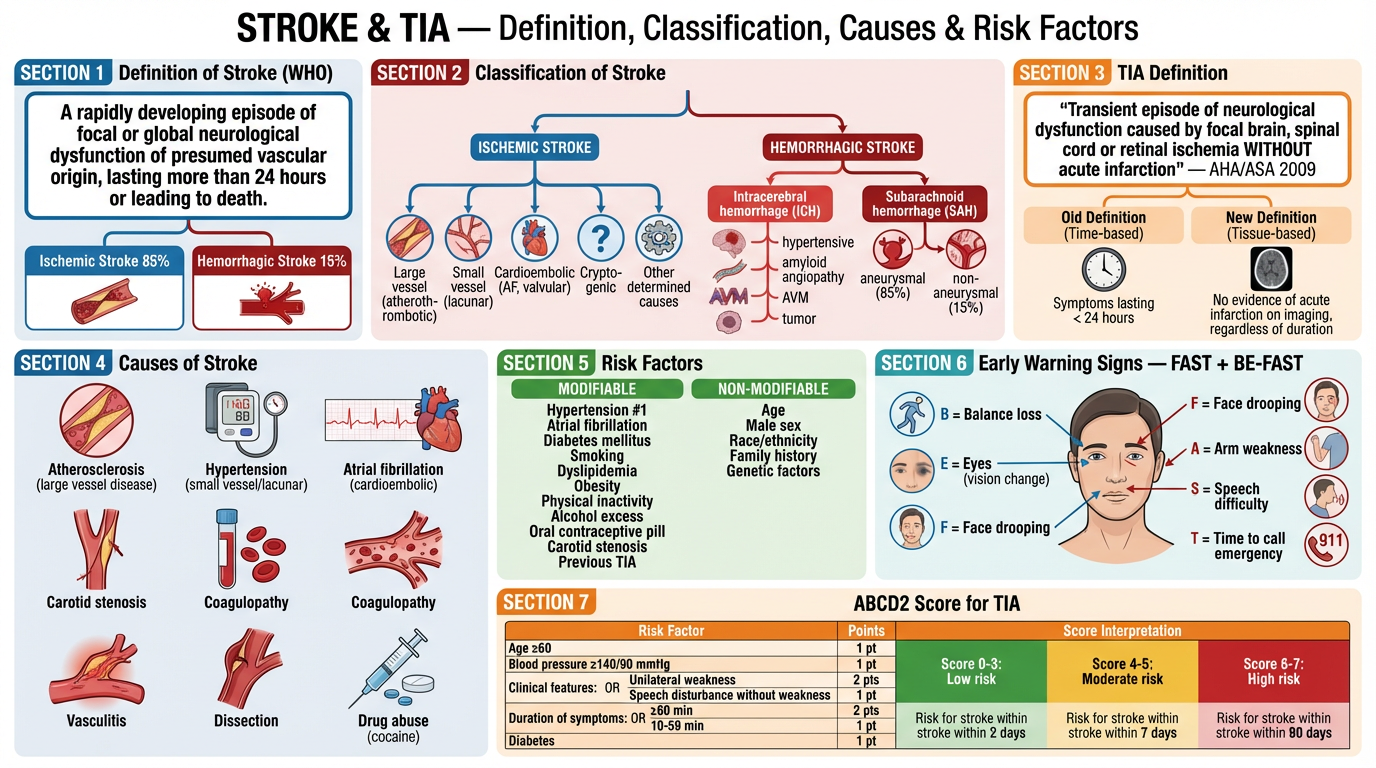
SECTION A: DEFINITION OF STROKE
"A rapidly developing episode of focal or global neurological dysfunction of vascular origin, lasting more than 24 hours or leading to death, with no apparent non-vascular cause."
"An infarction of the central nervous system (brain, spinal cord, or retinal cells) attributable to ischemia, based on neuropathological, neuroimaging, or clinical evidence."
