Endocrine Hormone types: steroids, peptides, amines. Major Glands - Pituitary, thyroid, adrenal, pancreas, gonads. Hormones: functions and regulation. Disorders - Diabetes, Cushing's syndrome, hypothyroidism.
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from the textbooks. Let me compile the full response.
Endocrine System: A Comprehensive Overview
1. Hormone Types: Chemical Classification
Hormones fall into three chemical classes that determine how they act on target cells.
Steroid Hormones
Derived from cholesterol. Include glucocorticoids (cortisol), mineralocorticoids (aldosterone), sex steroids (estrogen, progesterone, testosterone), and calcitriol (vitamin D).
Mechanism of action: Because steroids are lipid-soluble, they diffuse freely across the cell membrane and bind to intracellular (cytosolic or nuclear) receptor proteins. The hormone-receptor complex dimerizes and binds via zinc fingers to steroid-responsive elements (SREs) in the 5′ region of target genes, acting as a transcription factor that regulates mRNA synthesis → new protein production. This is why steroid effects are slow (hours), compared to peptide hormones.
"The hormone-receptor complex has now become a transcription factor that regulates the rate of transcription of that gene. New mRNA is transcribed, leaves the nucleus, and is translated to new proteins that have specific physiologic actions." — Costanzo Physiology, 7th Ed.
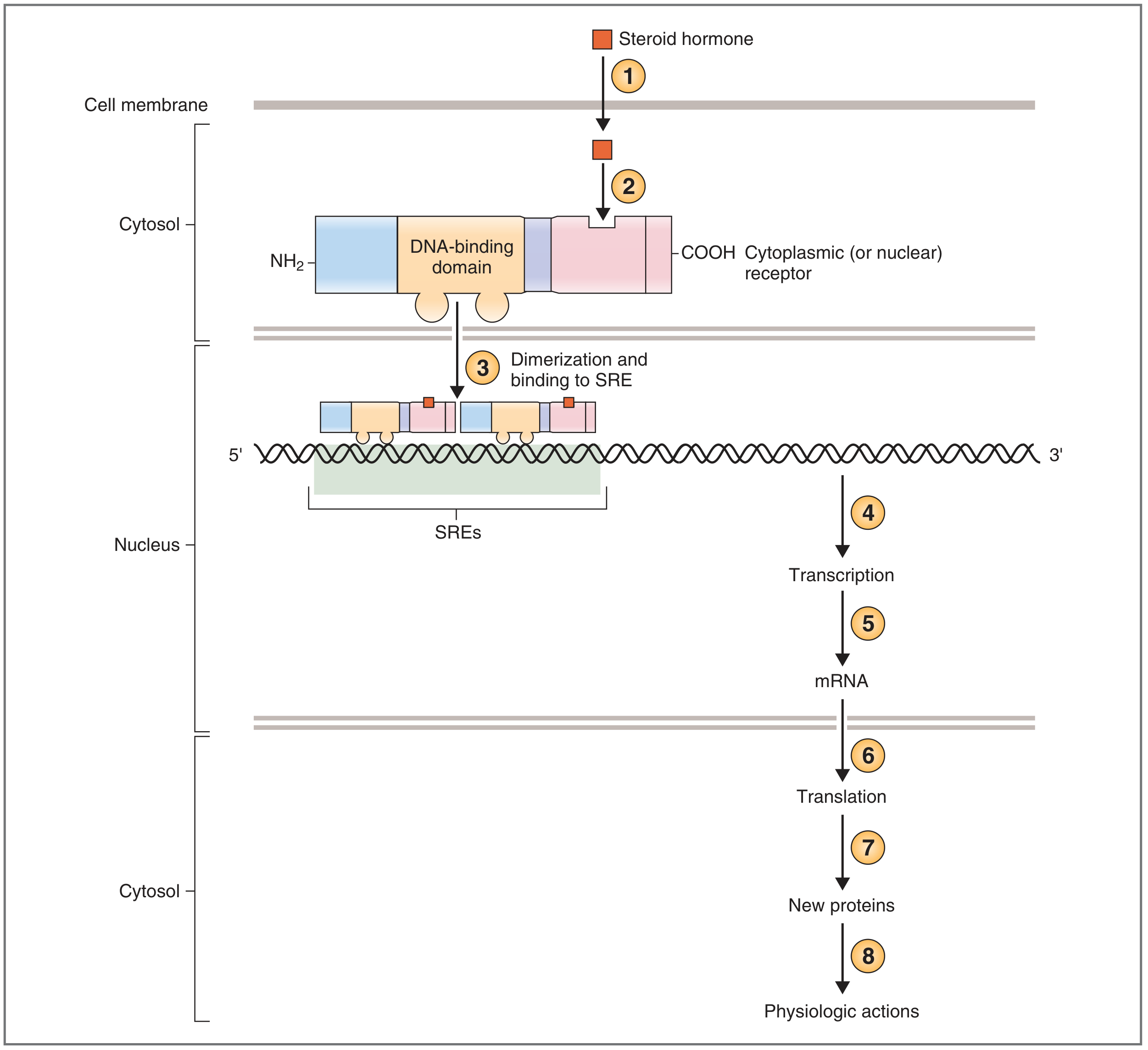
Peptide/Protein Hormones
Water-soluble; cannot cross the lipid bilayer. Include insulin, glucagon, ADH, oxytocin, GH, TSH, LH, FSH, PTH, and most hypothalamic releasing hormones.
