All about pathology of heart from latest Robbins book for pathology
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive content from all major sections. Let me compile the full answer.
Cardiac Pathology — Robbins, Cotran & Kumar: Pathologic Basis of Disease (Latest Edition, ISBN 9780443264528)
Overview
Cardiac disease has an interplay of genetic and environmental factors. Molecular diagnostics plays an increasing role in understanding individual patients. The chapter covers the following major entities: heart failure, congenital heart disease, ischemic heart disease, hypertensive heart disease, valvular heart disease, cardiomyopathies, myocarditis, pericarditis, and cardiac tumors.
1. HEART FAILURE (CHF)
Epidemiology: Affects >6.5 million Americans (~2% of the population); ~1 million hospitalizations/year; ~400,000 deaths/year. ~50% of patients die within 5 years of diagnosis.
Definition: The heart cannot pump blood to meet peripheral metabolic demands, or does so only at elevated filling pressures. It is the final common pathway of many chronic heart diseases.
Compensatory Mechanisms
- Frank-Starling mechanism — increased filling → myocyte stretch → more forceful contraction
- Neurohumoral activation — norepinephrine (↑HR, ↑contractility, ↑vascular resistance), RAAS (salt/water retention), atrial natriuretic peptide (counterbalancing diuresis)
- Myocardial hypertrophy & ventricular remodeling — collective molecular, cellular, and structural changes in response to injury/altered loading
Classification
| Type | EF | Dominant Dysfunction |
|---|---|---|
| HFrEF | ≤40% | Systolic (contractile failure) |
| HFpEF | ≥50% | Diastolic (impaired filling) |
HFpEF patients tend to be older, female, with higher prevalence of obesity and type 2 diabetes.
Left-Sided Heart Failure
- Causes: IHD, hypertension, aortic/mitral valve disease, myocardial disease
- Morphology: left ventricular dilation or hypertrophy; lungs show pulmonary congestion, hemosiderin-laden macrophages ("heart failure cells"), interstitial edema, and ultimately pulmonary fibrosis
- Clinical: dyspnea, orthopnea, paroxysmal nocturnal dyspnea, systemic hypoperfusion causing renal/cerebral dysfunction
Right-Sided Heart Failure
- Usually a consequence of left heart failure; less commonly primary pulmonary disorders
- Morphology: right ventricular dilation; systemic/portal venous congestion → hepatomegaly (nutmeg liver), splenomegaly, pleural effusions, ascites, peripheral edema
- Clinically dominated by peripheral edema and visceral congestion
2. CONGENITAL HEART DISEASE (CHD)
Definition: Abnormalities of the heart or great vessels present at birth; most arise from faulty embryogenesis during gestational weeks 3–8.
Incidence: ~1% of live births for serious defects; small VSDs/ASDs detected in >5% by routine echo. ~1.5 million adults in the USA live with CHD.
Frequencies of Common Malformations (Table 12.2)
| Malformation | Incidence (%) |
|---|---|
| Ventricular septal defect (VSD) | 42% |
| Atrial septal defect (ASD) | 10% |
| Pulmonary stenosis | 8% |
| Patent ductus arteriosus (PDA) | 7% |
| Tetralogy of Fallot | 5% |
| Coarctation of aorta | 5% |
| AV septal defect | 4% |
| Aortic stenosis | 4% |
| Transposition of great arteries | 4% |
Key Entities
- VSD: Most common; 90% are membranous; small ones close spontaneously; large → Eisenmenger syndrome (L→R shunt → pulmonary hypertension → shunt reversal → cyanosis)
- ASD: Often asymptomatic for decades; secundum type most common; ostium primum type associated with Down syndrome
- PDA: Failure of ductus arteriosus to close after birth; prostaglandins maintain patency; indomethacin can close it
- Tetralogy of Fallot: (1) VSD, (2) pulmonary/infundibular stenosis, (3) overriding aorta, (4) right ventricular hypertrophy — classical "boot-shaped heart" on X-ray; cyanosis from R→L shunt
- Transposition of Great Arteries: Aorta arises from RV, pulmonary artery from LV — incompatible with life unless a mixing defect (ASD, VSD, PDA) is present
- Coarctation of Aorta: Male predominance; associated with Turner syndrome; causes upper extremity hypertension and lower extremity hypotension/claudication; "rib notching" on X-ray
3. ISCHEMIC HEART DISEASE (IHD)
Epidemiology: Single largest cause of mortality worldwide; >15% of global deaths; ~9 million deaths/year in high-income countries. US death rate from IHD has fallen >50% since the mid-1960s.
