Pathophysiology of asthma in PG level
Reading File
Finding Sources
Finding Sources
Searching PubMed
"asthma"[MeSH Terms] AND "pathophysiology"[MeSH Terms]
Reading File
Reading File
Reading File
Searching Images
asthma pathophysiology airway inflammation diagram
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material from top-tier sources. Let me compile the complete PG-level answer:
Pathophysiology of Asthma
Sources: Robbins & Cotran Pathologic Basis of Disease (10th ed.), Fishman's Pulmonary Diseases and Disorders, Murray & Nadel's Textbook of Respiratory Medicine
Definition & Core Triad
Asthma is a chronic inflammatory disease of the airways characterized by three interacting pathophysiologic components:
- Airway hyperresponsiveness (AHR)
- Variable airflow obstruction (reversible bronchoconstriction)
- Chronic airway inflammation with structural remodeling
The complex interplay between these factors — not any one element alone — defines the clinical expression of disease.
Types of Asthma
| Type | Mechanism |
|---|---|
| Atopic (Allergic) | Th2/IgE-mediated; most common (65–80%) |
| Non-atopic (Intrinsic) | Non-IgE; triggered by cold, exercise, pollutants |
| Drug-induced | ASA/NSAIDs via COX inhibition → ↑ leukotrienes |
| Occupational | Sensitization to workplace antigens |
Central Immunopathogenesis: T2 Inflammatory Pathway
Step 1 — Allergen Sensitization (Adaptive Response)
- Inhaled allergen is trapped in the mucus layer and endocytosed by dendritic cells (DCs) whose dendrites interdigitate between bronchial epithelial cells.
- DCs migrate to local lymph nodes, presenting antigen via MHC II to naïve CD4⁺ T cells.
- Under the influence of IL-4 (from mast cells and basophils), naïve T cells differentiate into Th2 cells.
- Th2 cells secrete the critical cytokine triad:
- IL-4 → drives IgE class switching in B cells; promotes AHR in smooth muscle
- IL-5 → eosinophil maturation, recruitment, and activation
- IL-13 → goblet cell mucus hypersecretion; promotes IgE production
- IgE produced by plasma cells binds the high-affinity FcεRI receptors on submucosal mast cells and circulating basophils.
Step 2 — Re-exposure: Mast Cell Activation
On re-exposure, allergen cross-links IgE on mast cells → degranulation.
Preformed mediators released:
- Histamine
- Tryptase, chymase, heparin
- TNF-α, VEGF
Newly synthesized mediators:
- Leukotrienes (LTC4, LTD4, LTE4): potent, prolonged bronchoconstriction + ↑ vascular permeability + ↑ mucus
- PGD2: bronchoconstriction + vasodilation
- Thromboxane A2, PAF
- Cytokines: IL-4, IL-5, IL-8, IL-13, GM-CSF
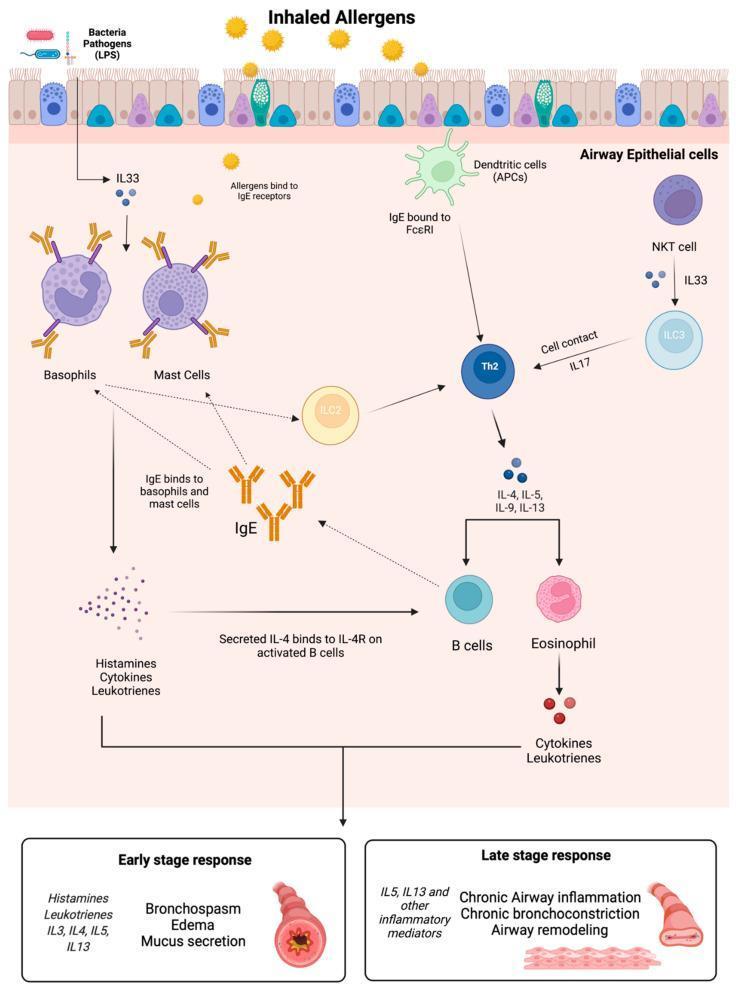
Biphasic Bronchial Response
Early Phase Reaction (Minutes 0–30)
- Bronchoconstriction: mast cell–released histamine and leukotrienes act directly + via vagal reflexes on subepithelial parasympathetic receptors → airway smooth muscle (ASM) contraction
- Increased vascular permeability → edema
- Mucus hypersecretion (goblet cell stimulation)
- Vasodilation
Late Phase Reaction (Hours 2–8)
- Recruitment of eosinophils, neutrophils, basophils, lymphocytes, monocytes to the airway wall
- Eosinophil-derived:
- Major Basic Protein (MBP), Eosinophil Cationic Protein (ECP) → epithelial damage, ciliary dysfunction
- LTC4 → bronchoconstriction
- Reactive oxygen species (ROS) → oxidative stress
- Charcot-Leyden crystals (crystallized galectin-10/GAL10 from eosinophils) → strong inducers of inflammation and mucus production
- This phase underlies persistent AHR and sets the stage for remodeling
Key Cellular Players
| Cell | Role in Asthma |
|---|---|
| Mast cells | Early-phase trigger; preformed + newly synthesized mediators |
| Th2 / ILC2 cells | Master regulators of T2 inflammation; IL-4, IL-5, IL-13 |
| Eosinophils | Late-phase tissue damage; MBP, ECP, LTC4, ROS |
| Basophils | IgE-dependent histamine + IL-4/IL-13 release; Th2 polarization |
| Dendritic cells | Antigen presentation; Th2 polarization |
| Macrophages | Source of ROS; amplify inflammation |
| Neutrophils | Recruited by LTB4; prominent in severe/steroid-resistant asthma |
| Airway epithelial cells | Release alarmins (TSLP, IL-25, IL-33) after injury |
Innate Immune Contribution: Alarmins & ILC2s
Damaged epithelial cells release epithelial-derived alarmins:
- TSLP (thymic stromal lymphopoietin)
- IL-33
- IL-25
These activate type 2 innate lymphoid cells (ILC2s), which produce IL-4, IL-5, and IL-13 without antigen recognition. This explains non-allergic asthma and explains why inflammation can occur even without adaptive immune sensitization.
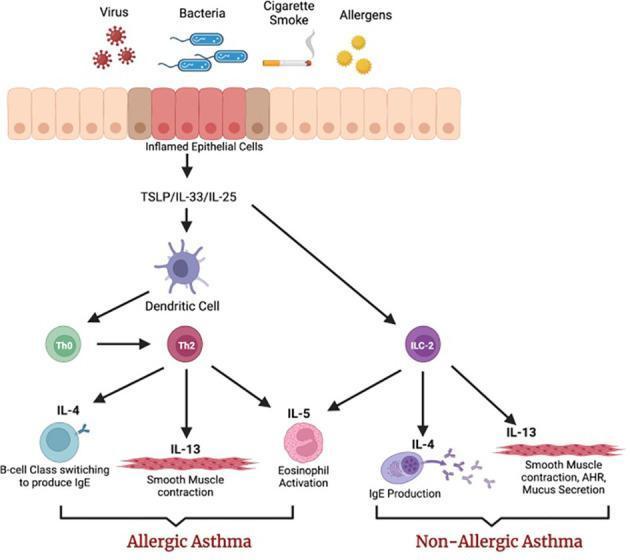
Airway Hyperresponsiveness (AHR)
AHR is defined as an exaggerated bronchoconstrictor response to stimuli (cold air, exercise, methacholine, histamine) that would be innocuous in normal individuals.