Mechanism of action: Bind to cell surface receptors and act via second messengers:
- Adenylyl cyclase / cAMP pathway – hormone binds receptor → G protein (Gs) activates adenylyl cyclase → cAMP produced → protein kinase A activated → rapid cellular response. Examples: glucagon, TSH, ACTH, PTH.
- Phospholipase C / IP₃–Ca²⁺ pathway – G protein (Gq) activates PLC → IP₃ and DAG produced → intracellular Ca²⁺ rise → kinase activation. Examples: GnRH, TRH, oxytocin.
- Tyrosine kinase pathway – receptor itself has intrinsic kinase activity; ligand binding triggers receptor autophosphorylation and downstream signaling. Prime example: insulin.
Peptide hormones act rapidly (within minutes).
Amine Hormones
Derived from tyrosine (catecholamines, thyroid hormones) or tryptophan (melatonin).
| Amine | Source | Solubility | Mechanism |
|---|---|---|---|
| Epinephrine / Norepinephrine | Adrenal medulla | Water-soluble | Cell surface (α/β-adrenergic → cAMP/IP₃) |
| Thyroid hormones (T₃/T₄) | Thyroid follicular cells | Lipid-soluble | Nuclear receptor (like steroids) |
| Dopamine | Hypothalamus, CNS | Water-soluble | Cell surface (D receptors) |
2. Major Endocrine Glands: Hormones, Functions & Regulation
🧠 Pituitary Gland (Hypophysis)
The pituitary has two lobes with distinct embryological origins, connected to the hypothalamus by the infundibulum.
Anterior Pituitary (Adenohypophysis)
Regulated by hypothalamic releasing and inhibiting hormones delivered via the hypophyseal portal blood system:
| Hypothalamic Hormone | Effect on Anterior Pituitary |
|---|---|
| TRH (thyrotropin-releasing hormone) | ↑ TSH, prolactin |
| CRH (corticotropin-releasing hormone) | ↑ ACTH |
| GnRH (gonadotropin-releasing hormone) | ↑ LH, FSH |
| GHRH / somatostatin | ↑ / ↓ GH |
| Dopamine | ↓ Prolactin |
Anterior pituitary hormones:
- TSH (thyroid-stimulating hormone) → stimulates T₃/T₄ synthesis and release
- ACTH (adrenocorticotropic hormone) → stimulates cortisol secretion from adrenal cortex
- GH (growth hormone) → promotes IGF-1 production, anabolic/lipolytic effects
- LH / FSH (gonadotropins) → regulate gonadal steroidogenesis and gametogenesis
- Prolactin → stimulates mammary gland lactation
- MSH (melanocyte-stimulating hormone) → pigmentation
Regulation is chiefly by negative feedback: rising levels of target-gland hormones (e.g., cortisol, T₃/T₄, estrogen) suppress both hypothalamic releasing hormones and pituitary trophic hormones.
