VIT D synthesis
Reading File
search_medical_text
Vitamin D synthesis metabolism pathway biochemistry
search_medical_image
Vitamin D synthesis pathway diagram
GradeResponse
GradeResponse
Vitamin D Synthesis
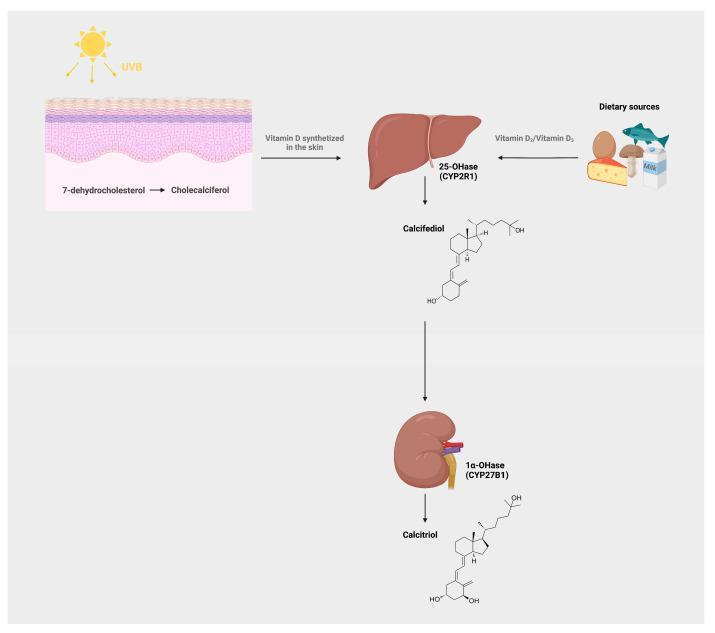
Overview
Vitamin D synthesis is a multi-organ, multi-step process involving the skin, liver, and kidneys. The end product is calcitriol (1,25-dihydroxyvitamin D₃), the biologically active hormone. (Harrison's, p. 1691)
Step-by-Step Pathway
| Step | Location | Substrate → Product | Enzyme/Mechanism |
|---|---|---|---|
| 1 | Skin | 7-dehydrocholesterol → Pre-vitamin D₃ | UV-B photolysis |
| 2 | Skin | Pre-vitamin D₃ → Vitamin D₃ (cholecalciferol) | Temperature-dependent isomerization |
| 3 | Liver | Vitamin D₃ → 25(OH)D₃ (calcifediol) | 25-hydroxylase (CYP2R1) |
| 4 | Kidney | 25(OH)D₃ → 1,25(OH)₂D₃ (calcitriol) | 1α-hydroxylase (CYP27B1) |
Details at Each Step
1 & 2 — Skin (Photosynthesis)
- Epidermal 7-dehydrocholesterol absorbs UV-B radiation (280–315 nm) and is photolyzed to pre-vitamin D₃.
- Pre-vitamin D₃ then undergoes spontaneous thermal isomerization to form the stable vitamin D₃.
- Vitamin D₃ diffuses into dermal capillaries and enters systemic circulation.
Key point: Prolonged UV exposure does NOT cause vitamin D toxicity — excess pre-vitamin D₃ and vitamin D₃ are degraded by UV light into inactive photoproducts (lumisterol, tachysterol).
3 — Liver (First Hydroxylation)
- Vitamin D₃ (from skin or diet) is hydroxylated at C-25 to form calcifediol (25-hydroxyvitamin D₃).
- This is the major circulating form and the standard lab test for vitamin D status.
- The reaction is largely unregulated — serum 25(OH)D reflects body stores/input.
4 — Kidney (Second Hydroxylation — Rate-Limiting Step)
- Calcifediol is hydroxylated at C-1α to form calcitriol (1,25-dihydroxyvitamin D₃) — the biologically active form.
- CYP27B1 (1α-hydroxylase) is tightly regulated:
- ↑ by: PTH, low serum calcium, low serum phosphate, prolactin, growth hormone
- ↓ by: calcitriol itself (negative feedback), FGF-23, high calcium/phosphate
Dietary Sources (Exogenous Pathway)
- Vitamin D₂ (ergocalciferol): from plant sources (UV-irradiated fungi/yeast)
- Vitamin D₃ (cholecalciferol): from animal sources (fatty fish, egg yolk, fortified milk)
- Both enter the same hepatic and renal hydroxylation pathway.
