How to proceed with infertilty treatment in females
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching PubMed
female infertility treatment
I now have comprehensive information to provide a thorough, well-structured answer. Here is a complete clinical guide to female infertility treatment:
Female Infertility: Evaluation & Treatment
Definition: Inability to achieve pregnancy after 12 months of regular unprotected intercourse (or 6 months if the female partner is >35 years; immediate evaluation if >40 years). Affects ~17.5% of couples globally.
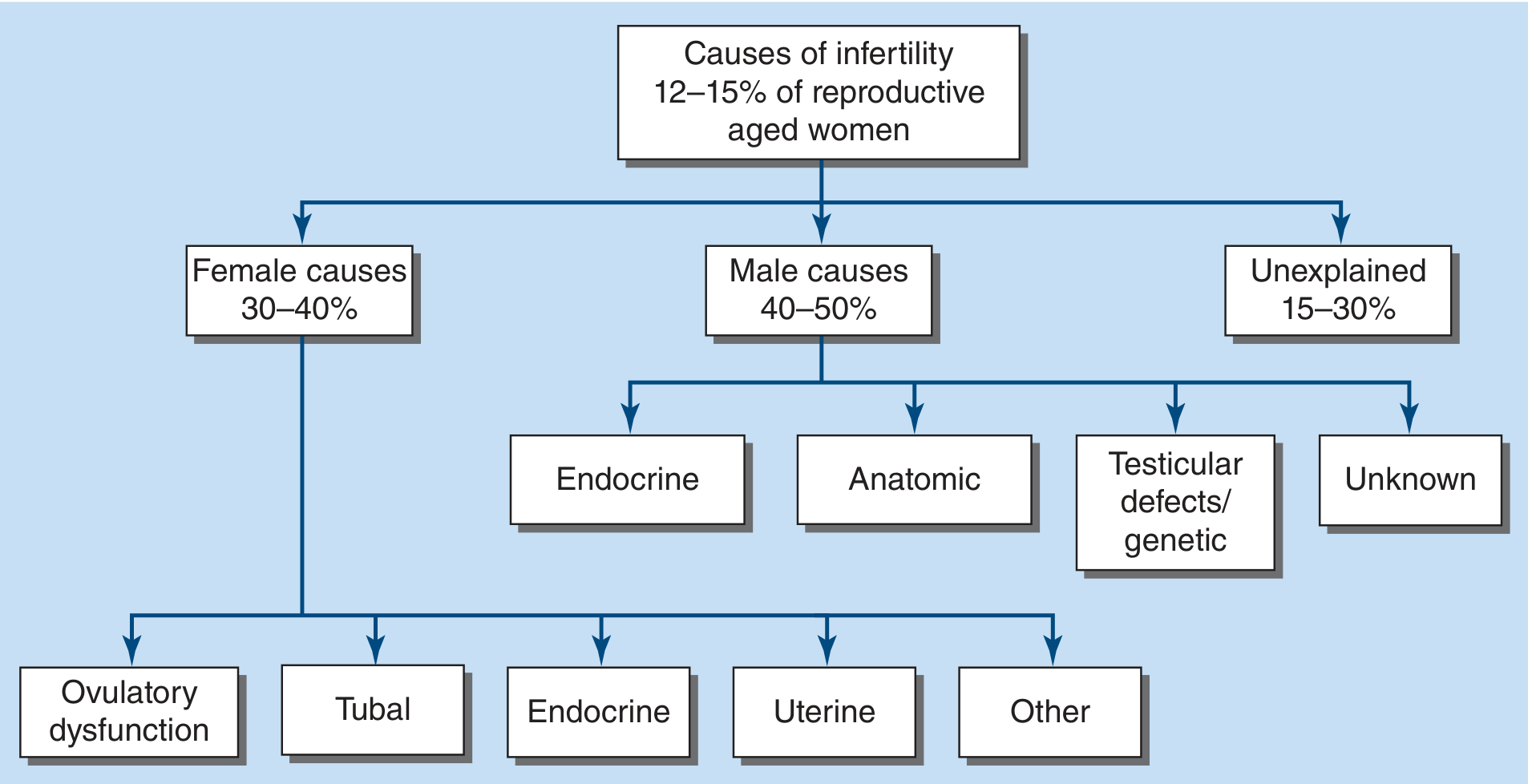
Step 1 — Complete Evaluation Before Starting Treatment
Perform a full workup of both partners before initiating any treatment, as multiple causes may coexist.