Posterior Pituitary (Neurohypophysis)
Not a true gland — it is a collection of axon terminals whose cell bodies are in the hypothalamus (supraoptic and paraventricular nuclei). Hormones are synthesized there and transported down axons for storage and release in the posterior lobe.
| Hormone | Stimulus for Release | Key Actions |
|---|---|---|
| ADH (vasopressin) | Increased plasma osmolality; hypovolemia | ↑ water reabsorption in renal collecting duct (V2 receptors); vasoconstriction (V1) |
| Oxytocin | Labour, suckling | Uterine contraction; milk ejection (let-down reflex) |
🦋 Thyroid Gland
Hormones: Thyroxine (T₄, ~93% secreted) and triiodothyronine (T₃, ~7%; biologically more potent). T₄ is converted to T₃ peripherally by deiodinases (Dio1, Dio2).
Functions of T₃/T₄:
- Increase basal metabolic rate (BMR) — calorigenic effect
- Essential for normal CNS development and myelination in infants
- Positive chronotropic/inotropic cardiac effects
- Permissive for GH and catecholamine actions
- Normal bone growth and maturation
Regulation: Classic hypothalamic-pituitary-thyroid (HPT) axis:
- Low T₃/T₄ → hypothalamus releases TRH → anterior pituitary releases TSH → TSH stimulates thyroid follicular cells (via cAMP) → T₃/T₄ synthesis and secretion
- Rising T₃/T₄ feeds back negatively to suppress TRH and TSH
Also secretes calcitonin (from parafollicular C-cells), which lowers serum Ca²⁺ by inhibiting osteoclast activity.
🫘 Adrenal Glands
Structurally divided into cortex (steroid hormones) and medulla (catecholamines).
Adrenal Cortex — Three Zones
| Zone | Hormone | Stimulus | Key Function |
|---|---|---|---|
| Zona glomerulosa | Aldosterone (mineralocorticoid) | Angiotensin II; hyperkalemia | ↑ Na⁺ reabsorption, ↑ K⁺/H⁺ excretion in kidney → ↑ ECF volume / BP |
| Zona fasciculata | Cortisol (glucocorticoid) | ACTH | ↑ gluconeogenesis, ↓ glucose uptake, lipolysis, protein catabolism; anti-inflammatory |
| Zona reticularis | DHEA (weak androgen) | ACTH | Precursor for sex steroids |
Cortisol is released in a diurnal pattern (peak early morning), amplified by stress. CRH → ACTH → cortisol axis, with cortisol negatively feeding back on both.
Adrenal Medulla
Chromaffin cells (modified postganglionic sympathetic neurons) secrete epinephrine (~80%) and norepinephrine (~20%) in response to preganglionic sympathetic stimulation (acetylcholine → nicotinic receptors). They are released during the fight-or-flight response: ↑ HR, ↑ BP, ↑ glycogenolysis, ↑ lipolysis, bronchodilation.
🫁 Pancreas (Islets of Langerhans)
| Cell Type | Hormone | Stimulus | Major Actions |
|---|---|---|---|
| β cells (~70%) | Insulin | ↑ blood glucose; amino acids; GIP; GLP-1; parasympathetic | ↓ blood glucose: ↑ GLUT4-mediated glucose uptake (muscle/fat), ↑ glycogenesis, ↑ lipogenesis, ↓ gluconeogenesis, ↓ glycogenolysis |
| α cells (~20%) | Glucagon | ↓ blood glucose; amino acids; exercise; sympathetic | ↑ blood glucose: ↑ glycogenolysis, ↑ gluconeogenesis in liver |
| δ cells | Somatostatin | Post-meal nutrients (glucose, amino acids, fatty acids) | Locally inhibits both insulin and glucagon; slows GI motility and absorption |
Insulin acts via a receptor tyrosine kinase (tyrosine kinase mechanism): insulin binding → receptor autophosphorylation → IRS-1 activation → PI3K/Akt cascade → GLUT4 translocation.
Normal fasting blood glucose: 80–90 mg/100 mL; postprandial peak ~120–140 mg/100 mL, restored by feedback within 2 hours.
