A note on Occupational diseases 2nd yr mbbs patho harsh mohan
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from Robbins Pathologic Basis of Disease and Robbins & Kumar Basic Pathology (the standard references equivalent to Harsh Mohan for 2nd year MBBS pathology). Let me compile the full notes.
Occupational Diseases — Pathology Notes (2nd Year MBBS)
(Based on Robbins Pathologic Basis of Disease & Robbins & Kumar Basic Pathology)
Definition
Pneumoconiosis — originally coined to describe lung disease caused by inhalation of mineral dusts in the workplace; now broadened to include diseases induced by organic and inorganic particulates, and chemical fumes and vapors.
Classification (Table)
| Agent | Disease | Occupation |
|---|---|---|
| Mineral Dusts | ||
| Coal dust | Anthracosis, Coal macules, Progressive massive fibrosis, Caplan syndrome | Coal mining (especially hard coal) |
| Silica | Silicosis, Caplan syndrome | Metal casting, sandblasting, hard rock mining, stone cutting |
| Asbestos | Asbestosis, Pleural plaques, Mesothelioma, Lung/larynx/stomach/colon carcinoma, Caplan syndrome | Mining, milling, insulation installation/removal |
| Beryllium | Acute beryllium disease, Beryllium granulomatosis | Mining, manufacturing |
| Iron oxide | Siderosis | Welding |
| Barium sulfate | Baritosis | Mining |
| Tin oxide | Stannosis | Mining |
| Organic Dusts (Hypersensitivity Pneumonitis) | ||
| Moldy hay | Farmer's lung | Farming |
| Bagasse (sugarcane) | Bagassosis | Wallboard, paper manufacturing |
| Bird droppings | Bird breeder's lung | Bird handling |
| Organic Dusts (Asthma) | ||
| Cotton, flax, hemp | Byssinosis | Textile manufacturing |
| Red cedar dust | Asthma | Lumbering, carpentry |
| Chemical Fumes/Vapors | ||
| NO₂, SO₂, NH₃, benzene, insecticides | Bronchitis, asthma, pulmonary edema, ARDS | Occupational/accidental exposure |
General Pathogenesis of Pneumoconioses
Key factors influencing disease development:
- Dust retention — determined by ambient dust concentration, duration of exposure, and effectiveness of mucociliary clearance (cigarette smoking significantly impairs clearance)
- Particle size — most dangerous particles: 1–5 μm in diameter (reach terminal airways and alveolar sacs); particles >5–10 μm are filtered in upper airways; particles <0.5 μm move in and out of alveoli without significant deposition
- Particle solubility & cytotoxicity — small, highly soluble injurious particles → rapid acute lung injury; larger insoluble particles → chronic fibrosis
- Inflammasome activation — phagocytosis by macrophages activates the inflammasome → release of IL-1, IL-18 → recruitment of inflammatory cells → fibroblast activation → collagen deposition
- Tobacco smoking — worsens ALL inhaled mineral dust diseases, most notably asbestos
The pulmonary alveolar macrophage is the key cellular element in initiation and perpetuation of inflammation, lung injury, and fibrosis.
1. Coal Workers' Pneumoconiosis (CWP)
Etiology
Inhalation of coal dust (mainly carbon) + contaminating silica and trace minerals.
Spectrum of Disease
| Stage | Features |
|---|---|
| Anthracosis | Most innocuous; carbon pigment engulfed by macrophages; accumulates along lymphatics; seen also in urban dwellers and smokers; asymptomatic |
| Simple CWP | Coal macules (1–2 mm, dust-laden macrophages + delicate collagen network) and coal nodules; upper lobes and upper zones of lower lobes; centrilobular emphysema may develop |
| Complicated CWP / PMF | Progressive Massive Fibrosis; requires years; intensely blackened scars >1–2 cm (up to 10 cm); dense collagen + pigment; necrosis of center due to ischemia |
Clinical Features
- Usually benign with little lung function decline
- PMF → progressive pulmonary dysfunction, pulmonary hypertension, cor pulmonale
- No increased risk of lung carcinoma (distinguishes CWP from silicosis and asbestosis)
- Less than 10% of simple CWP progresses to PMF
2. Silicosis
Etiology
Inhalation of crystalline silicon dioxide (silica) — most common: quartz (also cristobalite, tridymite). Most prevalent chronic occupational disease in the world.