Physiological Effects of Calcitriol (1,25(OH)₂D₃)
| Target | Action |
|---|---|
| Small intestine | ↑ calcium & phosphate absorption |
| Kidney | ↑ calcium & phosphate reabsorption |
| Bone | Promotes mineralization (indirectly via Ca/Pi) |
| Parathyroid gland | Suppresses PTH synthesis |
| Immune cells | Immunomodulation (anti-inflammatory) |
| Skin (keratinocytes) | Inhibits proliferation, promotes differentiation |
Clinical Relevance
- Deficiency causes: rickets (children), osteomalacia (adults), secondary hyperparathyroidism
- Factors reducing synthesis: dark skin (melanin blocks UV-B), high latitude/winter, sunscreen, aging (↓ skin 7-dehydrocholesterol), chronic kidney disease (↓ CYP27B1), liver disease (↓ CYP2R1)
- Toxicity: almost always from supplementation, not sun exposure; causes hypercalcemia
Blood products and uses
search_medical_text
blood products types indications transfusion
search_medical_image
blood products components transfusion chart
GradeResponse
GradeResponse
search_medical_text
packed red blood cells platelets fresh frozen plasma cryoprecipitate indications dosing
search_medical_text
transfusion reactions complications blood products
GradeResponse
Blood Products and Their Uses
Overview of Major Blood Products
| Product | Derived From | Key Contents | Storage |
|---|---|---|---|
| Packed Red Blood Cells (pRBCs) | Whole blood (plasma removed) | RBCs, Hb | 2–6°C, up to 42 days |
| Fresh Frozen Plasma (FFP) | Whole blood (plasma fraction) | All clotting factors, fibrinogen | −18°C, up to 1 year |
| Platelets | Whole blood or apheresis | Platelets | 20–24°C (agitated), 5–7 days |
| Cryoprecipitate | FFP (cold precipitation) | Fibrinogen, Factor VIII, vWF, Factor XIII, fibronectin | −18°C, up to 1 year |
| Whole Blood | Unfractionated | All components | 2–6°C, up to 35 days |
| Albumin | Pooled plasma | Albumin (4–5% or 20–25%) | Room temp, 3–5 years |
| Prothrombin Complex Concentrate (PCC) | Pooled plasma | Factors II, VII, IX, X | Lyophilized, room temp |
| Factor Concentrates | Pooled plasma / recombinant | Specific factors (e.g., VIII, IX) | Varies |
1. Packed Red Blood Cells (pRBCs)
Each unit raises Hb by ~1 g/dL and Hct by ~3%
Indications (Bailey & Love, p. 42):
- Acute blood loss — to restore circulating volume and oxygen delivery
- Perioperative anaemia — maintaining adequate O₂ delivery during surgery
- Symptomatic chronic anaemia — without haemorrhage or impending surgery
Transfusion Thresholds (general guidance):
| Clinical Setting | Trigger Hb |
|---|---|
| Stable, non-cardiac inpatient | < 7 g/dL |
| Perioperative / cardiovascular disease | < 8 g/dL |
| Active ACS / symptomatic | < 10 g/dL |
| ICU (stable, mechanically ventilated) | < 7 g/dL |
Restrictive transfusion strategies are preferred — transfuse to symptoms, not numbers alone.