Mechanisms include:
- Subepithelial exposure of sensory nerves (from epithelial damage) → exaggerated reflex bronchoconstriction
- ASM hypertrophy + hyperplasia → increased contractile mass
- Increased mucosal permeability (allows allergens/irritants deeper access)
- Neural plasticity: upregulation of substance P and other neuropeptides
- Persistent eosinophilic inflammation lowering the threshold of ASM contraction
Mediator Profile Summary
| Mediator | Source | Effect |
|---|---|---|
| Histamine | Mast cells, basophils | Bronchoconstriction, edema |
| LTC4/LTD4/LTE4 | Mast cells, eosinophils | Prolonged bronchoconstriction, mucus, edema |
| LTB4 | Neutrophils | Neutrophil chemotaxis |
| PGD2 | Mast cells | Bronchoconstriction, vasodilation |
| IL-5 | Th2, ILC2 | Eosinophil maturation/activation |
| IL-13 | Th2, ILC2 | Mucus hypersecretion, AHR |
| Acetylcholine | Parasympathetic nerves | ASM contraction via muscarinic receptors |
| MBP / ECP | Eosinophils | Epithelial necrosis, ciliostasis |
| GAL10 (Charcot-Leyden) | Eosinophils | Inflammation, mucus induction |
| TGF-β | Multiple cells | Subepithelial fibrosis (remodeling) |
Genetics & Environment
- Asthma is polygenic — multiple loci on chromosomes 5q, 6p, 11q, 12q, 14q, and 16p are implicated.
- Gene-environment interactions are critical: the same genetic predisposition can result in very different phenotypes depending on:
- Allergen load and type (house dust mite, pollens, cockroach, pet dander)
- Hygiene hypothesis: reduced microbial exposure in early life fails to skew immunity away from Th2, promoting atopy
- Viral URTIs (particularly rhinovirus) → major trigger of exacerbations via TLR3-mediated epithelial alarmin release
Airway Remodeling (Chronic Asthma)
With persistent disease, structural changes accrue and add an irreversible component to obstruction:
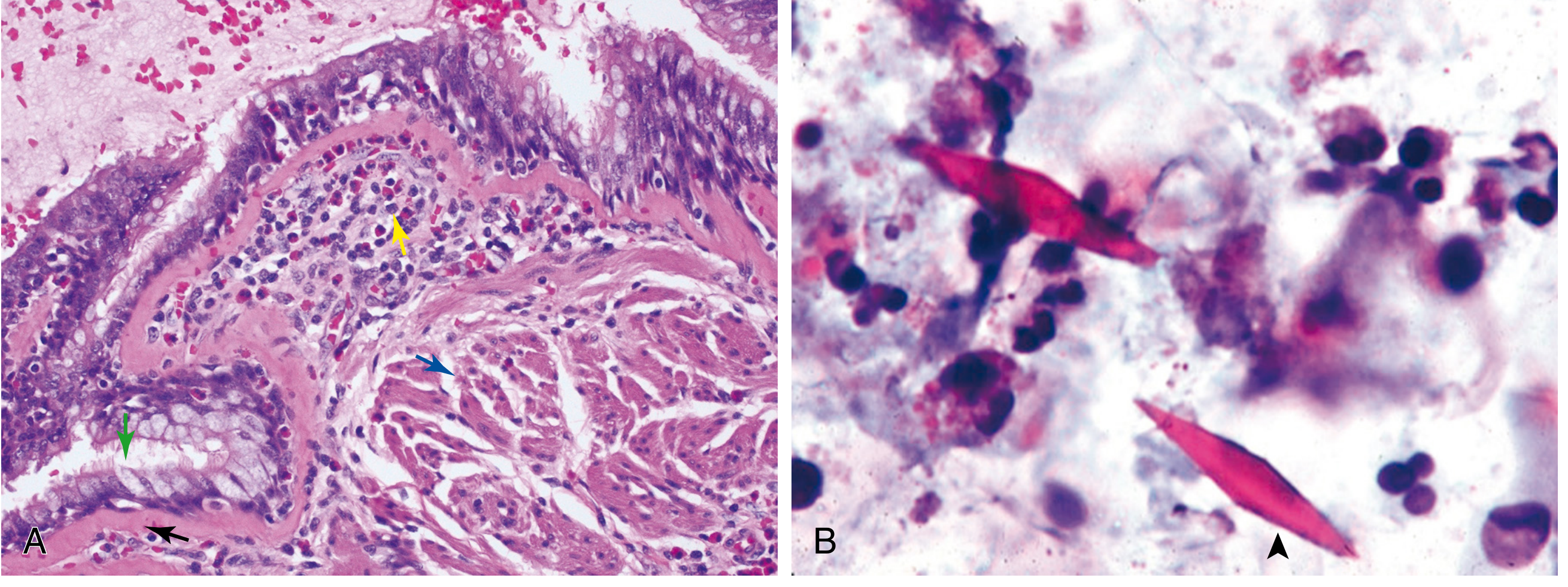
Histology (Robbins Fig. 15.11): (A) Asthmatic bronchus — goblet cell hyperplasia (green arrow), sub-basement membrane fibrosis (black arrow), eosinophilic inflammation (yellow arrow), muscle hypertrophy (blue arrow). (B) Sputum: Charcot-Leyden crystals.
