Describe the functionally distinct compartment of body water in a 70 kg man, elaborate on osmolality and tonicity.what are primary method of regulation of body fluid water. Write a notes on this question for MD Anaesthesiology exam
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Body Water Compartments, Osmolality, Tonicity & Regulation of Body Fluid
Notes for MD Anaesthesiology Examination
I. TOTAL BODY WATER (TBW) IN A 70 KG MAN
Total body water (TBW) constitutes approximately 60% of body weight in an average adult male — equating to 42 litres in a 70 kg man.
TBW correlates inversely with body fat. Women (~50–55% body weight is water) and obese individuals have proportionally less TBW; neonates and lean athletes have more (up to 75%).
II. FUNCTIONALLY DISTINCT COMPARTMENTS OF BODY WATER
Body water is segregated into two major compartments by the cell membrane, with the extracellular compartment further subdivided by the capillary wall (endothelium):
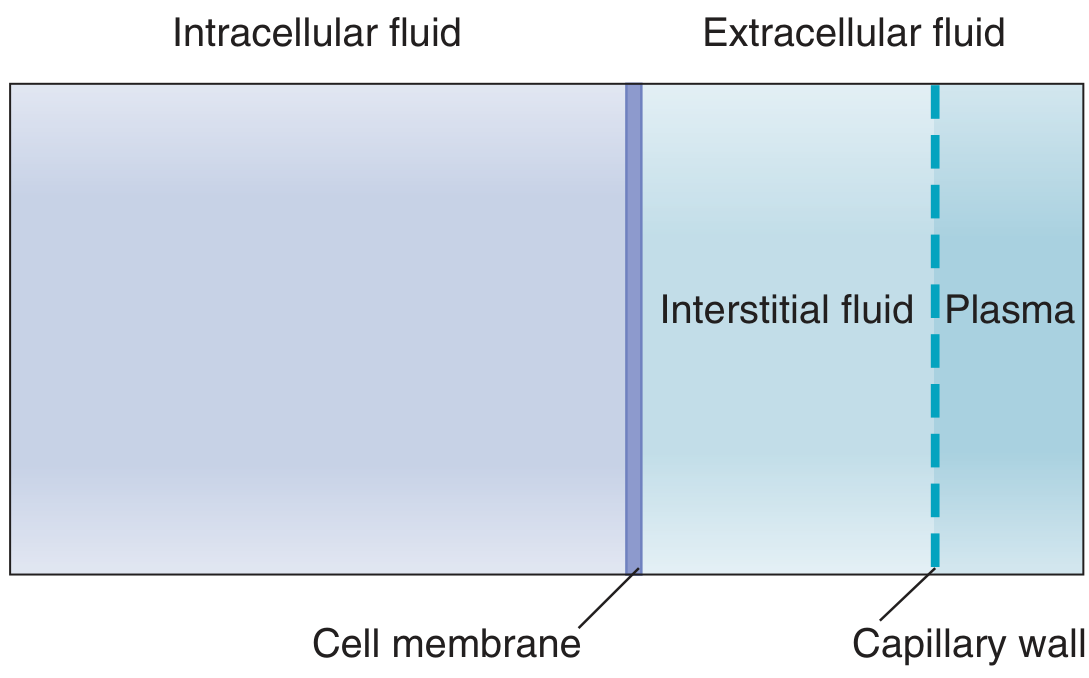
Fig. Body fluid compartments — Costanzo Physiology 7th Ed.