🧬 Gonads
| Gland | Hormones | Regulation | Functions |
|---|---|---|---|
| Testes | Testosterone (Leydig cells); Inhibin (Sertoli cells) | LH → testosterone; FSH → spermatogenesis | Spermatogenesis, virilization, libido, anabolic effects, bone/muscle mass |
| Ovaries | Estradiol (granulosa cells), Progesterone (corpus luteum), Inhibin | FSH → estradiol; LH surge → ovulation; LH → progesterone | Folliculogenesis, endometrial proliferation; progesterone: secretory phase, maintains pregnancy |
Negative feedback: estradiol/testosterone suppress GnRH and LH/FSH. Mid-cycle positive feedback from rising estradiol triggers the LH surge → ovulation.
3. Major Endocrine Disorders
🔴 Diabetes Mellitus
"Diabetes mellitus is a syndrome of impaired carbohydrate, fat, and protein metabolism caused by either lack of insulin secretion or decreased sensitivity of the tissues to insulin." — Guyton & Hall Textbook of Medical Physiology
| Feature | Type 1 | Type 2 |
|---|---|---|
| Mechanism | Autoimmune/viral destruction of β cells → absolute insulin deficiency | Insulin resistance → relative deficiency; eventual β-cell failure |
| Onset | Typically childhood/adolescent (any age) | Adult (increasingly younger) |
| Body habitus | Usually lean | Usually obese (esp. visceral adiposity) |
| Ketosis | Prone (DKA) | Uncommon (unless stressed) |
| Prevalence | ~5–10% of DM | ~90–95% of DM |
| Treatment | Insulin replacement | Lifestyle, metformin, SGLT2i, GLP-1 RA, insulin if needed |
Pathophysiology of hyperglycemia: Absent/ineffective insulin → reduced peripheral glucose uptake (GLUT4 not translocated) + increased hepatic gluconeogenesis → plasma glucose can reach 300–1200 mg/dL. The renal threshold (~180–200 mg/dL) is exceeded → glycosuria → osmotic diuresis → polyuria, polydipsia, polyphagia (the classic triad). Chronic hyperglycemia → non-enzymatic glycation of proteins, oxidative stress → microvascular (retinopathy, nephropathy, neuropathy) and macrovascular complications.
Insulin resistance contributors: Obesity (visceral fat), Cushing syndrome, acromegaly, PCOS, glucocorticoid therapy, lipodystrophy, insulin receptor mutations.
🟠 Cushing's Syndrome (Hypercortisolism)
Definition: State of excess glucocorticoid activity, either exogenous or endogenous.
Causes:
- Exogenous (most common overall): iatrogenic glucocorticoid therapy
- ACTH-dependent (endogenous):
- Cushing disease (pituitary ACTH-secreting adenoma) — 80–90% of endogenous cases → bilateral adrenal hyperplasia
- Ectopic ACTH (small-cell lung cancer, bronchial carcinoids) — 10–20%
- ACTH-independent (10–25%): primary adrenal adenoma or carcinoma (suppressed ACTH)
Distinguish: ACTH level is the key — elevated in pituitary/ectopic disease; suppressed (<5 pg/mL) in primary adrenal causes. Pituitary ACTH in Cushing disease is typically >100× elevated on high-dose dexamethasone suppression.
Clinical features:
- Centripetal obesity: truncal fat, moon facies, buffalo hump, thin limbs
- Skin: purple/violaceous striae, easy bruising, thin skin
- Musculoskeletal: proximal muscle wasting, osteoporosis, pathologic fractures
- Metabolic: hyperglycemia (glucose intolerance → diabetes), hypokalemia (mineralocorticoid effect of cortisol), hypertension
- Neuropsychiatric: emotional lability, depression, psychosis
- Endocrine: hirsutism and menstrual irregularity in women; growth retardation in children
Treatment: Directed at the underlying cause — surgical resection of pituitary adenoma (transsphenoidal), adrenalectomy for adrenal tumor, or tumor resection for ectopic ACTH.
🟡 Hypothyroidism
Definition: Insufficient thyroid hormone production or action.
Primary hypothyroidism (thyroid gland failure):
- Hashimoto thyroiditis (autoimmune) — most common in iodine-sufficient regions
- Iodine deficiency (most common worldwide)
- Post-radioiodine/surgical thyroidectomy
- TSH ↑ (compensatory), T₃/T₄ ↓
Secondary/tertiary hypothyroidism: Pituitary (TSH deficiency) or hypothalamic (TRH deficiency) failure; TSH low/normal with low T₄.