- Duration > 24 hours OR death (if < 24 hours → TIA if no infarction on imaging)
- Must exclude metabolic causes (hypoglycemia, Todd's paresis, migraine, tumor)
- Also called Cerebrovascular Accident (CVA)
SECTION B: CLASSIFICATION OF STROKE
B1. By Pathological Type
| Type | Frequency | Mechanism |
|---|---|---|
| Ischemic Stroke | 85% | Occlusion of cerebral artery → infarction |
| Hemorrhagic Stroke | 15% | Rupture of cerebral vessel → blood in/around brain |
B2. Ischemic Stroke — TOAST Classification
| Subtype | Mechanism | Features |
|---|---|---|
| Large artery atherothrombosis | Atherosclerotic plaque → thrombosis or artery-to-artery embolism | Carotid/vertebrobasilar stenosis; TIA precedes |
| Cardioembolic | Embolus from heart → cerebral artery | Atrial fibrillation (most common), valvular disease, mural thrombus post-MI, endocarditis |
| Small vessel (Lacunar) | Lipohyalinosis of perforating arteries → lacunar infarct | Hypertension, diabetes; pure motor/sensory stroke; no cortical signs |
| Cryptogenic | No identifiable cause after full workup | ~30% of ischemic strokes; PFO may be implicated |
| Other determined | Vasculitis, dissection, coagulopathy, drugs | Younger patients; specific investigations required |
B3. Hemorrhagic Stroke
| Subtype | Frequency | Key Features |
|---|---|---|
| Intracerebral hemorrhage (ICH) | 10% of all strokes | Bleeding into brain parenchyma; basal ganglia most common site |
| Subarachnoid hemorrhage (SAH) | 5% of all strokes | Bleeding into subarachnoid space; "thunderclap headache"; aneurysm in 85% |
B4. By Vascular Territory
| Territory | Artery | Deficits |
|---|---|---|
| Anterior circulation | ICA, MCA, ACA | Hemiplegia, hemisensory loss, aphasia (dominant), neglect (non-dominant), homonymous hemianopia |
| Posterior circulation | Vertebral, basilar, PCA, PICA | Diplopia, vertigo, ataxia, dysarthria, dysphagia, crossed signs, locked-in syndrome |
| Lacunar | Perforating arteries | Pure motor hemiplegia, pure sensory stroke, ataxic hemiparesis, dysarthria-clumsy hand |
B5. Oxford (Bamford) Clinical Classification
| Type | Abbrev. | Criteria | Prognosis |
|---|---|---|---|
| Total Anterior Circulation Infarct | TACI | All 3: hemimotor/sensory + homonymous hemianopia + higher cortical dysfunction | Worst |
| Partial Anterior Circulation Infarct | PACI | 2 of 3, or higher cortical dysfunction alone | Moderate |
| Lacunar Infarct | LACI | Pure motor, pure sensory, sensorimotor, or ataxic hemiparesis | Best |
| Posterior Circulation Infarct | POCI | Ipsilateral CN palsy + contralateral motor/sensory, OR cerebellar/brainstem signs | Variable |
SECTION C: CAUSES OF STROKE
Ischemic Causes
| Category | Specific Causes |
|---|---|
| Atherosclerosis | Large vessel disease; carotid stenosis; vertebrobasilar disease |
| Cardiac embolism | Atrial fibrillation (most common), acute MI/mural thrombus, dilated cardiomyopathy, valvular disease (mitral stenosis, prosthetic valve), infective endocarditis, patent foramen ovale |
| Small vessel disease | Hypertension → lipohyalinosis; diabetes mellitus |