At-risk workers: sandblasters, stone cutters, hard rock miners, jewelers, concrete demolition workers, stressed denim sandblasters.
Pathogenesis
- Silica phagocytosed by macrophages → inflammasome activation → IL-1 and IL-18 release → inflammatory cell recruitment → interstitial fibroblast activation → collagen deposition
- Crystalline forms far more fibrogenic than amorphous forms
- Clay coating of silica in coal/hematite miners reduces toxicity
- Disease is progressive even after exposure stops
Morphology
- Early: Tiny, pale-to-black discrete nodules in hilar lymph nodes and upper zones of lung
- Progressive: Nodules coalesce into hard, collagenous scars
- PMF: Expansion and coalescence of lesions
- Hallmark histology: Central area of whorled collagen fibers with peripheral zone of dust-laden macrophages; weakly birefringent particles under polarized microscopy
- Eggshell calcification of hilar lymph nodes — radiographic hallmark
- May cavitate due to superimposed tuberculosis or ischemia
Clinical Features
- Onset: Slow and insidious (most common, 10–30 years); accelerated (<10 years); acute/rapid (months after intense exposure — accumulation of lipoproteinaceous material, resembles alveolar proteinosis)
- Chest X-ray: fine nodularity in upper zones
- Pulmonary function: normal or mildly reduced early; worsens with PMF
- Increased susceptibility to tuberculosis (silica inhibits macrophage killing of mycobacteria) — "silicotuberculosis"