| Structural Change | Mechanism | Consequence |
|---|---|---|
| Goblet cell hyperplasia | IL-13, IL-4 | Mucus plugging |
| Sub-basement membrane fibrosis | TGF-β → collagen deposition | Irreversible narrowing |
| ASM hypertrophy + hyperplasia | Growth factors, chronic contraction | ↑ AHR, fixed obstruction |
| Submucosal gland hypertrophy | Cholinergic + inflammatory stimuli | Mucus hypersecretion |
| Angiogenesis | VEGF from mast cells | Mucosal edema |
| Nerve remodeling | Substance P, VIP changes | Neural AHR |
Growth factors driving remodeling: TGF-β, EGF, FGF-2, VEGF, PDGF.
Aspirin-Exacerbated Respiratory Disease (AERD)
A distinct phenotype with:
- Chronic rhinosinusitis + nasal polyps + severe asthma
- Mechanism: Aspirin/NSAIDs inhibit COX → ↓ PGE2 → loss of tonic inhibition of 5-lipoxygenase → massive surge in cysteinyl leukotrienes (LTC4/D4/E4)
- Mast cells are the primary source of cysteinyl leukotrienes in this syndrome
- Diagnosed by elevated urinary LTE4
Non-T2 (Th1/Th17) Pathway
In ~20–35% of asthmatic patients (typically severe, steroid-resistant):
- Th17 cells → IL-17 → neutrophil recruitment → neutrophilic inflammation
- Th1 cells → IFN-γ → further epithelial damage
- Often associated with obesity, smoking, pollutant exposure
- Less responsive to inhaled corticosteroids
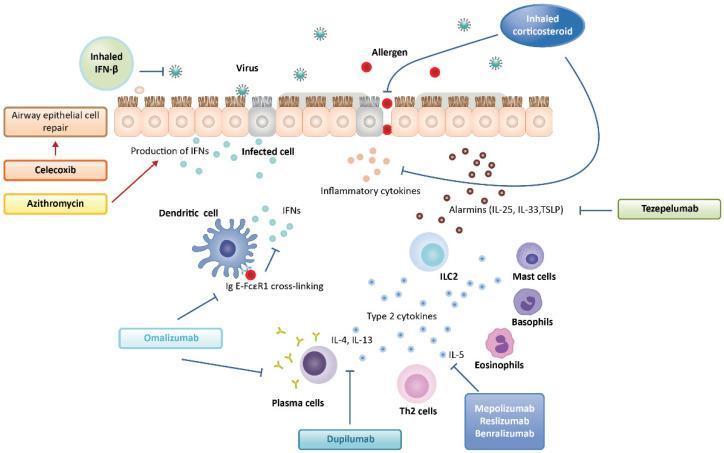
Physiologic Consequences
- FEV1/FVC ratio < 0.70 (obstructive pattern); reversible with bronchodilator (≥12% + 200 mL improvement)
- Air trapping → ↑ RV, ↑ FRC
- Ventilation-perfusion mismatch → hypoxemia
- Hypocapnia (early, due to hyperventilation); hypercapnia in severe/status asthmaticus (fatigue, impending respiratory failure)
- Pulsus paradoxus > 10 mmHg in severe attacks
- Mucus plugging → atelectasis
Summary Diagram Logic
Allergen → Dendritic cell → Th2 polarization → IgE production → Mast cell sensitization
↓ (Re-exposure)
IgE cross-linking → Mast cell degranulation
↓
Early phase: Bronchoconstriction + edema + mucus (0–30 min)
↓
Late phase: Eosinophil/neutrophil infiltration + epithelial damage (2–8 h)
↓ (Repeated)
Chronic inflammation → Airway remodeling → Fixed obstruction
Key references: Robbins, Cotran & Kumar — Pathologic Basis of Disease, 10th ed., p. 639–641; Fishman's Pulmonary Diseases and Disorders, Ch. 43; Murray & Nadel's Textbook of Respiratory Medicine
Give me a clear flowchart for ppt
Reading File
Reading File
Reading File
Running Command
Running Command
Writing File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.