| Investigation | Purpose |
|---|---|
| Semen analysis | Exclude male factor (~40–50% of cases) |
| Confirmation of ovulation | Urinary LH kits, day 21 progesterone, basal body temperature |
| Ovarian reserve | Day 3 FSH, estradiol, Anti-Müllerian hormone (AMH), antral follicle count |
| Tubal patency | Hysterosalpingography (HSG) |
| Uterine cavity | Sonohysterography or hysteroscopy |
| Hormonal screen | TSH, prolactin, androgens (PCOS workup) |
Female causes account for 30–40% of infertility: ovulatory dysfunction, tubal/peritoneal factors, uterine pathology, and endocrine disorders.
Step 2 — Preconception Counseling (All Patients)
Before any pharmacologic intervention:
- Weight management: Obesity increases anovulatory cycles, miscarriage rates, and maternal complications. Weight loss in obese women improves spontaneous and drug-induced ovulation.
- Smoking cessation: Smoking damages oocyte DNA, reduces ovarian reserve (hastens menopause by 1–4 years), and raises ectopic pregnancy risk.
- Caffeine restriction: Limit to ≤2 cups/day during attempts and pregnancy.
- Timing intercourse: Fertile window is 5–6 days before ovulation; every 1–2 days during this window. LH kits can help time intercourse.
- Folic acid supplementation: 400 µg/day preconceptionally.
- Aneuploidy counseling: Advanced maternal age (>35) is associated with higher chromosomal error rates in oocytes.
Step 3 — Treatment by Etiology
A. Ovulatory Dysfunction (30–40% of female infertility; most treatable)
Most common cause: PCOS
- First-line: Letrozole (aromatase inhibitor) — preferred over clomiphene for PCOS (higher live birth rates)
- First-line alternative: Clomiphene citrate (selective estrogen receptor modulator)
- 60–80% of women with PCOS respond to these oral agents
- Second-line: Metformin — added to oral agents in obese women with PCOS to further increase ovulation rates
- Hypothyroidism / Hyperprolactinemia: Treat the underlying endocrine disorder before using ovulation induction agents
- Hypothalamic amenorrhea: Weight gain + reduced exercise first; if no response → low-dose injectable gonadotropins (FSH ± LH) for monofollicular stimulation
- Injectable gonadotropins (FSH/LH): Used when oral agents fail; require close monitoring (ultrasound + hormone levels) to prevent ovarian hyperstimulation syndrome (OHSS)
B. Tubal Factor Infertility (30–35% of female infertility)
- Hydrosalpinx / tubal occlusion from STI/PID: IVF is preferred — bypasses tubes entirely, offers highest success rates. Tubal repair is generally not recommended due to poor outcomes and high ectopic pregnancy risk.
- Salpingectomy for hydrosalpinges before IVF improves pregnancy rates by preventing efflux of toxic tubal fluid into the uterine cavity.
- Proximal tubal blockage on HSG: Attempt radiographically-guided tubal cannulation first.
- Post-tubal ligation: Decision between microsurgical reanastomosis vs. IVF depends on age, ovarian reserve, number of children desired, partner's sperm parameters, surgeon experience, and cost.