Summary Table (70 kg man)
| Compartment | % Body Weight | % TBW | Volume (L) |
|---|---|---|---|
| Intracellular (ICF) | 40 | 67 | 28 |
| Extracellular (ECF) | 20 | 33 | 14 |
| — Interstitial | 15 | 25 | 10.5 |
| — Intravascular (plasma) | 5 | 8 | 3.5 |
| Total | 60 | 100 | 42 |
Source: Morgan & Mikhail's Clinical Anaesthesiology, 7e, Table 49-1
A. Intracellular Fluid (ICF) — 28 L
- Two-thirds of TBW; contained within cells
- Bounded by the cell (plasma) membrane
- Dominant cation: K⁺ (140 mEq/L) — maintained by the Na⁺/K⁺-ATPase (3 Na⁺ out : 2 K⁺ in)
- High intracellular protein concentration (16 g/dL) acts as non-diffusible anion
- The 3:2 unequal exchange ratio of the pump is critical — without it, trapped intracellular proteins would create hyperosmolality and osmotic cell swelling
- Mg²⁺ (50 mEq/L) and phosphate (75 mEq/L) are the other major intracellular solutes
- Ischaemia or hypoxia → inhibition of Na⁺/K⁺-ATPase → progressive cell swelling
B. Extracellular Fluid (ECF) — 14 L
- One-third of TBW; outside cells
- Dominant cation: Na⁺ (145 mEq/L) — the primary determinant of ECF osmotic pressure and volume
- Changes in total body Na⁺ content = changes in ECF volume
1. Interstitial Fluid (ISF) — 10.5 L
- Fluid bathing the cells; forms from plasma by filtration across the capillary wall
- Capillary wall is virtually impermeable to plasma proteins → ISF protein content is very low (~2 g/dL)
- Most interstitial water is in gel form, bound to extracellular proteoglycans; free fluid is minimal
- Interstitial fluid pressure is normally slightly negative (≈ −5 mm Hg)
- When ISF volume progressively increases and pressure becomes positive → free fluid accumulates → clinical oedema
- The interstitial compartment acts as an overflow reservoir for the intravascular compartment
2. Intravascular Fluid (Plasma) — 3.5 L
- Confined to the vascular endothelium
- Small ions (Na⁺, K⁺, Cl⁻) freely cross the endothelium → plasma and ISF electrolyte composition are nearly identical
- Plasma proteins (albumin, 7 g/dL) do not cross capillary clefts → generate colloid oncotic pressure (~25 mm Hg), the main force retaining fluid within vessels
- Plasma proteins are the only osmotically active solutes not freely exchanged between plasma and ISF
3. Transcellular Fluid (minor compartment, ~1–2 L)
- Includes CSF, synovial, pleural, pericardial, peritoneal, intraocular, and GI secretions
- Functionally distinct because it is actively secreted and has a different composition
- Clinically relevant in conditions like bowel obstruction or ascites where large volumes accumulate as "third space"
Electrolyte Composition of Body Fluid Compartments
| Solute | ICF (mEq/L) | Plasma (mEq/L) | ISF (mEq/L) |
|---|---|---|---|
| Na⁺ | 10 | 145 | 142 |
| K⁺ | 140 | 4 | 4 |
| Ca²⁺ | <1 | 3 | 3 |
| Mg²⁺ | 50 | 2 | 2 |
| Cl⁻ | 4 | 105 | 110 |
| HCO₃⁻ | 10 | 24 | 28 |
| Phosphate | 75 | 2 | 2 |
| Protein (g/dL) | 16 | 7 | 2 |
Source: Morgan & Mikhail's Clinical Anaesthesiology, 7e, Table 49-2
III. OSMOLALITY AND TONICITY
A. Definitions and Distinctions
| Term | Definition |
|---|---|
| Osmosis | Net movement of water across a semipermeable membrane from low to high solute concentration |
| Osmotic pressure | Pressure required to prevent net water movement to the high-solute side |
| Osmolarity | Osmoles of solute per litre of solution (mOsm/L) |
| Osmolality | Osmoles of solute per kilogram of solvent/water (mOsm/kg H₂O) |
| Tonicity | Effective osmolality — the osmotic effect of a solution on cell volume |
In clinical practice, osmolarity ≈ osmolality (1 L water ≈ 1 kg water), so the terms are often used interchangeably. Normal value: 280–295 mOsm/kg H₂O.
- 1 mOsm/L difference between two solutions generates an osmotic pressure of 19.3 mm Hg
- Substances that ionize contribute n osmoles per mole, where n = number of ionic species (NaCl → 2 Osm theoretically, but due to ionic interaction NaCl behaves as though only ~75% ionized)
B. Calculation of Plasma Osmolality
$$\text{Plasma Osmolality} = 2 \times [Na^+] + \frac{\text{Glucose (mg/dL)}}{18} + \frac{\text{BUN (mg/dL)}}{2.8}$$
- Na⁺ multiplied by 2 to account for accompanying anions (Cl⁻ and HCO₃⁻)
- Normal: 280–295 mOsm/kg
- Osmolar gap = Measured − Calculated > 10 mOsm/kg → presence of unmeasured osmoles (methanol, ethanol, mannitol, ethylene glycol)
C. Tonicity vs. Osmolality — The Critical Distinction
Total osmolality includes all solutes, whether or not they cross cell membranes.