- 2-fold increased risk of lung cancer
3. Asbestos-Related Diseases
Types of Asbestos
| Form | Examples | Properties |
|---|---|---|
| Serpentine (chrysotile) | Chrysotile | Curly, flexible; 90% of industrial use; less pathogenic — trapped in upper airways, more soluble |
| Amphibole | Crocidolite, amosite, anthophyllite | Straight, stiff; less common but more pathogenic — penetrate deeper, less soluble; especially more carcinogenic |
Asbestos-Related Diseases (6)
- Asbestosis — parenchymal interstitial fibrosis
- Localized pleural plaques (most common manifestation) or, rarely, diffuse pleural fibrosis
- Recurrent pleural effusions
- Lung carcinoma
- Malignant mesothelioma (pleural and peritoneal)
- Laryngeal cancer (± ovarian, colon)
Pathogenesis
- Fibers land at bifurcations of small airways and ducts, penetrate, and are directly toxic
- Macrophages phagocytose fibers → inflammasome activation → reactive oxygen species, proteases, cytokines, growth factors → generalized interstitial fibrosis
- Asbestos acts as both tumor initiator and tumor promoter via free radical generation (iron in fibers)
- Tobacco smoke carcinogens adsorb onto asbestos fibers → remarkable synergy for lung carcinoma
Morphology — Asbestosis
- Diffuse pulmonary interstitial fibrosis — distinguished from other ILD only by presence of asbestos bodies