Definition: Myocardial ischemia due to imbalance between supply and demand for oxygenated blood. In >90% of cases, caused by obstructive atherosclerotic lesions in epicardial coronary arteries.
Clinical Presentations
- Myocardial infarction (MI)
- Angina pectoris (stable, unstable, Prinzmetal's vasospastic)
- Chronic IHD with heart failure
- Sudden cardiac death (SCD)
Pathogenesis
- Stable plaque → stable angina
- Disrupted/ruptured plaque → superimposed thrombus → acute coronary syndrome (unstable angina, NSTEMI, STEMI) or SCD
- Non-atherosclerotic causes: coronary emboli, vasculitis, spasm, anomalous origin
Myocardial Infarction
Types:
- STEMI: Transmural infarct, complete occlusion; classic ST elevation
- NSTEMI: Subendocardial infarct, often partial occlusion; no ST elevation
Evolution of MI (morphologic timeline):
| Time | Gross Changes | Microscopic Changes |
|---|---|---|
| 0–6 hrs | None (may see pallor) | None visible on H&E; wavy fibers |
| 6–24 hrs | Pale/mottled | Coagulative necrosis begins; neutrophil infiltration starts |
| 1–3 days | Yellow-tan pallor | Dense neutrophil infiltration; nuclear pyknosis |
| 3–7 days | Hyperemic border; central yellowing | Macrophage replacement of neutrophils; begin granulation tissue |
| 10 days–8 weeks | Depressed scar | Progressive granulation tissue → fibrosis |
| >2 months | White fibrous scar | Dense fibrous scar (completed remodeling) |
Complications of MI:
- Arrhythmias (most common cause of death in the first 24 hours)
- Cardiogenic shock
- Cardiac tamponade from free wall rupture (days 3–7)
- Interventricular septal rupture
- Papillary muscle rupture → acute mitral regurgitation
- Mural thrombus → systemic embolism
- Fibrinous pericarditis (Dressler syndrome when autoimmune, weeks later)
- Ventricular aneurysm
Sudden Cardiac Death (SCD)
Death from cardiac causes within 1 hour of symptom onset. Most due to lethal arrhythmia (ventricular fibrillation) on a background of chronic IHD; in young people, causes include HCM, ARVC, congenital coronary anomalies, aortic stenosis, and channelopathies.