2. Fresh Frozen Plasma (FFP)
Each unit contains ~200–250 mL with all coagulation factors
Indications (Selected Topics in MCS, p. 10):
- PT or aPTT > 1.5× the reference range with active bleeding or prior to invasive procedure
- Massive transfusion protocol (MTP) — typically given in 1:1:1 ratio with pRBCs and platelets
- Reversal of warfarin (when PCC unavailable)
- TTP (Thrombotic Thrombocytopenic Purpura) — as plasma exchange fluid
- Liver disease with coagulopathy and bleeding
Dose: 10–15 mL/kg (typically 4 units in adults)
3. Platelets
One apheresis unit or pool of 4–6 whole-blood-derived units raises platelet count by ~30,000–50,000/µL
Indications (Selected Topics in MCS, p. 10):
- Therapeutic: Active bleeding with platelet count < 50,000/µL, or platelet dysfunction (e.g., aspirin, uremia)
- Prophylactic: Platelet count < 20,000/µL in stable patients; < 50,000/µL pre-procedure; < 100,000/µL pre-neurosurgery or ophthalmologic surgery
Contraindications: TTP, HIT (heparin-induced thrombocytopenia) — platelets are generally avoided as they worsen thrombosis.
4. Cryoprecipitate
Rich in fibrinogen (~250 mg/unit), Factor VIII, von Willebrand Factor, Factor XIII
Indications (Selected Topics in MCS, p. 10):
- Fibrinogen deficiency: fibrinogen < 100 mg/dL (some guidelines use < 150–200 mg/dL in active bleeding)
- Disseminated Intravascular Coagulation (DIC)
- Haemophilia A and von Willebrand disease (when specific concentrates unavailable)
- Massive hemorrhage / obstetric hemorrhage
Dose: 1 unit/5 kg body weight (typical adult dose = 10 units pooled)
5. Albumin
| Concentration | Indication |
|---|---|
| 4–5% (iso-oncotic) | Volume expansion in sepsis, SBP (spontaneous bacterial peritonitis), post-large-volume paracentesis |
| 20–25% (hyperoncotic) | Hypoalbuminaemia with oedema, hepatorenal syndrome |
6. Prothrombin Complex Concentrate (PCC)
- 4-factor PCC: Rapid reversal of warfarin (preferred over FFP — faster, lower volume)
- 3-factor PCC: Factors II, IX, X (less Factor VII)
- Also used for reversal of direct oral anticoagulants (DOACs) in life-threatening bleeding
Massive Transfusion Protocol (MTP)
Activated for > 10 units pRBCs in 24 hours or ongoing hemorrhagic shock:
- Standard ratio: pRBCs : FFP : Platelets = 1:1:1
- Consider Tranexamic acid (TXA) early (within 3 hours of injury)
- Monitor with thromboelastography (TEG/ROTEM) for targeted therapy
- Correct hypothermia, acidosis, hypocalcaemia (the "lethal triad")
Transfusion Reactions
| Reaction | Onset | Mechanism | Features |
|---|---|---|---|
| Acute Haemolytic | During/< 24h | ABO incompatibility (clerical error) | Fever, flank pain, haemoglobinuria, shock |
| Febrile Non-Haemolytic (FNHTR) | During transfusion | Cytokines from donor leukocytes | Fever, chills — no haemolysis |
| Allergic / Anaphylaxis | Minutes | IgE-mediated (donor plasma proteins) | Urticaria → bronchospasm, hypotension |
| TRALI (Transfusion-Related Acute Lung Injury) | < 6 hours | Donor anti-HLA antibodies | Acute hypoxia, bilateral pulmonary infiltrates |
| TACO (Transfusion-Associated Circulatory Overload) | During/< 6h | Volume overload | Pulmonary oedema, hypertension, dyspnoea |
| Delayed Haemolytic | 3–10 days | Anamnestic antibody response | Falling Hb, jaundice |
| Septic | During transfusion | Bacterial contamination (esp. platelets) | High fever, rigors, shock |
Special Modifications of Blood Products
| Modification | Purpose | Indication |
|---|---|---|
| Leukoreduced | Remove WBCs | Reduce FNHTR, CMV transmission, HLA alloimmunization |
| Irradiated | Prevent donor T-cell engraftment | Immunocompromised patients, HLA-matched products |
| CMV-negative | Reduce CMV transmission | CMV-seronegative immunocompromised patients |
| Washed | Remove plasma proteins | IgA deficiency, severe allergic reactions |
Cut off time for neck of femur fracture replacement
search_medical_text
neck of femur fracture surgery timing cutoff hemiarthroplasty total hip replacement
search_medical_text
hip fracture surgical delay outcomes mortality guidelines
GradeResponse
GradeResponse
search_medical_text
femoral neck fracture 48 hours 36 hours surgery timing avascular necrosis internal fixation young patient
Timing of Surgery for Neck of Femur (NOF) Fracture
The Core Principle: "As Soon As Medically Safe"
There is no single universal hard cut-off, but guidelines and evidence converge on within 36–48 hours of admission as the operative window — provided the patient is medically optimised.