| Arterial dissection | Carotid or vertebral dissection (young patients, trauma, neck manipulation) |
| Coagulopathy | Antiphospholipid syndrome, Factor V Leiden, protein C/S deficiency, polycythemia, thrombocytosis |
| Vasculitis | Primary CNS vasculitis, systemic lupus, Takayasu's, giant cell arteritis |
| Drug-induced | Cocaine (vasospasm), amphetamines, OCP (thrombosis) |
| Rare | Fabry disease, MELAS (mitochondrial), sickle cell disease, CADASIL |
Hemorrhagic Causes
| Category | Specific Causes |
|---|---|
| Hypertension | Most common cause of ICH; affects basal ganglia, thalamus, pons, cerebellum |
| Cerebral amyloid angiopathy | Elderly; lobar hemorrhages; recurrent; β-amyloid deposits in vessel walls (Harrison's p. 12293) |
| Arteriovenous malformation (AVM) | Young patients; recurrent hemorrhage |
| Aneurysm rupture | Berry aneurysm → SAH; commonest at Circle of Willis bifurcations |
| Anticoagulation | Warfarin, NOACs, heparin |
| Thrombolysis complication | tPA-related ICH |
| Tumour | Metastases (melanoma, renal, choriocarcinoma) → haemorrhagic |
| Venous sinus thrombosis | Haemorrhagic venous infarction |
| Cocaine/sympathomimetics | Acute severe hypertension |
SECTION D: RISK FACTORS FOR STROKE / CVA
Modifiable Risk Factors
| Risk Factor | Relative Risk | Notes |
|---|---|---|
| Hypertension | ×4–6 | Single most important modifiable risk factor; target BP < 130/80 |
| Atrial Fibrillation | ×5 | Cardioembolic; anticoagulation reduces risk by 65% |
| Smoking | ×2–3 | Doubles risk; cessation reduces risk within 5 years |
| Diabetes Mellitus | ×2–3 | Accelerates atherosclerosis; tight control reduces risk |
| Dyslipidaemia | ×2 | High LDL; statins reduce stroke risk by ~30% |
| Obesity / Physical inactivity | ×1.5–2 | BMI > 30; exercise reduces risk |
| Excess alcohol | ×2–3 | >14 units/week; abstinence/reduction beneficial |
| OCP / HRT | ×2–3 | Especially in smokers or those with migraine with aura |
| Carotid stenosis | ×2–4 | >70% stenosis; endarterectomy reduces risk |
| Previous TIA | ×10 in first 48 hrs | Strongest short-term predictor of stroke |
| Obstructive sleep apnoea | ×2 | Treat with CPAP |
| Cocaine / stimulants | High | Vasospasm + acute hypertension |
Non-Modifiable Risk Factors
| Risk Factor | Notes |
|---|---|
| Age | Risk doubles every decade after age 55 |
| Male sex | Men have 25% higher lifetime risk |
| Race/Ethnicity | Black and South Asian populations have higher risk (hypertension, diabetes) |
| Family history | First-degree relative with stroke increases risk ×3 |
| Genetic syndromes | CADASIL, Fabry disease, sickle cell, MELAS |
| Previous stroke | 30–43% recurrence risk within 5 years |
SECTION E: EARLY WARNING SIGNS OF STROKE
BE-FAST Acronym
| Letter | Sign | Clinical Meaning |
|---|---|---|
| B | Balance | Sudden loss of balance or coordination |
| E | Eyes | Sudden vision change (loss, double vision, hemianopia) |
| F | Face | Sudden facial drooping (ask patient to smile) |
| A | Arms | Sudden arm weakness (ask to raise both arms — one drifts) |
| S | Speech | Sudden slurred, confused, or absent speech |
| T | Time | Time to call emergency services IMMEDIATELY |
Other Early Warning Features