4. HYPERTENSIVE HEART DISEASE (HHD)
Systemic (Left-Sided) HHD
- Minimal criteria: (1) LV hypertrophy (usually concentric) without other cardiac pathology, and (2) clinical/pathologic evidence of hypertension
- Heart weight may exceed 500 g; LV wall thickness may exceed 2.0 cm
- Microscopy: increased transverse diameter of myocytes; perivascular and interstitial fibrosis
- Complications: diastolic dysfunction, left atrial enlargement, atrial fibrillation, IHD, CHF, SCD
- Effective BP control can cause regression of hypertrophy
Pulmonary (Right-Sided) HHD — Cor Pulmonale
- Right ventricular hypertrophy, dilation, and potential right-sided failure due to pulmonary hypertension
- Caused by: COPD (most common), pulmonary fibrosis, pulmonary embolism, primary pulmonary arterial hypertension
5. VALVULAR HEART DISEASE
Two fundamental defects:
- Stenosis — failure to open completely; almost always chronic (calcification/scarring)
- Insufficiency/Regurgitation — failure to close completely; acute (chordal rupture, IE) or insidious (scarring)
Most frequent causes:
| Lesion | Most Common Cause |
|---|---|
| Aortic stenosis | Calcification of normal or bicuspid valve |
| Aortic insufficiency | Dilation of ascending aorta (hypertension/aging) |
| Mitral stenosis | Rheumatic heart disease |
| Mitral insufficiency | Myxomatous degeneration (MVP) or LV dilation |
Calcific Valvular Degeneration
- Most common cause of aortic stenosis in adults; calcium deposits in valve cusps at flexion points
- Morphology: focal calcific deposits on the outflow (aortic) side of aortic cusps; no commissural fusion (unlike RHD)
- Bicuspid aortic valve (1–2% prevalence) calcifies earlier (5th–6th decade) vs. tricuspid (7th–8th decade)
Rheumatic Heart Disease (RHD)
- Immune-mediated; follows pharyngeal Group A streptococcal infection
- Aschoff bodies = pathognomonic granuloma-like lesions with central fibrinoid necrosis, surrounded by lymphocytes, plasma cells, and Anitschkow cells (caterpillar cells — macrophages with characteristic chromatin pattern)
- Acute RHD: carditis (pancarditis — endo, myo, peri); small, warty vegetations along the line of valve closure
- Chronic RHD: fibrous thickening, commissural fusion, leaflet calcification; the mitral valve is most commonly affected, followed by aortic, then tricuspid
- "Fish-mouth" or "button-hole" mitral stenosis on gross exam
Endocarditis Comparison (Key Robbins diagram):
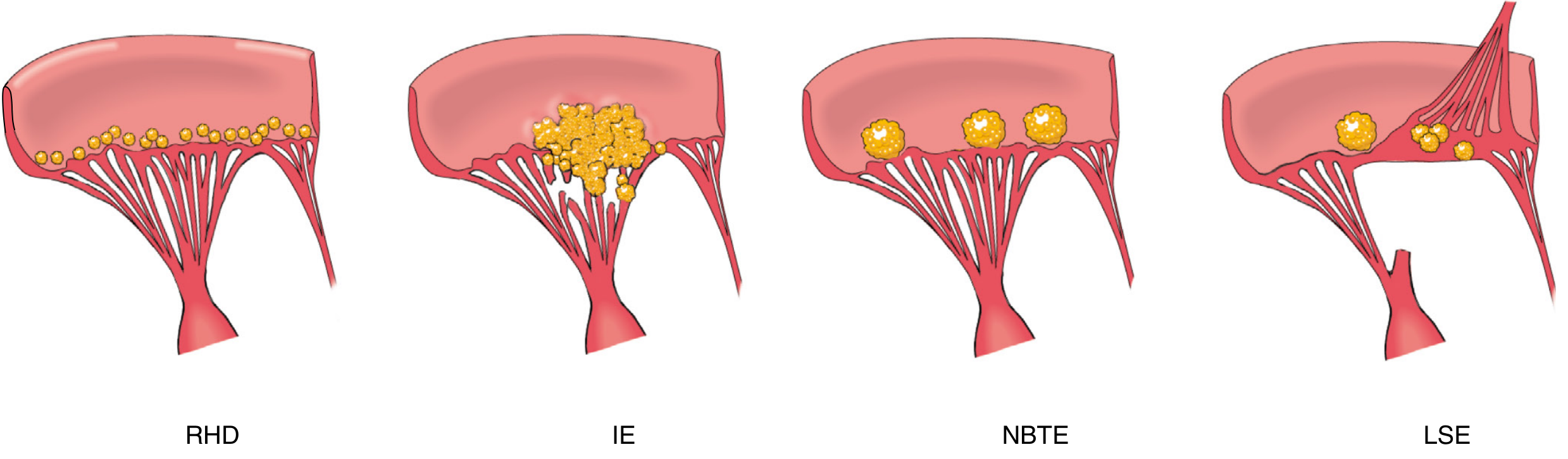
Infective Endocarditis (IE)
- Microbial infection of valves/mural endocardium → vegetations of thrombotic debris + organisms + valve destruction
- Acute IE: Highly virulent organisms (S. aureus) on previously normal valves; rapid, destructive; requires surgery
- Subacute IE: Low-virulence organisms (Streptococcus viridans) on deformed valves; indolent; often responds to antibiotics alone
Causative organisms:
| Setting | Organism |
|---|---|
| Native, previously damaged valves | S. viridans (50–60%) |
| Overall (most common) | S. aureus |
| IV drug users | S. aureus (right-sided — tricuspid) |
| Prosthetic valve (early, <2 months) | S. aureus, S. epidermidis |
| Prosthetic valve (late, >1 year) | Streptococci, S. aureus |
HACEK organisms: Haemophilus, Actinobacillus, Cardiobacterium, Eikenella, Kingella — oral commensals; important in culture-negative IE.