Key Guideline Recommendations
| Guideline | Recommended Timing |
|---|---|
| NICE (UK, NG124) | Within 36 hours of admission (or injury if admitted same day) |
| AAOS / American Geriatrics Society | Within 48 hours of admission |
| SIGN (Scottish) | Within 24–48 hours |
| BOA (British Orthopaedic Association) | Within 36 hours |
| AO Foundation | Within 24–48 hours |
Bottom line: Most major guidelines target < 36–48 hours. The UK NICE guideline specifically sets 36 hours as the benchmark for audit purposes.
Why Timing Matters
| Consequence of Delay | Evidence |
|---|---|
| ↑ 30-day and 1-year mortality | Each 10-hour delay associated with measurable mortality increase |
| ↑ Pressure ulcer risk | Immobility in elderly |
| ↑ Venous thromboembolism (DVT/PE) | Prolonged bed rest |
| ↑ Pneumonia / delirium | Especially in elderly |
| ↑ Hospital length of stay | Functional decline |
| Avascular necrosis (AVN) | Critical in displaced fractures in young patients — see below |
Special Case: Young / Active Patient with Displaced Femoral Neck Fracture
This is the scenario where timing is most critical:
- The femoral head blood supply (mainly via medial femoral circumflex artery) is disrupted by displacement.
- Urgent reduction and internal fixation within 6–12 hours is traditionally recommended to restore blood flow and prevent AVN.
- Some evidence supports emergency surgery (< 6 hours) in young patients to preserve the native femoral head.
- In practice, many centres aim for < 24 hours even in young patients if logistics delay emergency fixation.
| Patient Group | Fracture Type | Preferred Procedure | Timing Goal |
|---|---|---|---|
| Young (< 60) | Displaced | Reduction + internal fixation (cannulated screws) | < 6–12 hours (urgent) |
| Elderly (> 60–65), active | Displaced (Garden III/IV) | Total Hip Replacement (THR) | < 36–48 hours |
| Elderly, low-demand / comorbid | Displaced | Hemiarthroplasty | < 36–48 hours |
| Any age | Undisplaced (Garden I/II) | Cannulated screws | < 48 hours (elective urgent) |
Does Accelerated Surgery (< 6 hours) Help in Elderly?
A recent multicentre RCT of 2,970 patients showed that accelerated surgery within 6 hours did NOT significantly reduce mortality or major complications compared to standard care (10–42 hours) in older patients (Management of Severe Peri-Operative Bleeding, p. 38).
This supports the approach of optimising the patient medically first rather than rushing to theatre — but still within the 36–48 hour window.
Acceptable Reasons to Delay Beyond 36–48 Hours
Surgery may be delayed if the patient has reversible, correctable conditions:
- Uncontrolled anticoagulation (warfarin: wait for INR < 2; DOACs: guidance varies — delay not clearly beneficial)
- Severe electrolyte disturbance
- Decompensated heart failure or uncontrolled arrhythmia
- Active chest infection requiring treatment
- Unstable angina / recent MI
Delay is NOT justified for: mild anaemia alone, diet-controlled diabetes, stable chronic conditions, or out-of-hours timing (meta-analysis shows no difference in outcomes for daytime vs. after-hours surgery) (Management of Severe Peri-Operative Bleeding, p. 38).
Summary
| Scenario | Cut-off |
|---|---|
| Standard elderly NOF fracture | 36–48 hours from admission |
| Young patient, displaced NOF | 6–12 hours (emergency) |
| Medically unstable (correctable cause) | Delay until safe, ideally < 72 hours |
| Daytime vs. after-hours | No difference in outcomes — operate when ready |
This is a shared conversation. Sign in to Orris to start your own chat.