Mycobacterium avium intercellulare full details
Reading File
search_medical_text
Mycobacterium avium intracellulare complex MAC microbiology pathogenesis epidemiology
search_medical_text
Mycobacterium avium complex clinical presentation diagnosis treatment HIV immunocompromised
search_medical_image
Mycobacterium avium complex chest imaging histology
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive overview of Mycobacterium avium-intracellulare (MAI) / Mycobacterium avium Complex (MAC):
Mycobacterium avium-intracellulare (MAI) / MAC
1. Microbiology & Classification
- Family: Mycobacteriaceae; Genus: Mycobacterium
- MAC = a group of closely related nontuberculous mycobacteria (NTM), primarily:
- Mycobacterium avium
- Mycobacterium intracellulare
- Mycobacterium paratuberculosis (and others)
- Staining: Acid-fast bacilli (AFB) — positive on Ziehl-Neelsen and auramine-rhodamine stains
- Growth: Slow-growing (colonies appear in 2–4 weeks); grows at 37°C; non-chromogenic
- Morphology: Slightly curved, rod-shaped organisms; intracellular pathogen — survives within macrophage phagolysosomes by inhibiting phagolysosomal fusion
- Serovars: >28 serovars identified; serovars 1–6 predominantly cause human disease
2. Epidemiology
(Prevention and Treatment of Opportunistic Infections, p. 326)
- Ubiquitous environmental organism: found in soil, water (including tap water, hot tubs, swimming pools), aerosols, animals, and food
- Not transmitted person-to-person; acquired from environmental exposure
- In the United States, NTM infections outnumber M. tuberculosis infections and are an important cause of pulmonary morbidity in adults
- The number of known NTM species grew from 50 (1997) to >125 (2006), with MAC remaining the most clinically significant
- Increasing incidence observed in some U.S. regions and globally
- Disseminated MAC is rare in immunocompetent individuals; primarily affects immunocompromised hosts
3. Pathogenesis
- Entry via inhalation (respiratory tract) or ingestion (gastrointestinal tract)
- Organisms are phagocytosed by macrophages but resist intracellular killing:
- Inhibit phagolysosome acidification
- Produce glycopeptidolipids (GPL) that impair macrophage activation
- Down-regulate TNF-α and IL-12 production
- In immunocompetent hosts → granuloma formation contains infection
- In HIV/AIDS or severe immunosuppression (CD4 <50 cells/μL) → uncontrolled macrophage infection → disseminated disease
4. Clinical Syndromes
MAC causes four distinct clinical syndromes:
A. Pulmonary MAC Disease (most common in immunocompetent hosts)
Two major patterns:
| Feature | Classic / Fibrocavitary | Lady Windermere / Nodular-Bronchiectatic |
|---|---|---|
| Population | Older men, smokers, COPD | Post-menopausal women, slender build, scoliosis |
| Symptoms | Productive cough, hemoptysis, fever | Chronic cough, fatigue, weight loss |
| Radiology | Upper lobe cavitary lesions (TB-like) | Middle lobe/lingula bronchiectasis, nodules |
| Course | Aggressive, progressive | Indolent |
CT Findings (Harrison's, p. 5254):
Characteristic CT pattern includes bronchiectasis and tree-in-bud pattern (bronchiolar inflammation), distributed in both lungs, particularly the middle lobe and lingula.
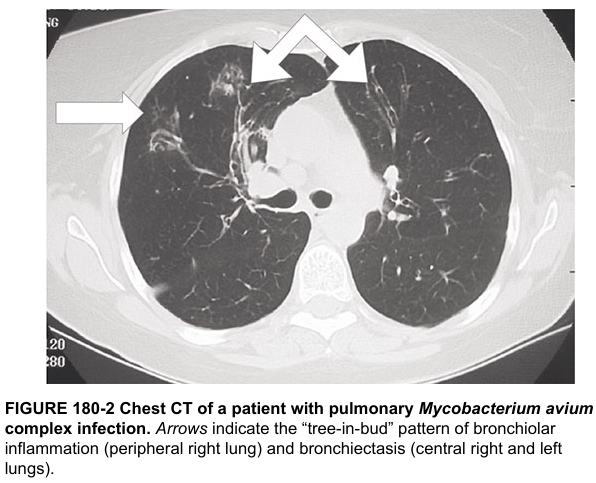
B. Disseminated MAC (DMAC) — primarily in HIV/AIDS
- Occurs when CD4 count <50 cells/μL
- Symptoms: High spiking fevers, drenching night sweats, profound weight loss, fatigue, anorexia
- Signs: Hepatosplenomegaly, lymphadenopathy, anemia, elevated alkaline phosphatase
- Bacteremia: Organisms are recoverable from blood cultures (lysis-centrifugation or BACTEC)
- Without ART and prophylaxis, incidence was ~40% per year in advanced AIDS
C. Lymphadenitis (Scrofula)
- Primarily in immunocompetent children (ages 1–5 years)
- Unilateral, painless cervical or submandibular lymphadenopathy
- Skin may become violaceous; fistula formation possible
- Diagnosis: Excisional biopsy (preferred over FNA)
D. Hypersensitivity/Hot Tub Lung
- Non-infectious, granulomatous hypersensitivity pneumonitis from aerosolized MAC (hot tubs, showers)
- Presents with dyspnea, cough, fever after hot tub exposure
- Ground-glass opacities on CT; responds to antigen avoidance ± steroids
5. Diagnosis
Pulmonary MAC (ATS/IDSA Diagnostic Criteria)
All three criteria must be met:
- Clinical: Pulmonary symptoms with nodular or cavitary opacities on CXR, or HRCT bronchiectasis with multiple small nodules — after excluding other diagnoses