- Asbestos bodies: Golden-brown, fusiform or beaded rods with translucent center — asbestos fiber coated with iron-containing proteinaceous material (ferruginous bodies)
- Fibrosis begins at lower lobes and subpleurally (unlike CWP/silicosis which start upper lobes)
- Progresses → honeycomb pattern → pulmonary hypertension, cor pulmonale
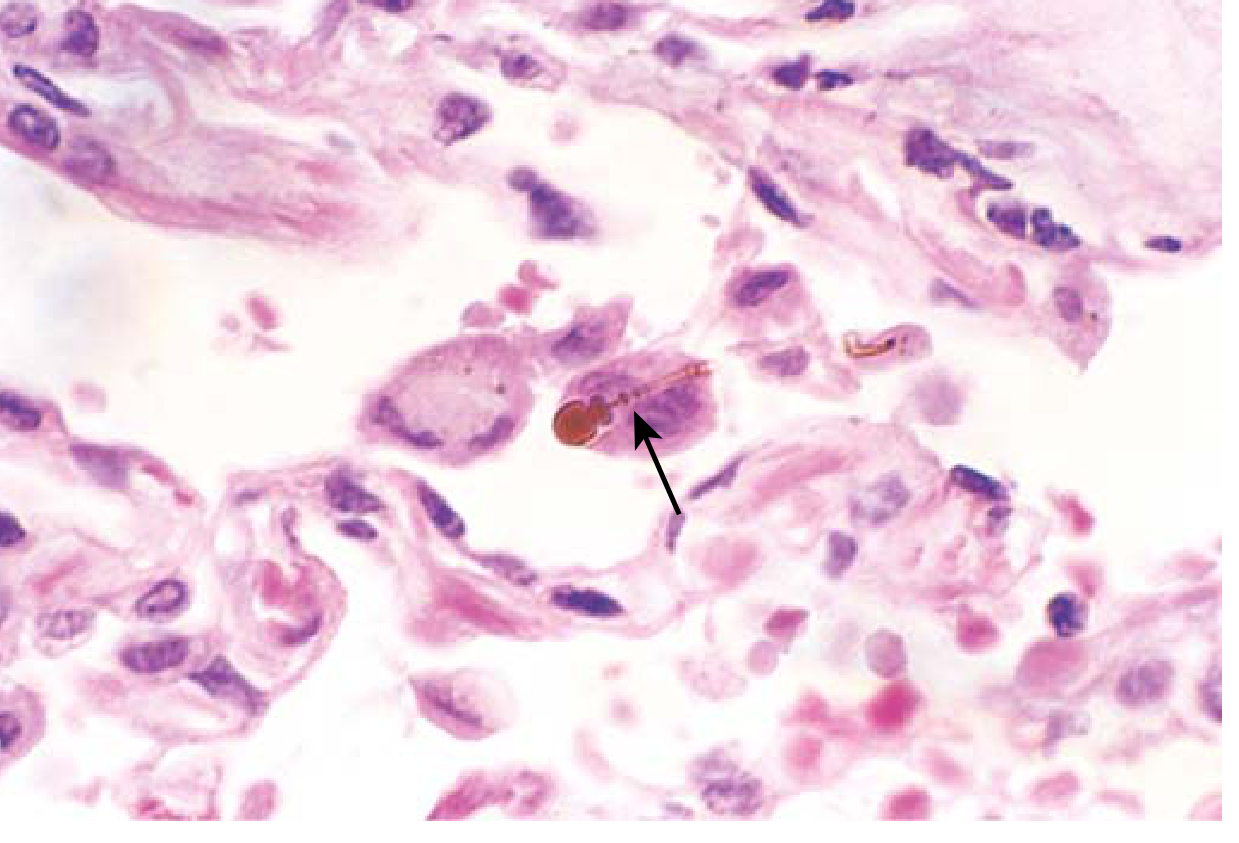
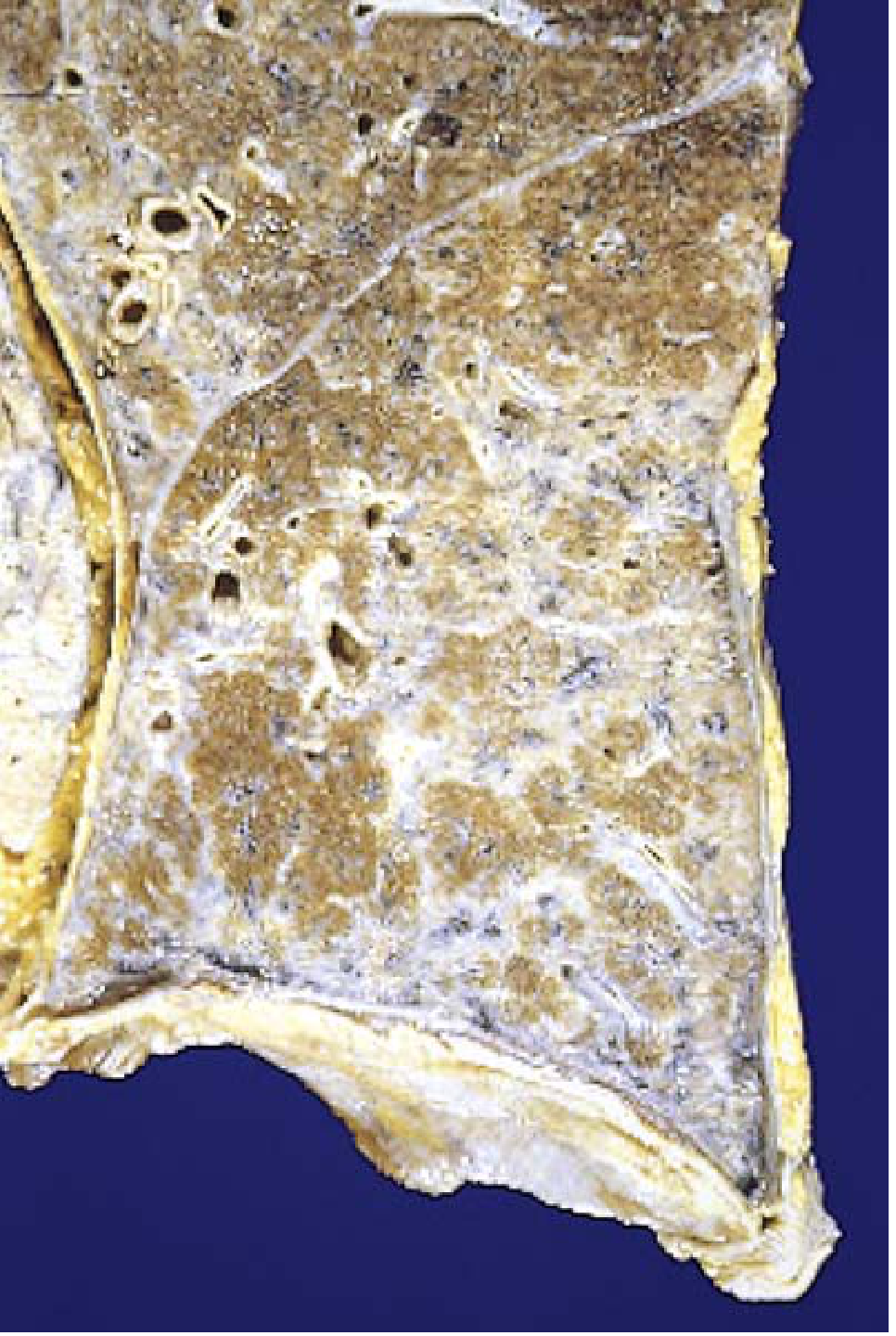
Morphology — Pleural Plaques
- Most common manifestation of asbestos exposure
- Well-circumscribed plaques of dense, often calcified collagen
- Anterior and posterolateral parietal pleura; over domes of diaphragm
- Usually asymptomatic; detected on X-ray
Clinical Features of Asbestosis
- Symptoms appear 10–20 years after first exposure (rarely <10 years)
- Dyspnea — first symptom (exertional → resting)
- Cough + sputum (usually due to smoking, not asbestosis itself)
- CXR: irregular linear densities in both lower lobes → honeycomb pattern
- May progress to respiratory failure, cor pulmonale, death
Cancer Risk
| Cancer | Relative Risk |
|---|---|
| Lung carcinoma | 5× increased with asbestos alone; 55× increased with asbestos + smoking |
| Mesothelioma | >1000× increased (normally very rare: 2–17/million) |
| Smoking + asbestos | Synergistic for lung carcinoma; smoking does NOT increase mesothelioma risk |
Family members of asbestos workers also have increased risk due to secondary exposure (asbestos fibers on clothing, etc.)