- Sudden severe headache with no known cause ("worst headache of life" → SAH)
- Sudden numbness or tingling in face, arm, or leg (especially one-sided)
- Sudden confusion or difficulty understanding
- Sudden dizziness, vertigo, or unsteady gait
- Sudden diplopia (double vision)
- Transient monocular blindness (amaurosis fugax) → carotid TIA
SECTION F: TRANSIENT ISCHEMIC ATTACK (TIA)
Definition
AHA/ASA 2009 (Tissue-Based Definition): "A transient episode of neurological dysfunction caused by focal brain, spinal cord, or retinal ischemia, WITHOUT acute infarction."
- Old time-based definition: symptoms resolve within 24 hours
- New definition: tissue-based — symptoms may be brief but imaging may show infarction
- Most TIAs last < 1–2 hours (AHA/ASA Guidelines)
- If symptoms last > 24 hours with NO infarction on imaging → still TIA by new definition
- If imaging unavailable and symptoms last > 24 hours → classified as clinical stroke
Clinical Features of TIA
| Vascular Territory | Symptoms |
|---|---|
| Carotid (Anterior) | Contralateral hemiparesis, hemisensory loss; aphasia (dominant hemisphere); amaurosis fugax (ipsilateral monocular blindness — retinal TIA) |
| Vertebrobasilar (Posterior) | Diplopia, vertigo, dysarthria, dysphagia, ataxia, drop attacks, bilateral weakness/sensory loss |
ABCD² Score — Risk Stratification After TIA
| Feature | Criteria | Score |
|---|---|---|
| A — Age | ≥ 60 years | 1 |
| B — Blood Pressure | Systolic ≥ 140 mmHg OR diastolic ≥ 90 mmHg | 1 |
| C — Clinical features | Unilateral weakness | 2 |
| Speech disturbance without weakness | 1 | |
| Other | 0 | |
| D — Duration | ≥ 60 minutes | 2 |
| 10–59 minutes | 1 | |
| < 10 minutes | 0 | |
| D² — Diabetes | Present | 1 |
| Total | Max = 7 |
| Score | Risk Category | 2-Day Stroke Risk |
|---|---|---|
| 0–3 | Low | ~1% |
| 4–5 | Moderate | ~4% |
| 6–7 | High | ~8–12% |
Clinical Significance of TIA
- Short-term stroke risk: 10–20% risk of stroke within 90 days; highest in first 48 hours (up to 10%)
- "Stroke waiting to happen": TIA patients who present early and are managed aggressively have 80% reduction in subsequent stroke risk
- SOS-TIA Clinic data: Urgent same-day evaluation and treatment can reduce 90-day stroke risk from ~10% to ~2%
- Crescendo TIA: Multiple TIAs in rapid succession = imminent stroke; requires emergency hospital admission
- Investigation window: Opportunity to find and treat the underlying cause before disabling stroke occurs
Investigations After TIA
| Investigation | Purpose |
|---|---|
| MRI brain with DWI (preferred) | Detect acute infarction (rules out "true TIA" vs minor stroke) |
| CT brain | Exclude hemorrhage if MRI unavailable |
| 12-lead ECG | Detect atrial fibrillation |
| 24/48-hour Holter monitor | Paroxysmal AF (missed on ECG) |
| Carotid Doppler USS | Carotid stenosis (anterior circulation TIA) |
| Echocardiography | Cardiac source: thrombus, valvular disease, PFO |
| FBC, ESR, CRP | Vasculitis, polycythaemia |
| Coagulation screen | Coagulopathy, antiphospholipid syndrome |
| Fasting lipids, glucose, HbA1c | Cardiovascular risk factors |