Morphology: Vegetations are irregular, destructive masses on valve cusps that can extend onto chordae, with areas of necrosis. Microscopy: fibrin, inflammatory cells, bacteria, necrotic debris.
Duke Criteria used clinically; complications include valve destruction, septic emboli, glomerulonephritis (immune complex deposition), mycotic aneurysms.
Nonbacterial Thrombotic Endocarditis (NBTE) / Marantic Endocarditis
- Small, sterile, bland vegetations along the line of valve closure
- Associated with: cancer (especially mucin-secreting adenocarcinoma), sepsis, burns, prolonged debilitating illness
- Mechanism: hypercoagulable state
Libman-Sacks Endocarditis (LSE)
- Small-medium vegetations on either or both sides of valve leaflets
- Pathognomonic of SLE; associated with antiphospholipid antibody syndrome
Mitral Valve Prolapse (MVP) / Myxomatous Degeneration
- Most common valvular abnormality (1–2.5% of adults); female predominance
- Morphology: enlarged, floppy leaflets that balloon into the LA during systole; myxomatous change (acid mucopolysaccharide accumulation) weakens the fibrosa layer
- Complication: chordal rupture, mitral regurgitation; small increased risk of IE and SCD
Carcinoid Heart Disease
- Right-sided valvular lesions (tricuspid/pulmonary) due to serotonin and other vasoactive substances released by carcinoid tumors
- Fibrous plaques on endocardial surfaces ("carcinoid plaques")
- Left heart spared because lungs inactivate serotonin; left-sided involvement occurs with pulmonary carcinoid or right-to-left shunts
6. CARDIOMYOPATHIES
Three primary types:
| Feature | DCM | HCM | RCM |
|---|---|---|---|
| Ventricle | Dilated, flabby | Thick-walled, stiff | Normal size, stiff |
| Dysfunction | Systolic | Diastolic | Diastolic |
| EF | Low | Normal/high | Normal |
| Key gene | TTN, lamin A/C | MYH7, MYBPC3 | Amyloid, sarcoid |
Dilated Cardiomyopathy (DCM)
- Progressive cardiac dilation + contractile failure, usually with hypertrophy
- Familial in up to 50% of cases; autosomal dominant most common
- TTN mutations (titin gene) = 10–20% of all DCM cases
- Other causes: myocarditis (Coxsackie B), alcohol/toxins (acetaldehyde, cobalt, chemotherapy), peripartum, Duchenne/Becker (dystrophin mutations), hemochromatosis
- Morphology: all four chambers dilated; pale, flabby myocardium; mural thrombi common; microscopy shows myocyte hypertrophy, interstitial fibrosis (nonspecific)
- Clinical: progressive CHF, arrhythmias, emboli; transplantation may be required
Hypertrophic Cardiomyopathy (HCM)
- Prevalence: 1 in 500; leading cause of SCD in young athletes
- Autosomal dominant; >400 mutations in 9 sarcomeric protein genes; most common: MYBPC3 (myosin-binding protein C) and MYH7 (β-myosin heavy chain) — together 70–80% of cases
- All are gain-of-function mutations → hypercontractility
- Morphology: massive ventricular hypertrophy (asymmetric septal hypertrophy); banana-shaped LV on cross-section; "subaortic hypertrophy" causing dynamic outflow obstruction (in ~1/3)
- Microscopy hallmark: myofiber disarray (haphazard arrangement of myocytes at odd angles to each other) + interstitial fibrosis