Tonicity (effective osmolality) includes only impermeant solutes — those that create sustained osmotic gradients across cell membranes.
Tonicity (effective osmolality) includes only impermeant solutes — those that create sustained osmotic gradients across cell membranes.
$$\text{Tonicity} \approx 2 \times [Na^+] + \frac{\text{Glucose (mg/dL)}}{18}$$
BUN is NOT included in tonicity — urea freely crosses cell membranes (via UT family transporters), equilibrates across the membrane, and therefore does not cause sustained changes in cell volume. It is an "ineffective osmole."
Clinical example:
- Adding mannitol (impermeant) to ECF → sustained cell shrinkage ✓
- Adding urea to ECF → transient cell shrinkage, then re-swelling as urea equilibrates ✗ (no sustained tonicity effect)
This is why hyperuraemia does not cause cellular dehydration, while hypernatraemia does.
D. Isotonic / Hypotonic / Hypertonic Solutions
| Classification | Effect on Cell Volume | Example |
|---|---|---|
| Isotonic (≈290 mOsm/L) | No change | 0.9% NaCl, Lactated Ringer's |
| Hypotonic (<290 mOsm/L) | Cell swelling (water enters) | 0.45% NaCl, free water |
| Hypertonic (>290 mOsm/L) | Cell shrinkage (water exits) | 3% NaCl, mannitol, hypertonic dextrose |
E. Fluid Exchange Between Compartments — Key Principles
- Volume of a compartment depends on its total solute content; ECF volume is governed by total body Na⁺
- In steady state, ICF osmolality = ECF osmolality — water shifts freely across membranes to maintain equality
- Solutes confined to ECF (NaCl, NaHCO₃, mannitol) — when added, expand only the ECF
- Permeant solutes (urea) distribute throughout TBW and do not change cell volume
Starling Forces across Capillaries (Intravascular ↔ Interstitial exchange):
$$J_v = K_f [(P_c - P_i) - \sigma(\pi_c - \pi_i)]$$
- Capillary hydrostatic pressure (P_c) → filtration out
- Plasma oncotic pressure (π_c) → reabsorption in
- Net: fluid filtered at arterial end, reabsorbed at venous end; ~10% excess enters lymphatics (~2 mL/min)
IV. PRIMARY METHODS OF REGULATION OF BODY FLUID WATER
Body water regulation is a closely integrated system involving the kidney, hypothalamus, and posterior pituitary, with secondary roles for the RAAS and natriuretic peptides.
A. Osmoreceptor–ADH (Vasopressin)–Renal Axis
The dominant mechanism for osmoregulation.
Sensing
- Osmoreceptor cells in the anterior hypothalamus (anterior to the 3rd ventricle, separate from but closely associated with supraoptic and paraventricular nuclei)
- Exquisitely sensitive — stimulated by increases in plasma osmolality of as little as 1–2% (~1–2 mOsm/kg above the set-point of ~285 mOsm/kg)
- Respond primarily to Na⁺ concentration in arterial blood
- Cell shrinkage = stimulation; cell swelling = inhibition
ADH (Vasopressin) Synthesis and Release
- Synthesized in supraoptic and paraventricular nuclei of the hypothalamus as prepro-hormone
- Transported down axons via the supraopticohypophysial tract → stored and released from the posterior pituitary
- Basal plasma ADH: 0.5–2 pg/mL → urine osmolality maintained above plasma (1–3 L/day urine)
- When ADH < 0.5 pg/mL → free water diuresis, urine osmolality < 100 mOsm/kg, up to 18–24 L/day
- At plasma osmolality ~295 mOsm/kg → urine maximally concentrated to 1000–1200 mOsm/kg
- Urine osmolality range: 50–1200 mOsm/L depending on ADH
Mechanism of Action — Aquaporin-2 (AQP2)
- ADH acts on V2 receptors in collecting duct principal cells
- V2 receptor → Gs protein → adenylate cyclase → ↑cAMP → PKA activation
- PKA phosphorylates aquaporin-2 (AQP2) vesicles → insertion into apical membrane of collecting duct
- ↑ Water permeability of late distal tubule and collecting duct → ↑ water reabsorption
- Water exits basolaterally through constitutively expressed AQP3 and AQP4 into medullary capillaries
| ADH Receptor | Location | Effect |
|---|---|---|