| CT/MR Angiography | Intracranial stenosis, dissection, vertebrobasilar disease |
| Bubble echocardiography | Patent foramen ovale (PFO) — cryptogenic TIA in young |
Management of TIA
URGENT SAME-DAY ASSESSMENT (ABCD² ≥ 4 or multiple TIAs)
↓
IMMEDIATE ANTIPLATELET THERAPY:
Aspirin 300 mg STAT (loading dose)
then Aspirin 75 mg OD + Clopidogrel 75 mg OD (dual antiplatelet x 21 days)
— POINT study: dual antiplatelet ↓ early recurrence by 25%
↓
If AF confirmed → ANTICOAGULATION:
DOAC (apixaban/rivaroxaban/dabigatran) or Warfarin
↓
STATIN: Atorvastatin 80 mg OD (started same day)
↓
ANTIHYPERTENSIVE: If SBP > 130 → treat (after acute phase)
ACE inhibitor + thiazide (PROGRESS trial)
↓
CAROTID ENDARTERECTOMY: If 70–99% ipsilateral carotid stenosis
— Within 2 weeks of TIA (NASCET, ECST trials)
↓
LIFESTYLE: Smoking cessation, diet, exercise, alcohol reduction
SECTION G: ISCHEMIC vs HEMORRHAGIC STROKE — COMPARISON
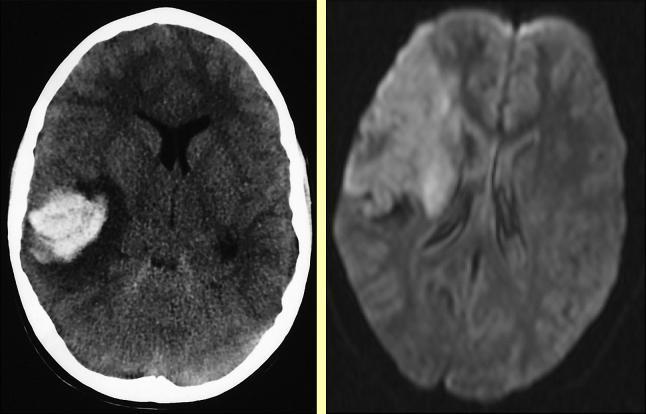
| Feature | Ischemic Stroke | Hemorrhagic Stroke |
|---|---|---|
| Frequency | 85% | 15% |
| Mechanism | Arterial occlusion → infarction | Vessel rupture → blood in/around brain |
| Onset | Sudden; may be stepwise | Sudden; often during activity/exertion |
| Preceding TIA | Common (20–30%) | Rare |
| Headache | Mild or absent | Severe ("worst headache of life" in SAH) |
| Vomiting | Uncommon | Common |
| Conscious level | Usually preserved initially | Often impaired (ICH/SAH) |
| Meningism | Absent | Present in SAH (neck stiffness, photophobia) |
| Seizures | Less common acutely | More common at onset |
| Blood pressure | May be normal or mildly elevated | Usually markedly elevated (hypertensive ICH) |
| CT appearance | Hypodense (dark) area — may be normal in first 6 hrs | Hyperdense (bright) blood collection |
| MRI DWI | Hyperintense (restricted diffusion) — positive within 30 min | Variable; hemoglobin signal changes over time |
| CSF | Normal (xanthochromia absent) | Xanthochromia / blood-stained CSF (SAH) |
| Thrombolysis | ✅ Eligible (if no contraindications) | ❌ ABSOLUTELY CONTRAINDICATED |
| Anticoagulation | Delayed (24–48 hrs for non-AF; earlier for cardioembolic) | ❌ Contraindicated acutely; reverse existing anticoagulation |
| Antiplatelet | ✅ Aspirin 300 mg within 24–48 hrs | ❌ Avoid acutely |
| Surgical treatment | Mechanical thrombectomy for large vessel occlusion | Surgical clipping/coiling (SAH); haematoma evacuation (select ICH) |
| BP management | Permissive hypertension (allow up to 220/120 unless thrombolysis) | Aggressive reduction (target SBP < 140 mmHg within 1 hr) |
| Prognosis | Better overall; large MCA infarcts have 30-day mortality ~20% | Worse; 30-day mortality 30–50% for ICH; 25–35% for SAH |
SECTION H: ACUTE MANAGEMENT OF STROKE
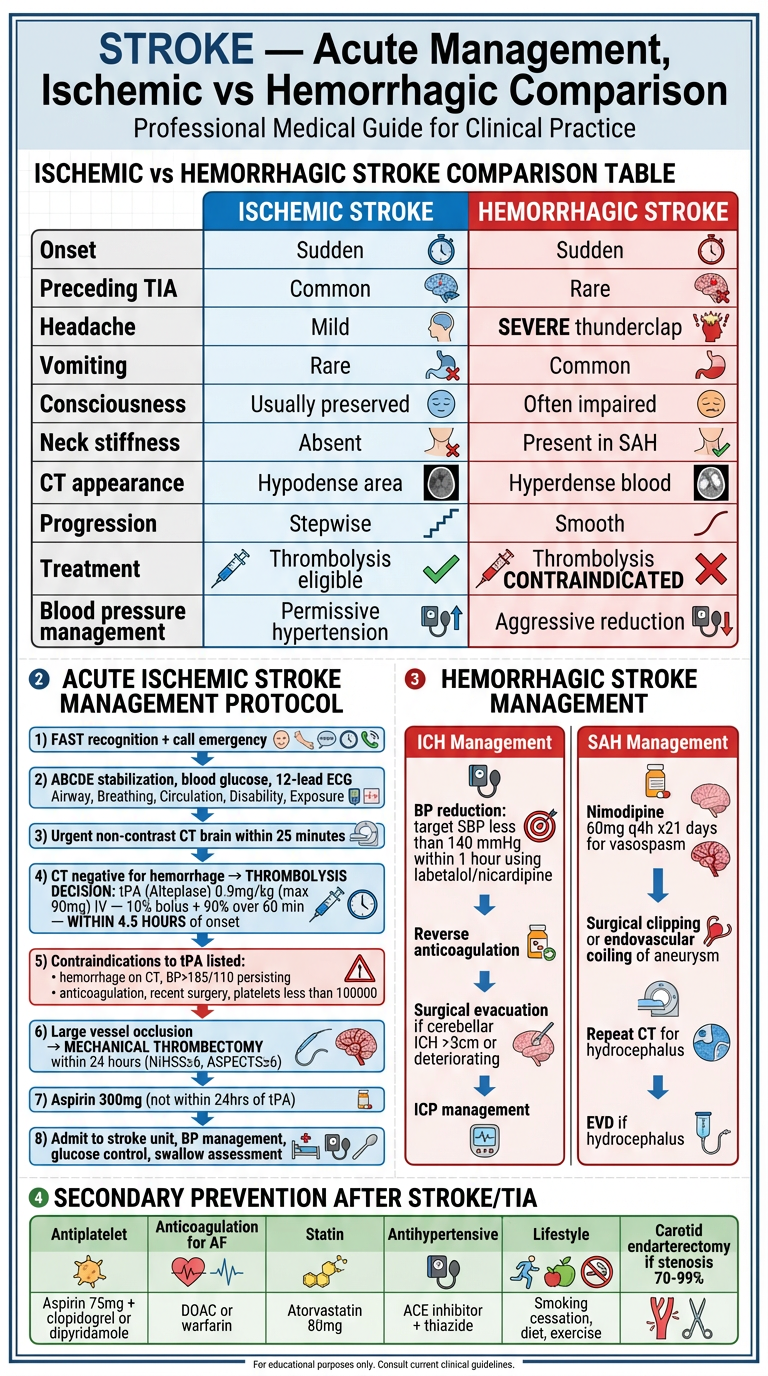
H1. Pre-Hospital Phase
1. RECOGNITION: BE-FAST symptom identification
2. CALL emergency services IMMEDIATELY
3. NOTE exact time of symptom onset (or "last seen well")
4. DO NOT give aspirin until hemorrhage excluded by CT
5. EMS: Rapid transfer to stroke-capable hospital
"Time is brain" — 1.9 million neurons lost per minute
H2. Emergency Department — First 60 Minutes ("Golden Hour")