- Systolic anterior motion (SAM) of the anterior mitral leaflet worsens outflow obstruction
- Clinical: diastolic dysfunction, angina, syncope, SCD; novel therapy: mavacamten (myosin inhibitor)
Restrictive Cardiomyopathy (RCM)
- Decreased ventricular compliance → impaired diastolic filling; normal systolic function
- Causes: amyloidosis (most common in adults), sarcoidosis, hemochromatosis, endomyocardial fibrosis (Löffler endocarditis — eosinophilic), glycogen storage diseases, radiation fibrosis
- Morphology: firm, non-compliant myocardium; microscopy depends on cause (e.g., Congo red–positive amyloid deposits, eosinophilic infiltration)
Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC)
- Autosomal dominant with variable penetrance
- Mutations in desmosomal proteins (plakoglobin, desmin) at the intercalated disk
- Naxos syndrome = ARVC + palmoplantar hyperkeratosis (plakoglobin mutation)
- Morphology: right ventricular wall severely attenuated; massive fatty infiltration + focal fibrosis replacing RV myocardium
- Clinical: right-sided heart failure, ventricular arrhythmias, SCD in young adults; can mimic DCM
7. MYOCARDITIS
Definition: Myocardial injury due to infectious microorganisms or primary inflammatory process (distinct from secondary inflammation in IHD).
Causes (Table 12.12)
- Infections: Viruses (Coxsackievirus A & B most common in the US; also CMV, HIV, influenza; COVID-19 causes focal myocardial necrosis via microthrombi)
- Trypanosoma cruzi → Chagas disease (endemic in South America; acute death 10%; chronic immune-mediated cardiomyopathy in 10–20 years)
- Trichinella spiralis (most common helminth)
- Diphtheria toxin (Corynebacterium diphtheriae)
- Borrelia burgdorferi (Lyme disease — self-limited conduction abnormalities)
- Immune-mediated: Poststreptococcal (RHD), SLE, drug hypersensitivity, transplant rejection, immune checkpoint inhibitor therapies
- Unknown: Sarcoidosis, giant cell myocarditis (idiopathic; rapidly fatal; pathognomonic multinucleated giant cells + extensive necrosis)
Morphology
- Gross: may be normal or show ventricular dilation
- Microscopy: lymphocytic (most common) — interstitial edema + mononuclear inflammatory infiltrate + myocyte necrosis; eosinophilic; giant cell; granulomatous (sarcoid)
Clinical
- Can range from asymptomatic (incidental finding) to fulminant CHF and SCD
- Some cases resolve completely; others progress to DCM
8. PERICARDIAL DISEASE
Acute Pericarditis
| Type | Key Associations |
|---|---|
| Serous | Rheumatic fever, SLE, scleroderma, tumors, uremia; sterile; rarely organizes |
| Fibrinous/Serofibrinous | Most frequent type; acute MI, Dressler syndrome, uremia, RHD, SLE, trauma, post-cardiac surgery; "bread and butter" pericarditis |
| Purulent/Suppurative | Bacterial invasion (S. aureus, streptococci, gram-negatives); high mortality; may require surgical drainage |
| Hemorrhagic | Blood + fibrinous exudate; tuberculosis, malignancy, post-cardiac surgery |
| Caseous | Tuberculosis; most frequent antecedent to constrictive pericarditis |
Clinical features of fibrinous pericarditis: Pleuritic, position-dependent chest pain; fever; pericardial friction rub.