| V1 | Vascular smooth muscle | Vasoconstriction, ↑ vasomotor tone |
| V2 | Collecting duct principal cells | Antidiuresis via AQP2 insertion |
| V3 | Anterior pituitary | ↑ ACTH secretion |
Corticopapillary Osmotic Gradient (enables concentration)
- Medullary interstitium osmolality rises from cortex (~300 mOsm/L) to papilla (~1200 mOsm/L)
- Maintained by countercurrent multiplication (loop of Henle deposits NaCl in medullary interstitium) and urea recycling (inner medullary collecting duct deposits urea)
- ADH-driven water reabsorption relies on this pre-existing gradient
Negative Feedback Loop
↑ Plasma osmolality
↓
Osmoreceptor shrinkage → ↑ ADH + ↑ Thirst
↓
↑ Water reabsorption (kidney) + Drinking
↓
↓ Plasma osmolality (restored to normal)
B. Thirst Mechanism
- Threshold for thirst activation (~5–10 mOsm/kg above the ADH threshold) — slightly higher than for ADH
- Thirst centre is distributed across multiple brain areas; stimulated by osmoreceptors in the hypothalamus
- Under normal conditions, water balance is regulated more by ADH secretion than by thirst; in severe dehydration, thirst becomes essential
- Thirst is also stimulated by angiotensin II and by non-osmotic stimuli (volume depletion, dry mouth)
C. Non-Osmotic Regulation of ADH
ADH is also released in response to haemodynamic and other stimuli:
| Stimulus | Receptor/Mechanism | Effect on ADH |
|---|---|---|
| ↓ Effective circulating volume | Carotid/aortic baroreceptors, atrial stretch receptors | ↑ ADH (less sensitive than osmotic; ~10–15% volume loss needed) |
| Angiotensin II | Hypothalamic AT1 receptors | ↑ ADH + ↑ Thirst |
| Nausea/vomiting | Emetic stimulus | ↑↑ ADH (potent — relevant in post-op patients) |
| Pain, surgical stress | Sympathetic activation | ↑ ADH |
| Alpha catecholamines | ↓ ADH | |
| Beta catecholamines | ↑ ADH | |
| Glucocorticoid deficiency | ↑ ADH action + ↑ release | |
| Cortisol (excess) | ↓ ADH release | |
| Ethanol | ↓ ADH → diuresis | |
| Prostaglandins | ↓ ADH effect on kidney | |
| Chlorpropamide | ↑ ADH action | |
| Lithium / demeclocycline | ↓ ADH action (nephrogenic DI) | |
| Pregnancy | ↑ vasopressinase | ↓ effective ADH → polyuria |
Anaesthetic relevance: Major surgery, laryngoscopy, and post-operative nausea are potent non-osmotic stimuli for ADH release, contributing to post-operative hyponatraemia, especially when hypotonic IV fluids are given.
D. Renin–Angiotensin–Aldosterone System (RAAS) — Volume Regulation
While RAAS primarily regulates Na⁺ (and hence ECF volume), it secondarily affects water balance:
- ↓ Renal perfusion → ↑ Renin → ↑ Angiotensin I → ACE → Angiotensin II
- Angiotensin II: vasoconstriction + ↑ aldosterone + stimulates thirst and ADH release in hypothalamus
- Aldosterone → ↑ Na⁺/K⁺-ATPase in collecting duct → ↑ Na⁺ reabsorption → passive water follows
- Net effect: ↑ ECF volume
E. Atrial Natriuretic Peptide (ANP) / Brain Natriuretic Peptide (BNP)
- Released from atrial myocytes in response to ↑ atrial stretch (ECF volume expansion)
- Actions: ↑ GFR, ↓ aldosterone, ↓ ADH release, ↓ renin → natriuresis and diuresis
- Negative feedback on RAAS and ADH axis — buffers volume expansion
F. Renal Mechanisms — The Final Effector
The kidney integrates all signals to produce urine of variable osmolality:
| Segment | Function |
|---|---|
| Proximal tubule | Iso-osmotic reabsorption of 65% of filtered water (obligatory) |
| Loop of Henle (descending) | Freely permeable to water → water leaves into hyperosmotic medulla |
| Loop of Henle (ascending, thick) | Impermeable to water; active NaCl reabsorption → dilutes tubular fluid |
| Distal tubule / collecting duct | ADH-regulated water reabsorption via AQP2 (facultative) |
Urine osmolality variability (50–1200 mOsm/L) is almost entirely a function of ADH acting on the collecting duct, working against the corticopapillary gradient.