Clinical features:
- Fatigue, cold intolerance, weight gain, constipation, bradycardia
- Dry skin, coarse hair, hair loss
- Periorbital puffiness, myxedema (non-pitting oedema — glycosaminoglycan deposition)
- Delayed deep tendon reflexes
- Hyperlipidaemia (LDL↑), hyponatremia
- Cognitive slowing, depression
- In infants: cretinism (irreversible intellectual disability and short stature) if untreated
Treatment: Levothyroxine (synthetic T₄), standard dose ~1.7 µg/kg/day. Goal: normalize TSH in primary hypothyroidism, or free T₄ in secondary. Dose starts low in elderly and cardiac patients (12.5–50 µg/day); titrated every 6 weeks. Levothyroxine monotherapy is the evidence-based standard; T₄+T₃ combination is not routinely superior. Dose must be increased ~30% during pregnancy.
Quick-Reference Summary Table
| Gland | Key Hormones | Type | Regulation | Disorder |
|---|---|---|---|---|
| Anterior pituitary | TSH, ACTH, GH, LH/FSH, Prolactin | Peptide | Hypothalamic releasing hormones; negative feedback | Panhypopituitarism, acromegaly, Cushing disease |
| Posterior pituitary | ADH, Oxytocin | Peptide | Neural (osmoreceptors, baroreceptors, suckling) | Diabetes insipidus, SIADH |
| Thyroid | T₃/T₄, Calcitonin | Amine (T₃/T₄); Peptide (calcitonin) | TRH/TSH axis; negative feedback | Hypothyroidism, hyperthyroidism, goitre |
| Adrenal cortex | Cortisol, Aldosterone, DHEA | Steroid | ACTH (cortisol); RAAS/K⁺ (aldosterone) | Cushing's, Addison's, Conn's |
| Adrenal medulla | Epinephrine, Norepinephrine | Amine | Sympathetic nervous system | Pheochromocytoma |
| Pancreas | Insulin, Glucagon, Somatostatin | Peptide | Blood glucose, amino acids, autonomic | Diabetes mellitus, insulinoma |
| Testes/Ovaries | Testosterone, Estradiol, Progesterone | Steroid | GnRH/LH/FSH axis | Hypogonadism, PCOS |
Sources:
- Costanzo Physiology, 7th Ed. — Mechanisms of hormone action, hypothalamic-pituitary relationships
- Guyton & Hall Textbook of Medical Physiology — Pancreatic hormones, diabetes mellitus, glucose regulation
- Mulholland & Greenfield's Surgery, 7th Ed. — Cushing's syndrome, hypercortisolism
- Goodman & Gilman's Pharmacological Basis of Therapeutics — Thyroid hormone replacement therapy
Dura master archoniod matter pia matter
Reading File
Finding Sources
Finding Sources
Searching Images
meninges dura mater arachnoid pia mater layers brain anatomy
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Excellent content. Now let me compile a comprehensive, well-illustrated response.
The Meninges: Dura Mater, Arachnoid Mater & Pia Mater
The meninges are three concentric connective tissue membranes that surround, protect, and suspend the brain and spinal cord within the cranial cavity and vertebral canal. From outermost to innermost: Dura → Arachnoid → Pia (mnemonic: PAD).
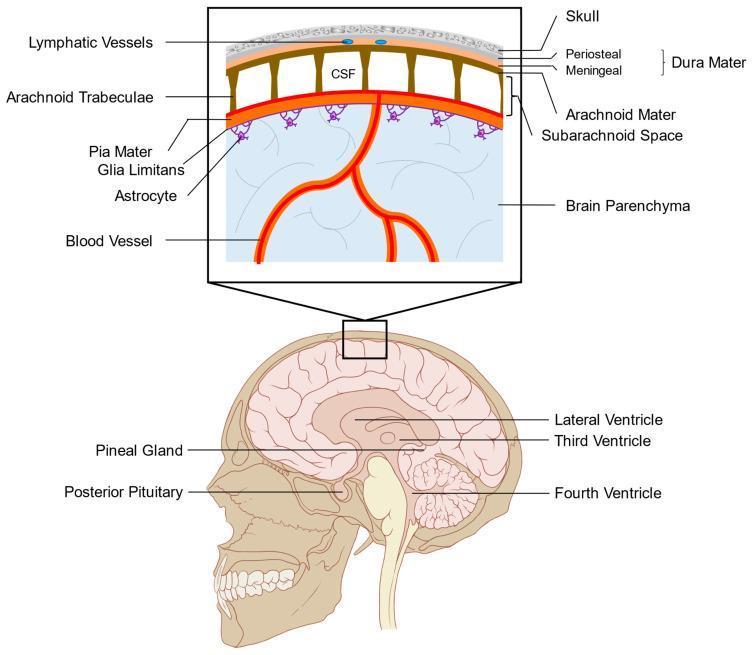
1. Dura Mater ("Tough Mother" / "Hard Mother")
The thickest, outermost meningeal layer. Composed of dense irregular collagenous connective tissue.