C. Uterine Factors (up to 15% of infertile couples)
| Pathology | Management |
|---|---|
| Submucosal fibroids | Hysteroscopic myomectomy |
| Endometrial polyps | Hysteroscopic polypectomy |
| Intrauterine adhesions (Asherman's) | Hysteroscopic adhesiolysis + estrogen support |
| Congenital anomalies (septum) | Hysteroscopic septum resection |
D. Endometriosis
- Mild endometriosis (no tubal/peritoneal adhesions): Superovulation with IUI may be tried
- Moderate-severe (tubal adhesions, endometrioma): IVF is the treatment of choice
- Surgical resection of endometriomas before IVF: May be considered but can reduce ovarian reserve — weigh carefully
E. Diminished Ovarian Reserve / Premature Ovarian Insufficiency
- Step up from oral ovulation induction + IUI → IVF
- Overall live birth rates are lower
- Donor oocytes: A highly effective option — donor undergoes oocyte retrieval, eggs are fertilized with partner's sperm, and embryos are transferred to the patient's uterus
Step 4 — Assisted Reproductive Technologies (ART)
Intrauterine Insemination (IUI)
- Washed, concentrated sperm deposited into the uterine cavity via a soft catheter
- Used for: unexplained infertility, mild male factor, mild ovulatory dysfunction, endometriosis (mild)
- Often combined with ovulation induction
- Pregnancy rate: ~8–16% per cycle
In Vitro Fertilization (IVF)
- Indications: Tubal factor, moderate-severe endometriosis, diminished ovarian reserve, failed IUI, severe male factor
- Process:
- Ovarian stimulation with gonadotropins (FSH ± LH)
- GnRH antagonist to prevent premature LH surge
- Transvaginal oocyte retrieval under sedation
- Fertilization in laboratory (conventional IVF or ICSI)
- Embryo culture: 3 days (cleavage stage) or 5 days (blastocyst)
- Transcervical embryo transfer under ultrasound guidance
- Luteal phase progesterone support
- Cryopreservation of surplus embryos
ICSI (Intracytoplasmic Sperm Injection)
- A single morphologically normal sperm is injected directly into the oocyte
- Used for severe male factor infertility (sperm count <5 million/mL or motility <20%)
- Achieves ART success rates comparable to non-male factor couples
Preimplantation Genetic Testing (PGT)
- PGT-A (aneuploidy screening): Reduces miscarriage risk, especially in advanced maternal age
- PGT-M (monogenic disorders): Used when known genetic disease risk exists
Step 5 — Special Situations
Unexplained Infertility
- After complete negative workup (~30% of cases)
- Initial: Superovulation (letrozole/clomiphene) + IUI
- Escalate to IVF if no success after 3–6 cycles of IUI
Fertility Preservation (Cancer Patients)
- Oocyte cryopreservation or embryo cryopreservation before chemotherapy/radiation
- Chemotherapy can hasten ovarian failure — referral to reproductive specialist should not be delayed
Key Complications to Monitor
| Complication | Notes |
|---|---|
| Ovarian hyperstimulation syndrome (OHSS) | Risk with gonadotropins; minimize with careful dose titration; GnRH antagonist protocols lower risk |
| Multiple gestation | Most serious complication of ART; single embryo transfer (SET) is strongly preferred |
| Ectopic pregnancy | Increased risk with tubal factor infertility; vigilant early monitoring required |
Summary Algorithm
Unexplained or Ovulatory → Letrozole/Clomiphene + IUI (3–6 cycles)
↓ (failure)
IVF
Tubal factor / Hydrosalpinx → IVF ± Salpingectomy first
Uterine pathology → Hysteroscopic correction, then reassess
Endocrine cause → Treat underlying cause → then Ovulation Induction
Diminished Reserve / POI → IVF ± Donor Oocytes
Severe male factor → IVF + ICSI
Sources:
- Harrison's Principles of Internal Medicine, 22nd Ed., Ch. 408
- Berek & Novak's Gynecology, Ch. on Infertility & ART
- Recent meta-analysis: Inositol for PCOS (PMID: 38163998, J Clin Endocrinol Metab 2024) supports inositol as an adjunct in PCOS management, consistent with current guidelines.
This is a shared conversation. Sign in to Orris to start your own chat.