Pericardial Effusion and Cardiac Tamponade
- Slowly accumulating effusions (up to 500 mL) tolerated well; rapidly accumulating effusion of 200–300 mL can cause tamponade (compression of heart, Beck's triad: hypotension, elevated JVP, muffled heart sounds)
Chronic/Healed Pericarditis — Constrictive Pericarditis
- Most commonly follows TB, hemopericardium from cardiac surgery, radiation
- Pericardium replaced by dense fibrous scar or calcification, encasing the heart
- Clinical: mimics RHD or tricuspid stenosis; elevated JVP, ascites, Kussmaul's sign (paradoxical rise in JVP on inspiration)
9. TUMORS OF THE HEART
Primary Cardiac Tumors
Uncommon; most are benign. Top 5 (benign, ~90% of primaries):
- Myxoma (most common in adults)
- Fibroma
- Lipoma
- Papillary fibroelastoma
- Rhabdomyoma (most common in infants/children; strongly associated with tuberous sclerosis)
Cardiac Myxoma:
- ~90% arise in the atria; L:R ratio = 4:1; origin from fossa ovalis
- Sporadic (no consistent genetic alteration) vs. syndromic:
- McCune-Albright syndrome (GNAS1 gain-of-function mutation — G protein signaling)
- Carney complex (PRKAR1A loss-of-function mutation — ↑cAMP signaling)
- Morphology: sessile or pedunculated, 1–10 cm; gelatinous or hemorrhagic; histology: stellate myxoma cells in abundant acid mucopolysaccharide matrix + peculiar vessel-like/gland-like structures
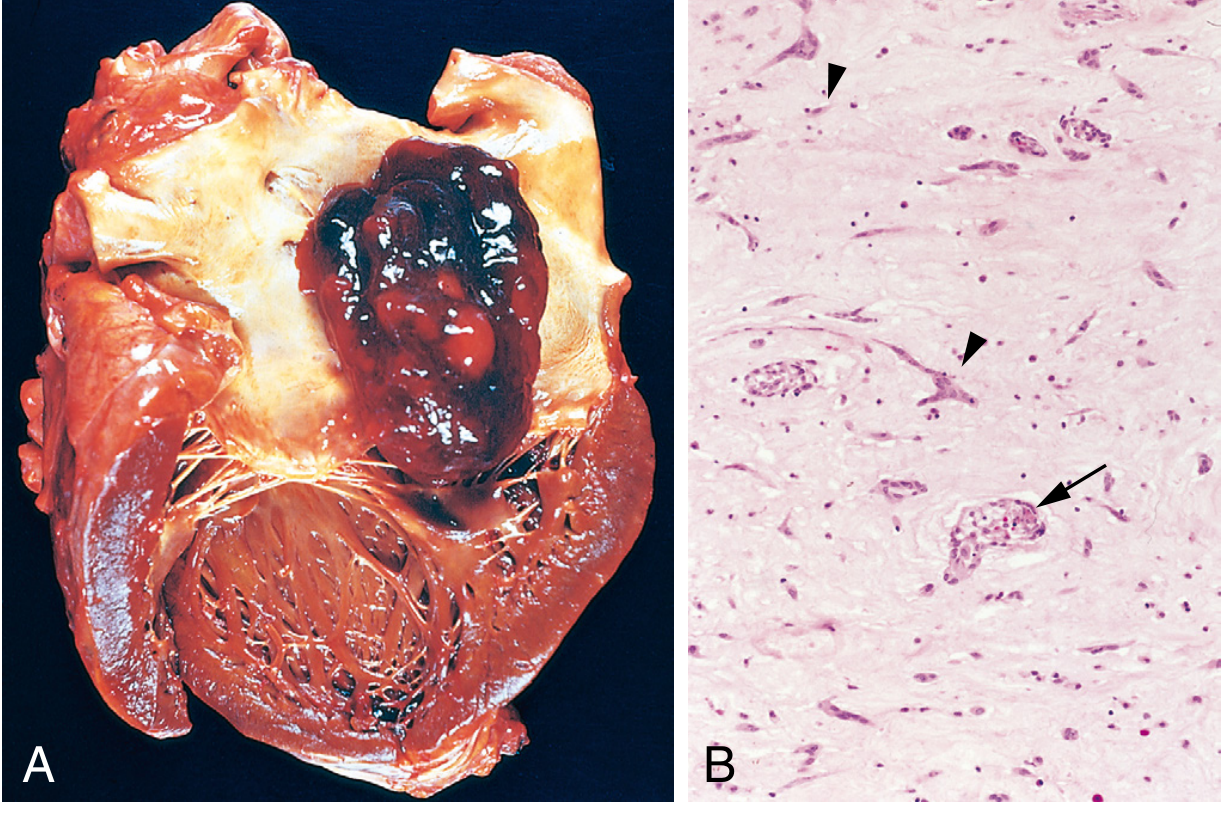
- Clinical manifestations: (1) "ball-valve" obstruction of mitral valve orifice (position-dependent symptoms), (2) systemic embolization, (3) constitutional symptoms (fever, malaise — due to IL-6 secretion)
- Treatment: surgical excision — usually curative; rare recurrence if incompletely excised
Rhabdomyoma:
- Most common primary cardiac tumor in infants and children
- Strongly associated with tuberous sclerosis (TSC1/TSC2 mutations — hamartomas)
- Often regress spontaneously; may cause outflow obstruction or arrhythmias