- Microbiologic (any of):
- ≥2 positive sputum cultures (separate specimens), OR
- ≥1 positive bronchial wash/lavage culture, OR
- Transbronchial or lung biopsy with mycobacterial histopathology + positive culture
- Expert consultation recommended
Disseminated MAC
- Blood cultures (mycobacterial — lysis-centrifugation method or BACTEC MYCO/F Lytic): high sensitivity
- Bone marrow biopsy: granulomas with AFB
- Liver biopsy: granulomatous hepatitis
- CBC: normocytic anemia, elevated ESR, elevated ALP
- Identification: DNA probes, HPLC of mycolic acids, 16S rRNA sequencing
Lymphadenitis
- Excisional biopsy: caseating granulomas with AFB; culture for definitive ID
6. Treatment
Pulmonary MAC — ATS/IDSA 2020 Guidelines
Three-drug regimen (all patients with nodular-bronchiectatic or fibrocavitary disease):
| Drug | Dose | Notes |
|---|---|---|
| Azithromycin 500 mg OR Clarithromycin 500–1000 mg/day | Once daily (AZI) or BID (CLA) | Macrolide backbone — CRITICAL |
| Rifampicin 600 mg/day | Daily | |
| Ethambutol 15 mg/kg/day | Daily |
- Duration: Until sputum culture-negative for 12 months
- Amikacin (IV or inhaled liposomal amikacin) added for severe/refractory disease
- Bedaquiline emerging as option for macrolide-resistant MAC
Disseminated MAC (HIV/AIDS)
Treatment (USPHS/IDSA Guidelines):
- Azithromycin 500–600 mg/day + Ethambutol 15 mg/kg/day (preferred)
- Alternative: Clarithromycin 500 mg BID + Ethambutol ± Rifabutin 300 mg/day
- Duration: ≥12 months + immune reconstitution (CD4 >100 cells/μL on ART for >6 months)
- ART initiation: Start within 2 weeks of MAC treatment initiation
Prophylaxis (primary prevention) in HIV:
- Indicated when CD4 <50 cells/μL
- Azithromycin 1200 mg once weekly (preferred) OR Clarithromycin 500 mg BID
- Can discontinue when CD4 ≥100 cells/μL sustained on ART for >3 months
Lymphadenitis
- Excision (complete excisional lymph node removal) is curative in most cases
- Antibiotics added for extensive/recurrent disease
7. Drug Susceptibility Testing (DST)
- Macrolide susceptibility testing (clarithromycin MIC) is mandatory before and during treatment
- Macrolide resistance (clarithromycin MIC >32 μg/mL) = poor prognosis, requires alternative regimens
- Acquired resistance occurs via 23S rRNA mutations from macrolide monotherapy (never use as monotherapy)
- Routine susceptibility testing for rifamycins and ethambutol has limited clinical correlation, except for macrolides and amikacin
8. Immune Reconstitution Inflammatory Syndrome (IRIS)
- In HIV patients starting ART while on MAC treatment
- Paradoxical worsening: fever, lymphadenopathy, new lesions
- Unmasking IRIS: MAC diagnosed after ART initiation reveals previously subclinical infection
- Management: Continue both ART and MAC treatment; NSAIDs; steroids for severe IRIS
9. Complications
| Complication | Context |
|---|---|
| Cavitation, respiratory failure | Severe pulmonary MAC |
| Severe anemia, cachexia | Disseminated MAC in AIDS |
| Macrolide resistance | Inadequate treatment regimens |
| IRIS | HIV patients on ART |
| Fistula formation | MAC lymphadenitis |
| Hepatitis, bone marrow suppression | Disseminated disease |
10. Prognosis
- Immunocompetent pulmonary MAC: Variable; some patients with nodular-bronchiectatic disease have stable or slowly progressive course; fibrocavitary form is more aggressive with ~50% treatment success rates
- Disseminated MAC in HIV (pre-ART era): Median survival ~4–5 months; with ART + treatment, dramatically improved
- Post-ART era: Disseminated MAC is far less common; mortality related primarily to HIV disease control
- Macrolide-resistant MAC: Cure rates <30%
- Children with lymphadenitis: Excellent prognosis with surgical excision
11. Prevention
- HIV patients: Primary MAC prophylaxis (azithromycin weekly) when CD4 <50 cells/μL
- ART: Most effective prevention strategy — immune reconstitution prevents disseminated MAC
- Environmental: No specific recommendations; avoid aerosolized water sources (hot tubs) in severely immunocompromised patients
- Surveillance and guideline updates via USPHS/IDSA and ATS/IDSA Joint Committees
Key References:
- Harrison's Principles of Internal Medicine, 21st Ed., p. 5254
- Prevention and Treatment of Opportunistic Infections in Adults/Adolescents with HIV (USPHS/IDSA), p. 460
- Prevention and Treatment of Opportunistic Infections in Children with and Exposed to HIV, p. 326
- ATS/IDSA NTM Guidelines (2007, updated 2020)
This is a shared conversation. Sign in to Orris to start your own chat.