V. SUMMARY DIAGRAM — INTEGRATED WATER BALANCE
WATER INTAKE WATER LOSS
Oral fluids ←──────────────────→ Urine (primary regulated output)
Food water Insensible (skin + lungs ~800 mL/day)
Metabolic water (~300 mL/day) Sweat (variable)
Faeces (~100 mL/day)
Sensors: Osmoreceptors (hypothalamus) → ↑ Osmolality ≥ 1–2%
Baroreceptors → ↓ Volume ≥ 10–15%
Effectors:
1. ADH (vasopressin) → AQP2 → ↑ water reabsorption by collecting duct
2. Thirst → ↑ water intake
3. RAAS → ↑ Na⁺ reabsorption → passive water retention
4. ANP/BNP → ↑ natriuresis + ↓ ADH (volume overload correction)
VI. CLINICALLY RELEVANT ANAESTHETIC CORRELATIONS
| Clinical Scenario | Physiological Basis |
|---|---|
| Post-op hyponatraemia | Elevated ADH from surgical stress + hypotonic fluid administration → water retention → dilutional ↓ Na⁺ |
| Mannitol for ICP | Hypertonic → draws water from ICF to ECF → cellular dehydration; effective osmole |
| Hypertonic saline (3%) for severe hyponatraemia | Corrects ECF tonicity; rate-limited to avoid osmotic demyelination |
| Diabetes insipidus (central) | ↓ ADH → failure of AQP2 insertion → massive free water diuresis (>4 L/day), urine osmolality < 300 mOsm/kg |
| SIADH | Inappropriately elevated ADH → water retention → hypo-osmolar hyponatraemia with concentrated urine |
| Isotonic saline (0.9% NaCl) | Expands ECF only; does not shift into ICF (Na⁺ is impermeant) |
| Hypo-osmolar IV fluids | Distribute into all compartments proportional to their TBW share (ICF:ECF = 2:1) |
KEY EXAMINATION POINTS
- TBW = 60% body weight = 42 L in 70 kg man; ICF = 28 L (2/3), ECF = 14 L (1/3)
- Na⁺ governs ECF volume; K⁺ governs ICF osmolality — maintained by Na⁺/K⁺-ATPase
- Osmolality ≠ Tonicity: BUN contributes to osmolality but NOT to tonicity (freely permeable); glucose and Na⁺ are effective osmoles
- ADH set-point ~285 mOsm/kg; threshold ~280 mOsm/kg; maximal response ~295 mOsm/kg
- Thirst threshold is 5–10 mOsm/kg higher than ADH threshold
- AQP2 insertion in the apical membrane of collecting duct principal cells is the molecular mechanism of ADH action
- The corticopapillary gradient (300–1200 mOsm/L) is the permissive gradient; ADH action is the regulated switch
- Non-osmotic ADH release is a critical perioperative concern — nausea, pain, and surgical stress are potent stimuli
- Starling forces govern ICF↔ECF distribution; capillary oncotic pressure (albumin) retains fluid intravascularly
Sources: Morgan & Mikhail's Clinical Anaesthesiology 7e (pp. 2135–2142) | Costanzo Physiology 7th Ed. (pp. 9–12, 304–307) | Medical Physiology — Boron & Boulpaep (pp. 207–210) | Henry's Clinical Diagnosis & Management by Laboratory Methods | Goldman-Cecil Medicine (Vasopressin chapter)
This is a shared conversation. Sign in to Orris to start your own chat.