Structure — Two Layers (in the Cranium)
| Layer | Description |
|---|---|
| Outer periosteal layer | Fused with the inner periosteum of the skull; acts as the cranial periosteum |
| Inner meningeal layer | Provides the actual meningeal covering; forms dural folds |
These two layers are normally fused, but separate in specific locations to form dural venous sinuses (e.g., superior sagittal sinus) — blood-filled channels that drain venous blood from the brain.
Dural Folds (Reflections of the Inner Layer)
The inner meningeal layer folds inward to form two major partitions:
| Fold | Location | Significance |
|---|---|---|
| Falx cerebri | Runs vertically in the interhemispheric fissure | Separates right and left cerebral hemispheres |
| Tentorium cerebelli | Horizontal tent-like sheet over the cerebellum | Separates supratentorial (cerebral hemispheres) from infratentorial (cerebellum, brainstem) compartments |
"The falx cerebri [is] a flat sheet of dura suspended from the roof of the cranium and separating the right and left cerebral hemispheres. The tentorium cerebelli [is] a tentlike sheet of dura that covers the upper surface of the cerebellum." — Neuroanatomy through Clinical Cases, 3rd Ed.
Spinal Dura
- Separated from the periosteum of the vertebrae by the epidural space (contains fat and venous plexuses — the target for epidural anesthesia)
- Only one layer (the outer periosteal layer stays with the bone)
Clinical — Extradural (Epidural) Hematoma
The space between the dura and skull is normally potential (no real space). Rupture of the middle meningeal artery (e.g., temporal bone fracture) bleeds into this space → extradural hematoma — a neurosurgical emergency with classic lucid interval.
2. Arachnoid Mater ("Spider-Web Mother")
Named for its web-like appearance (Greek: arachnoeides = spider-like). The middle layer, pressed against the internal surface of the dura.
Structure — Two Components
- Outer sheet — flat layer of connective tissue closely apposed to the dura
- Trabeculae — loose, web-like extensions of collagen and fibroblasts that span the subarachnoid space and connect to the pia mater below
The arachnoid is avascular (no nutritive capillaries), though larger blood vessels course through it. It does not follow the brain's contours — it bridges over gyri and sulci, creating the subarachnoid space.
Arachnoid Villi & Granulations
Finger-like projections where arachnoid penetrates the dura and protrudes into dural venous sinuses (especially the superior sagittal sinus):
- Arachnoid villi → individual projections
- Arachnoid granulations (Pacchionian bodies) → clustered villi
- Function: Reabsorb CSF back into venous blood → critical for CSF homeostasis
Clinical — Subdural Hematoma
No true subdural space exists anatomically — bleeding here represents dissection of the dural border cell layer (the innermost cells of the meningeal dura). Tearing of bridging cerebral veins (crossing from cortex into the superior sagittal sinus) causes a subdural hematoma — common in elderly (brain atrophy stretches bridging veins) and after trauma.
"The arachnoid, not the dura, hinders drug movement through the meninges — because of its cellular architecture." — Barash's Clinical Anesthesia, 9th Ed.
3. Pia Mater ("Tender/Soft Mother")
The innermost, thinnest meningeal layer. Directly and intimately applied to the surface of the brain and spinal cord.