4. Caplan Syndrome
- Combination of pneumoconiosis (coal/silica/asbestos) + rheumatoid arthritis
- Large, well-defined, rapidly developing nodules (rheumatoid nodules) superimposed on simple pneumoconiosis
- Represents an immunologic reaction
5. Byssinosis
- Inhalation of cotton, flax, or hemp dust in textile manufacturing
- Organic dust-induced asthma
- "Monday morning tightness" — symptoms worst on first day back after weekend
6. Berylliosis
- Acute: Chemical pneumonitis
- Chronic: Beryllium granulomatosis — non-caseating granulomas; histologically identical to sarcoidosis — must be differentiated by occupational history
- Possible increased risk of lung carcinoma
Key Distinguishing Features (High-Yield Summary)
| Feature | CWP | Silicosis | Asbestosis |
|---|---|---|---|
| Dust | Coal | Crystalline silica | Asbestos |
| Location | Upper lobes | Upper lobes, hilar nodes | Lower lobes, subpleural |
| Hallmark lesion | Coal macule/nodule | Whorled collagen nodule; eggshell calcification | Asbestos bodies (ferruginous bodies) |
| Lung cancer risk | None (no increase) | 2× ↑ | 5× ↑ (55× with smoking) |
| Mesothelioma | No | No | Yes (>1000× ↑) |
| TB susceptibility | Mild | Yes (silicotuberculosis) | Less prominent |
| Progress after stopping exposure | May stop | Continues | Continues |
Sources
- Robbins Pathologic Basis of Disease (10th ed.) — Chapter 15 (Lung)
- Robbins & Kumar Basic Pathology — Chapter 11 (Lung)
Vitamin k
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the full note on Vitamin K.
Vitamin K — Complete Notes (2nd Year MBBS)
(Biochemistry + Pharmacology + Pathology perspective)
Overview
Vitamin K is a fat-soluble vitamin that serves as an essential coenzyme for the post-translational γ-carboxylation of specific glutamic acid residues in a number of proteins, most critically the clotting factors.
Forms of Vitamin K
| Form | Name | Source |
|---|---|---|
| K₁ | Phylloquinone | Green leafy vegetables — spinach, cabbage, kale, broccoli; beef liver, egg yolk |
| K₂ | Menaquinone (MK-n) | Synthesized by intestinal bacteria (gut flora); also present in fermented foods |
| K₃ | Menadione | Synthetic form; converted to K₂ in the body |