0–10 min: Stroke team alert; ABCDE assessment
IV access x2; bloods (FBC, U&E, coag, glucose, group & save)
12-lead ECG
Pulse oximetry; O₂ if SpO₂ < 95%
0–25 min: Urgent non-contrast CT brain
(exclude hemorrhage before any antithrombotic therapy)
0–45 min: CT reviewed by stroke physician/radiologist
0–60 min: TREATMENT DECISION (door-to-needle < 60 min target)
H3. Acute Ischemic Stroke Management
Step 1 — Stabilization
| Parameter | Target | Action |
|---|---|---|
| Airway | Patent | Intubate if GCS ≤ 8 or airway at risk |
| Oxygenation | SpO₂ ≥ 95% | Supplemental O₂ only if hypoxic |
| Blood Pressure | < 185/110 for thrombolysis; allow up to 220/120 otherwise | IV labetalol, nicardipine if > 185/110 and thrombolysis planned |
| Blood glucose | 4–11 mmol/L | IV insulin infusion if > 11; glucose if < 4 |
| Temperature | Normothermia | Paracetamol + cooling if febrile |
| Cardiac monitoring | Continuous | Detect AF, arrhythmia |
Step 2 — IV Thrombolysis (Alteplase / tPA)
| Parameter | Detail |
|---|---|
| Drug | Alteplase (recombinant tPA) |
| Dose | 0.9 mg/kg IV (maximum 90 mg); 10% as IV bolus over 1 min; remaining 90% infused over 60 min |
| Time window | Within 4.5 hours of symptom onset (extended to 9 hrs in selected patients with mismatch on perfusion imaging) |
| Benefit | NNT = 7 for favourable outcome; greatest benefit if given within 90 minutes |
| Absolute | Relative |
|---|---|
| Hemorrhage on CT | Minor, rapidly improving symptoms |
| BP > 185/110 (uncontrolled) | Pregnancy |
| Active internal bleeding | Recent seizure at onset |
| Platelets < 100,000 | INR 1.7–3.0 (discuss with senior) |
| INR > 3.0 or heparin within 48 hrs | Prior stroke within 3 months |
| Recent major surgery (< 14 days) | Blood glucose < 2.7 or > 22 mmol/L |
| Recent serious head trauma (< 3 months) | Large established infarct (ASPECTS ≤ 5) |
| History of intracranial hemorrhage | Age < 18 years |
| Intracranial neoplasm/AVM |
Step 3 — Mechanical Thrombectomy (Endovascular Treatment)
| Parameter | Detail |
|---|---|
| Indication | Large vessel occlusion (ICA, M1/M2 MCA, basilar) + NIHSS ≥ 6 |
| Time window | 0–24 hours (extended window with perfusion imaging: DAWN and DEFUSE-3 trials) |
| Standard window | Within 6 hours of onset |
| Extended window | 6–24 hours with CT perfusion/MR perfusion showing salvageable penumbra |
| Technique | Stent retriever (Solitaire, Trevo) or aspiration catheter via transfemoral approach |
| Benefit | HERMES meta-analysis: NNT = 2.6 for functional independence; reduces disability even without tPA |
| Combined | tPA + thrombectomy superior to either alone in eligible patients |
Step 4 — Antiplatelet Therapy
| Drug | Dose | Timing |
|---|---|---|
| Aspirin | 300 mg loading dose | Within 24–48 hours of onset (NOT within 24 hrs of tPA) |
| Aspirin 75 mg + Clopidogrel 75 mg | Dual antiplatelet | POINT/CHANCE trials: 21 days dual, then single antiplatelet long-term |
| Clopidogrel 75 mg alone | Alternative if aspirin intolerant | Long-term |
Step 5 — Stroke Unit Admission
- Nurse-led neurological observation (GCS, pupils, limb power) q1–4h