Structure
- Composed of flattened mesenchymally-derived cells
- Follows every contour of the brain — dips into every gyrus, sulcus, and fissure
- Does not directly contact neurons; separated from the neural parenchyma by the glia limitans (a thin layer of astrocytic foot processes that firmly adheres to the pia)
- Together, pia + glia limitans form the pial-glial membrane, which separates CNS tissue from CSF
Blood Vessels & Perivascular Spaces
Blood vessels entering the brain are initially covered by pia mater and travel through perivascular (Virchow-Robin) spaces. As vessels branch into capillaries, the pia disappears, and capillaries are covered only by astrocytic processes (contributing to the blood-brain barrier).
Pia-Arachnoid = Leptomeninges
The pia and arachnoid are closely associated and collectively called the leptomeninges ("thin membranes"), in contrast to the dura alone, which is the pachymeninx ("thick membrane"). Inflammation of the leptomeninges = meningitis.
The Meningeal Spaces
| Space | Between | Contents | Real or Potential? |
|---|---|---|---|
| Extradural (epidural) | Dura & skull (cranium) / Dura & vertebral periosteum (spine) | Nothing normally (cranium); fat + veins (spine) | Potential (cranium); Real (spine) |
| Subdural | Dura & arachnoid | Nothing (artifact of dural border cell layer dissection) | Potential |
| Subarachnoid | Arachnoid & pia | CSF + blood vessels + trabeculae | Real (normally fluid-filled) |
"The only normally occurring fluid-filled space associated with the meninges is the subarachnoid space." — Gray's Anatomy for Students
Subarachnoid Space Details
- Contains cerebrospinal fluid (CSF) — clear, colorless, produced by choroid plexus in the ventricles
- Enlarged areas = subarachnoid cisterns (e.g., cisterna magna, pontine cistern)
- CSF circulates here around the entire brain and spinal cord
- Reabsorbed via arachnoid granulations into the superior sagittal sinus
- Subarachnoid hemorrhage (SAH): blood in this space — typically from ruptured berry aneurysm; presents with "thunderclap headache"
Summary Diagram
SKULL BONE
│
EXTRADURAL SPACE (potential; real in spine)
│
DURA MATER ── periosteal layer (fused with skull)
── meningeal layer (forms dural folds & venous sinuses)
│
SUBDURAL SPACE (potential; site of subdural hematoma)
│
ARACHNOID MATER ── outer sheet (against dura)
── trabeculae (spider-web bridges)
│
SUBARACHNOID SPACE ◄── CSF + blood vessels (REAL SPACE)
│ ◄── arachnoid villi drain CSF to venous sinuses
PIA MATER ── closely follows all brain/cord contours
│
GLIA LIMITANS (astrocytic foot processes)
│
BRAIN / SPINAL CORD
Clinical Summary
| Condition | Space/Structure Involved | Mechanism |
|---|---|---|
| Epidural hematoma | Extradural space | Rupture of middle meningeal artery (temporal fracture) |
| Subdural hematoma | Dural border cell layer | Tearing of bridging veins (trauma, elderly) |
| Subarachnoid hemorrhage | Subarachnoid space | Ruptured cerebral aneurysm; "thunderclap" headache |
| Meningitis | Leptomeninges (pia + arachnoid) | Infection/inflammation; CSF pleocytosis on LP |
| Lumbar puncture | Subarachnoid space (L3–L4) | Needle passes: skin → ligaments → epidural space → dura → subdural → arachnoid → subarachnoid |
| Hydrocephalus | Ventricular/subarachnoid system | Obstruction of CSF flow or failed reabsorption at arachnoid villi |
Sources:
- Gray's Anatomy for Students — Meninges, spaces, arrangement
- Junqueira's Basic Histology, 17th Ed. — Histology of dura, arachnoid, pia
- Neuroanatomy through Clinical Cases, 3rd Ed. — Dural folds, falx cerebri, tentorium cerebelli
- Morgan & Mikhail's Clinical Anesthesiology, 7th Ed. — Spinal meninges
Digestive System: Anatomy - Mouth, Pharynx, Esophagus, Deglutition, Stomach, Pancreas, Liver and Gallbladder, Small Intestine, Large Intestine, Phases of Digestion - Mechanical and chemical processes. Absorption of nutrients. Disorders - GERD, ulcers, celiac disease
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching Images
digestive system anatomy overview gastrointestinal tract diagram
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.