Chemistry
- Fat-soluble vitamin (quinone derivative)
- Active functional form: hydroquinone (reduced form)
- Inactive oxidized form: vitamin K epoxide
- Recycling enzyme: Vitamin K Epoxide Reductase (VKOR) — converts inactive epoxide back to active hydroquinone
Biochemical Mechanism of Action
γ-Carboxylation Reaction
Vitamin K acts as a cofactor for γ-glutamyl carboxylase (GGCX), which converts glutamic acid (Glu) residues → γ-carboxyglutamate (Gla) residues in target proteins.
Requirements for the reaction:
- γ-glutamyl carboxylase enzyme
- O₂ and CO₂
- Hydroquinone form of vitamin K (gets oxidized to epoxide during the reaction)
Why Gla Residues Matter
- Gla residues have two adjacent negatively charged carboxylate groups → excellent chelators of Ca²⁺ ions
- The protein-Ca²⁺ complex then binds to negatively charged phospholipid membranes on damaged endothelium and platelets
- This membrane attachment dramatically increases the rate of proteolytic activation of clotting factors (e.g., prothrombin → thrombin)
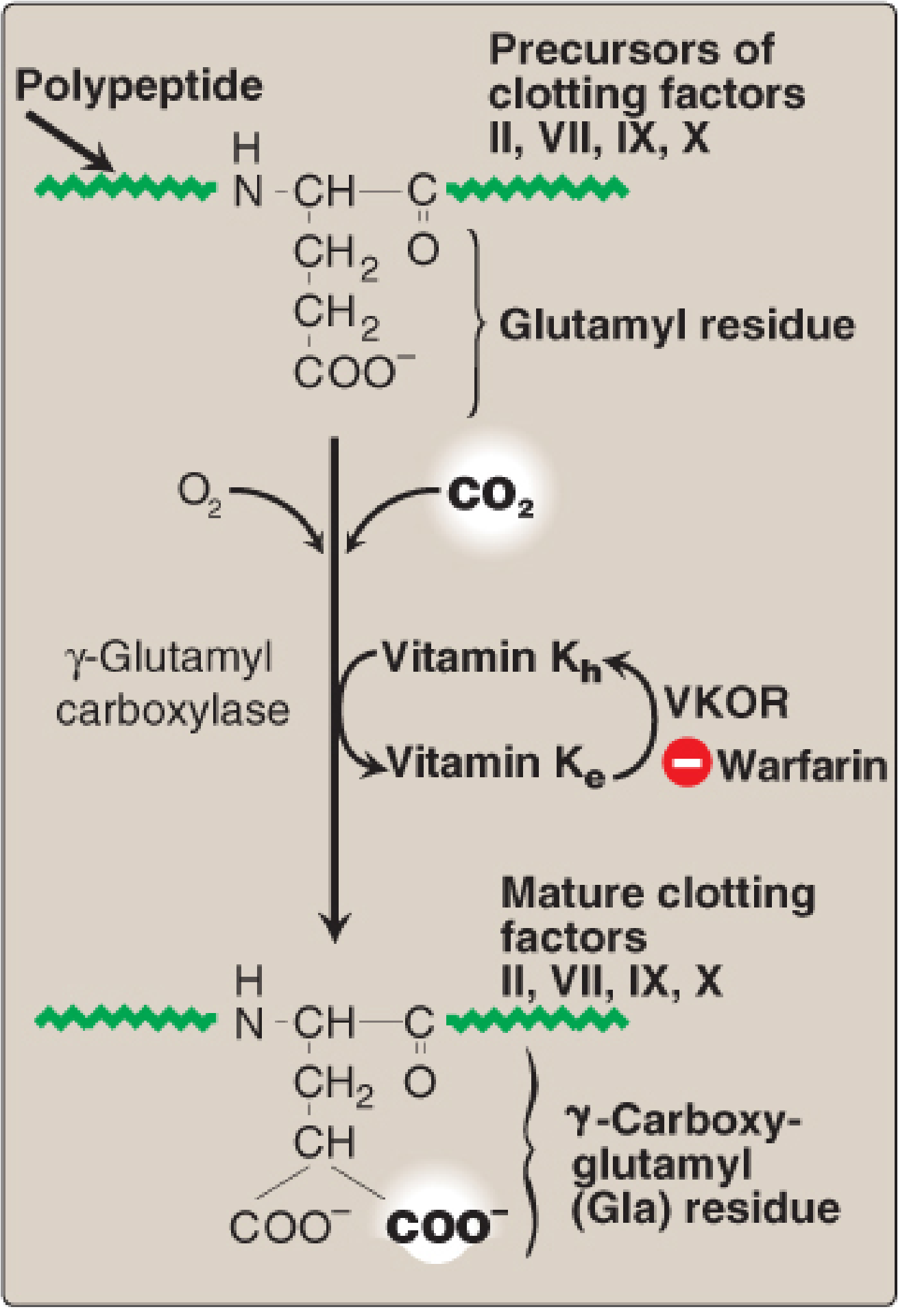
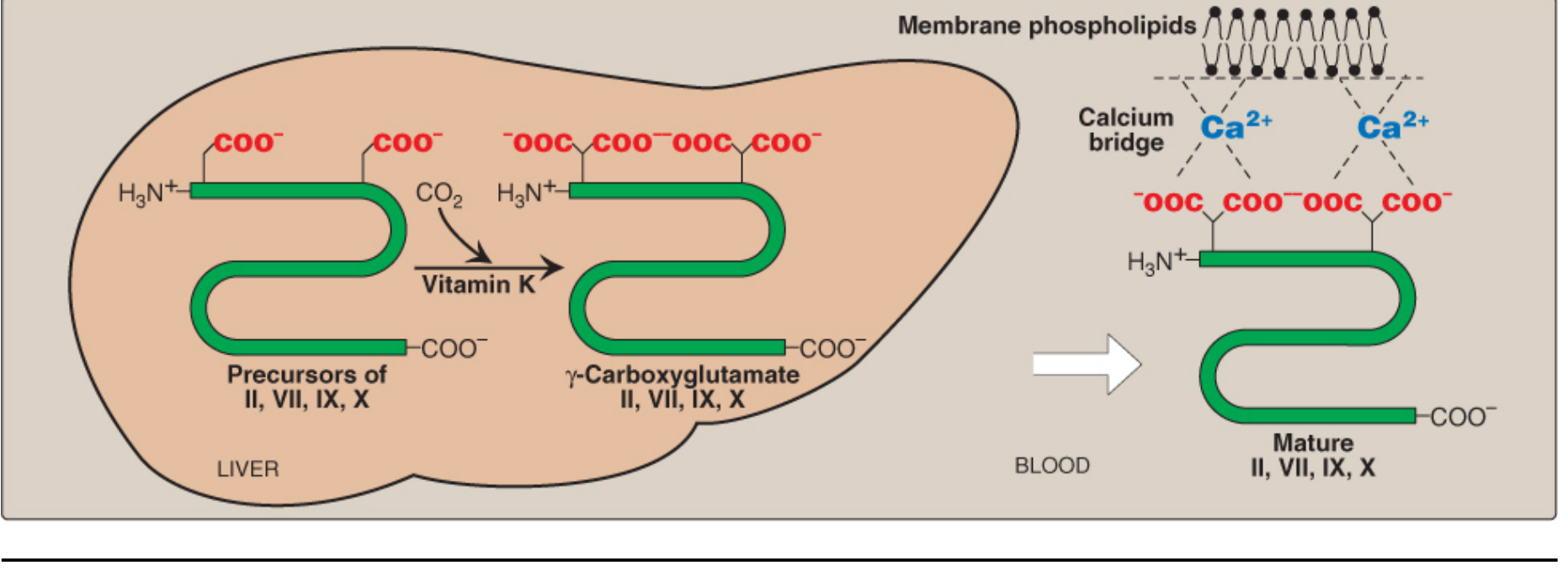
Vitamin K-Dependent Proteins
Procoagulant (Clotting Factors) — all synthesized in liver
| Factor | Name |
|---|---|
| Factor II | Prothrombin |
| Factor VII | Proconvertin |
| Factor IX | Christmas factor |
| Factor X | Stuart-Prower factor |
Mnemonic: 1972 (II, IX, VII, X) or "Ten PIG" (Ten = X; P = prothrombin/II; I = IX; G = VII)
Anticoagulant Proteins
- Protein C — inhibits Factors Va and VIIIa
- Protein S — cofactor for Protein C
Note: Vitamin K is required for both pro- and anticoagulant proteins. In early warfarin therapy, Protein C (short half-life) falls first → transient hypercoagulable state.
Bone and Other Proteins
- Osteocalcin — bone Gla protein; involved in bone mineralization
- Matrix Gla Protein (MGP) — inhibits vascular calcification
Dietary Requirement & Absorption
- Adult males: 120 μg/day (Adequate Intake)
- Adult females: 90 μg/day
- Absorption: Requires bile salts and dietary fat (fat-soluble vitamin); absorbed predominantly in the ileum
- Two major sources: dietary intake + gut bacterial synthesis