- Dysphagia screening before any oral intake (SALT referral)
- Physiotherapy: early mobilization within 24–48 hours
- Occupational therapy, speech and language therapy
- DVT prophylaxis: compression stockings; LMWH after 24–48 hours
- Bladder/bowel care; pressure area care
- Early rehabilitation planning
H4. Acute Hemorrhagic Stroke Management
Intracerebral Hemorrhage (ICH)
| Priority | Action |
|---|---|
| Airway | Intubate if GCS ≤ 8 |
| Stop anticoagulation | IMMEDIATELY stop all anticoagulants/antiplatelets |
| Reverse anticoagulation | See reversal table below |
| Blood pressure control | Target SBP < 140 mmHg within 1 hour (INTERACT2, ATACH-2 trials); IV labetalol 5–20 mg IV PRN or nicardipine infusion 5–15 mg/hr |
| ICP management | Head elevation 30°; mannitol 0.25–1 g/kg if herniation signs; neurosurgical consult |
| Seizure treatment | IV lorazepam if seizing; prophylactic AED if cortical ICH |
| Glucose control | Target 6–10 mmol/L |
| Avoid fever | Paracetamol; cooling |
| Anticoagulant | Reversal Agent |
|---|---|
| Warfarin | Vitamin K 10 mg IV slow + Prothrombin Complex Concentrate (PCC) 25–50 units/kg |
| Dabigatran | Idarucizumab 5g IV (Praxbind) |
| Rivaroxaban/Apixaban | Andexanet alfa OR PCC 50 units/kg |
| Heparin | Protamine sulphate 1 mg per 100 units heparin IV |
| LMWH | Protamine (partial reversal); andexanet alfa |
| Indication | Operation |
|---|---|
| Cerebellar ICH > 3 cm OR deteriorating | Urgent posterior fossa craniotomy + evacuation — most evidence for surgical benefit |
| Lobar ICH with mass effect + GCS 9–14 | Consider craniotomy (STICH II trial: borderline benefit for superficial lobar ICH) |
| Deep ICH (basal ganglia, thalamus) | Generally NO surgery (STICH I trial: no benefit); medical management |
| Hydrocephalus post-ICH | External ventricular drain (EVD) |
| Young patient with lobar ICH and underlying AVM | AVM resection / radiosurgery after stabilization |
| Minimally invasive surgery | MISTIE III trial: clot aspiration + alteplase instillation — reduced clot volume but no functional benefit |
Subarachnoid Hemorrhage (SAH) Management
| Priority | Action |
|---|---|
| Stabilization | ABCDE; ICU admission; strict bed rest |
| Analgesia | IV morphine / codeine for headache |
| BP control | Target SBP < 160 mmHg before aneurysm secured |
| Nimodipine | 60 mg PO/NG every 4 hours for 21 days — reduces cerebral vasospasm and delayed ischemic neurological deficit (DIND) |
| Fluids | IV isotonic fluids; maintain euvolemia; avoid hypovolemia (worsens vasospasm) |
| Aneurysm treatment | Urgent CTA or digital subtraction angiography (DSA) to identify aneurysm |
| Method | Technique | Indication |
|---|---|---|
| Endovascular coiling (ISAT trial preferred) | Microcatheter delivery of platinum coils to occlude aneurysm lumen | First-line for most aneurysms; better short-term outcomes |
| Surgical clipping | Craniotomy + titanium clip across aneurysm neck | MCA bifurcation aneurysms; wide-necked aneurysms; when coiling fails |
ISAT Trial: Coiling significantly better than clipping at 1 year (independent survival 23.7% vs 30.6% risk of dependency/death).