Secondary (Metastatic) Cardiac Tumors
- Far more common than primary tumors
- Most common sources: lung carcinoma, breast carcinoma, lymphoma/leukemia, malignant melanoma (highest rate of cardiac involvement per primary tumor)
- Reach the heart via direct invasion, hematogenous spread, lymphatic spread, or growth through the vena cava
Summary Table of Key Cardiac Pathologies
| Entity | Key Pathologic Feature | Key Mechanism |
|---|---|---|
| CHF | Ventricular dilation/hypertrophy, pulmonary congestion | Pressure/volume overload; ischemia |
| CHD | Structural defects (VSD, ASD, ToF, etc.) | Embryogenic errors wks 3–8 |
| Acute MI | Coagulative necrosis; neutrophil → macrophage → scar | Atherosclerotic plaque rupture + thrombosis |
| HHD | Concentric LV hypertrophy | Systemic hypertension |
| Aortic stenosis | Dystrophic calcification of cusps | Mechanical stress/aging |
| RHD | Aschoff bodies; commissural fusion | Anti-streptococcal immune response |
| Infective endocarditis | Large destructive vegetations | Microbial colonization of endocardium |
| NBTE | Small sterile vegetations | Hypercoagulable state |
| Libman-Sacks | Bilateral vegetations | SLE/antiphospholipid syndrome |
| DCM | All chambers dilated; systolic failure | Titin/cytoskeletal mutations; viral; toxins |
| HCM | Asymmetric septal hypertrophy; myofiber disarray | Sarcomeric gain-of-function mutations (MYH7, MYBPC3) |
| ARVC | Fatty + fibrous replacement of RV | Desmosomal protein mutations |
| Myocarditis | Inflammatory infiltrate + myocyte necrosis | Viral (Coxsackie B), immune, Chagas |
| Constrictive pericarditis | Dense fibrous/calcified pericardial scar | TB, post-surgical, radiation |
| Cardiac myxoma | Stellate cells in mucopolysaccharide matrix; left atrium | Primitive mesenchymal cells; Carney complex |
— Robbins, Cotran & Kumar: Pathologic Basis of Disease (ISBN 9780443264528), Chapter 12: Heart, pp. 481–544
Retry
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.