Vitamin K Cycle (VKOR Cycle)
Active hydroquinone (KH₂)
↓ [γ-glutamyl carboxylase + O₂ + CO₂]
Glu → Gla (on clotting factors)
↓
Inactive vitamin K epoxide (KO)
↓ [VKOR — Vitamin K Epoxide Reductase]
Active hydroquinone (KH₂) ← recycled
Warfarin/Coumarins block VKOR → cannot regenerate active vitamin K → inactive undercarboxylated clotting factors (called PIVKA — Proteins Induced by Vitamin K Absence/Antagonism)
Deficiency
Causes
- Poor dietary intake alone — rare (gut bacteria supplement dietary supply)
- Antibiotic therapy — destroys gut flora → reduced K₂ synthesis; especially dangerous when combined with poor oral intake (ICU patients)
- Fat malabsorption — obstructive jaundice (no bile salts), celiac disease, short bowel syndrome, IBD, cystic fibrosis
- Total Parenteral Nutrition (TPN) — without vitamin K supplementation
- Newborns — sterile gut (no bacteria); breast milk provides only ~⅕ of daily requirement → Hemorrhagic Disease of the Newborn (HDN)
- Warfarin/Coumarin therapy — competitive VKOR antagonism
- Certain cephalosporins (e.g., cefamandole) — warfarin-like VKOR inhibition
Consequences
- Bleeding diathesis (hypoprothrombinemia)
- Prolonged PT (most sensitive — Factor VII has shortest half-life, extrinsic pathway)
- Prolonged aPTT (intrinsic pathway factors IX, X affected)
- Undercarboxylated prothrombin must fall >50% before PT elevates
- Possible osteoporosis (loss of osteocalcin function)
- Vascular calcification (loss of MGP function)
Lab Tests in Deficiency
| Test | Result |
|---|---|
| PT (Prothrombin Time) | Prolonged (first and most sensitive) |
| aPTT | Prolonged |
| Bleeding time | Normal (platelets unaffected) |
| Platelet count | Normal |
| PIVKA-II | Elevated (undercarboxylated prothrombin) |
Hemorrhagic Disease of the Newborn (HDN)
- Newborns have sterile intestines → no K₂ synthesis
- Breast milk poor in vitamin K
- Presents: bleeding from umbilical stump, skin, GI tract, intracranial hemorrhage (late HDN)
- Prevention: Single IM dose of vitamin K₁ (phytomenadione, 1 mg) at birth — standard of care
Warfarin and Vitamin K — Antagonism
| Aspect | Detail |
|---|---|