| Complication | Timing | Management |
|---|---|---|
| Rebleeding | Peak 24 hrs | Urgent aneurysm occlusion |
| Vasospasm / DIND | Days 4–14 | Nimodipine; triple-H therapy (hypertension, hypervolemia, hemodilution) historically; now euvolemia + permissive hypertension; intra-arterial vasodilators |
| Hydrocephalus | Acute or chronic | EVD acutely; VP shunt if chronic |
| Hyponatremia (CSWS/SIADH) | Days 3–10 | Distinguish CSWS (fluid restriction harmful!) from SIADH; fludrocortisone for CSWS |
| Seizures | Any time | AED; levetiracetam preferred |
SECTION I: SECONDARY PREVENTION AFTER STROKE/TIA
| Intervention | Regimen | Evidence |
|---|---|---|
| Antiplatelet | Aspirin 75 mg + dipyridamole MR 200 mg BD (ESPS-2) or clopidogrel 75 mg OD | Reduces recurrent stroke by 20–25% |
| Anticoagulation (AF) | DOAC (apixaban, rivaroxaban, dabigatran, edoxaban) or Warfarin (INR 2–3) | Reduces cardioembolic stroke by 65% |
| Statin | Atorvastatin 80 mg OD (SPARCL trial) | Reduces recurrent stroke by 16% |
| Antihypertensive | Perindopril + indapamide (PROGRESS trial); target BP < 130/80 | Reduces recurrent stroke by 28% |
| Carotid endarterectomy | Symptomatic 70–99% stenosis within 2 weeks | NASCET: reduces 5-yr stroke risk from 26% to 9% |
| Carotid stenting | Alternative to CEA in high surgical risk | Slightly higher peri-procedural stroke rate |
| Lifestyle | Smoking cessation, weight reduction, exercise, reduced alcohol, Mediterranean diet | Comprehensive CVD risk reduction |
| Diabetes control | HbA1c < 48 mmol/mol | Slows progression of small vessel disease |
SECTION J: COMPLICATIONS OF STROKE
| System | Complication |
|---|---|
| Neurological | Cerebral edema, haemorrhagic transformation, seizures, hydrocephalus, recurrent stroke |
| Respiratory | Aspiration pneumonia (from dysphagia), neurogenic pulmonary oedema |
| Cardiovascular | DVT, pulmonary embolism, cardiac arrhythmias, stress cardiomyopathy |
| Metabolic | SIADH (hyponatremia), hyperglycemia, malnutrition |
| Musculoskeletal | Spasticity, contractures, shoulder subluxation, falls |
| Psychological | Post-stroke depression (30–40%), emotional lability, anxiety, PTSD |
| Cognitive | Vascular dementia, post-stroke cognitive impairment |
| Communication | Aphasia, dysarthria (speech and language therapy) |
SUMMARY COMPARISON TABLE — All 5 Questions Answered
| Topic | Key Point |
|---|---|
| Stroke definition | Focal neurological deficit > 24 hrs of vascular origin |
| TIA definition | Focal ischaemia WITHOUT infarction; resolves completely |
| TIA significance | 10–20% stroke risk within 90 days; highest in first 48 hours; ABCD² score guides urgency |
| Classification | Ischemic 85% (large vessel, lacunar, cardioembolic) vs Haemorrhagic 15% (ICH, SAH) |
| Main cause | Hypertension (for ICH + lacunar); AF (for cardioembolic); atherosclerosis (for large vessel) |
| Top risk factor | Hypertension (#1 modifiable); Age (#1 non-modifiable) |
| Ischemic tx | Alteplase 0.9 mg/kg within 4.5 hrs + mechanical thrombectomy within 24 hrs for LVO |
| Hemorrhagic tx | Reverse anticoagulation + SBP < 140 mmHg + surgery for cerebellar ICH / aneurysm coiling for SAH |
| Key difference | Thrombolysis → ✅ ischemic / ❌ absolutely contraindicated in hemorrhagic |
- Harrison's Principles of Internal Medicine, 21st Ed. (p. 12293)
- AHA/ASA TIA Guidelines 2009 — "Adult Patients With Suspected TIA in the Emergency Department" (p. 2)
- NICE Stroke Guidelines NG128 (2019)
- ISAT Trial — Neurosurgery vs coiling for SAH (Lancet 2002)
- POINT / CHANCE Trials — Dual antiplatelet after TIA
- HERMES meta-analysis — Mechanical thrombectomy
- INTERACT2, ATACH-2 — Blood pressure in ICH
- NASCET/ECST — Carotid endarterectomy
- PROGRESS Trial — BP lowering after stroke