| Mechanism | Warfarin competitively inhibits VKOR → blocks recycling of vitamin K epoxide → no active hydroquinone → factors II, VII, IX, X, Protein C, S remain inactive |
| Monitored by | PT/INR (extrinsic pathway, Factor VII) |
| Reversal | Oral/IV Vitamin K₁ (phytomenadione) — takes 12–24 hours for reversal |
| Emergency reversal | Fresh Frozen Plasma (FFP) or 4-factor Prothrombin Complex Concentrate (PCC) — immediate |
| Vitamin K bypasses | Warfarin block by providing sufficient substrate for carboxylation |
Toxicity
- Natural K₁ and K₂: No established Upper Limit (UL) — no known toxicity
- K₃ (Menadione — synthetic): Prolonged large doses → hemolytic anemia + jaundice in infants (toxic effects on RBC membrane) → no longer used therapeutically
Clinical Uses of Vitamin K
- Reversal of warfarin anticoagulation (supratherapeutic INR)
- Prevention of HDN — IM vitamin K at birth
- Liver disease with coagulopathy — trial of vitamin K to assess hepatic synthetic function
- Malabsorption-related coagulopathy — parenteral vitamin K (10 mg SC daily)
- Cephalosporin-associated hypoprothrombinemia — supplementation
For supratherapeutic INR (4.5–10.0) without active bleeding — stopping warfarin alone is usually sufficient; vitamin K administration provides no added benefit.
High-Yield Summary Points
| Feature | Detail |
|---|---|
| Type | Fat-soluble vitamin |
| Active form | Hydroquinone (KH₂) |
| Cofactor for | γ-glutamyl carboxylase (GGCX) |
| Reaction | Glu → Gla (γ-carboxylation) |
| Key enzyme inhibited by warfarin | VKOR |
| Clotting factors dependent | II, VII, IX, X, Protein C, Protein S |
| Most sensitive lab test | PT (Factor VII, shortest half-life) |
| Newborn prophylaxis | IM Vitamin K₁ at birth |
| Deficiency in newborn | Hemorrhagic Disease of Newborn |
| Toxic form | K₃ (menadione) — hemolytic anemia |
| Antidote for warfarin overdose | Vitamin K₁ + FFP/PCC for emergencies |
Sources: Lippincott Illustrated Reviews: Biochemistry, 8th ed. • Harper's Illustrated Biochemistry, 32nd ed. • Guyton & Hall Medical Physiology • Goldman-Cecil Medicine
Liver failure
Finding Sources
Reading File
Reading File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.