How does hyperinsulinemia increases the risk of atherosclerosis
Reading File
Finding Sources
Finding Sources
Searching PubMed
hyperinsulinemia atherosclerosis mechanism
Searching the Web
hyperinsulinemia atherosclerosis mechanisms pathophysiology
Reading File
Reading File
Reading File
Reading Web Page
hyperinsulinemia atherosclerosis mechanisms
Reading File
Finding Sources
Loading Image
I now have everything needed for a comprehensive, well-cited answer. Here it is:
How Hyperinsulinemia Increases the Risk of Atherosclerosis
Hyperinsulinemia — chronically elevated circulating insulin, typically arising as a compensatory response to peripheral insulin resistance — promotes atherosclerosis through at least six interconnected mechanisms.
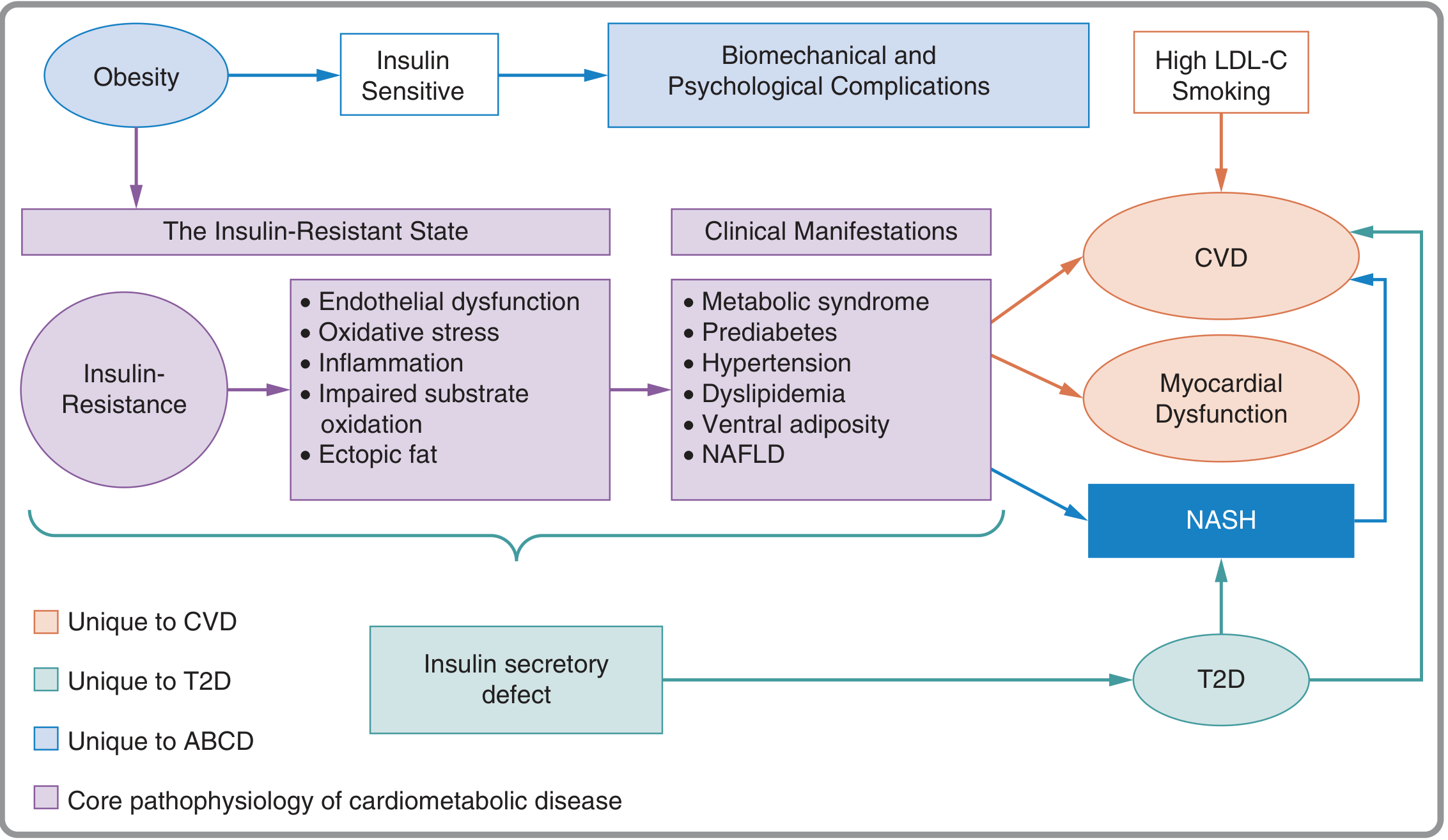
1. Atherogenic Dyslipidemia
Insulin normally suppresses hepatic VLDL secretion and stimulates lipoprotein lipase activity. In insulin resistance, this suppression fails, while the liver remains sensitive to insulin's lipogenic signalling (a phenomenon of selective insulin resistance).
- Excess free fatty acids (NEFAs) from unrestrained adipose lipolysis flood the liver, stabilizing Apolipoprotein B-100 (ApoB-100) — the structural core of VLDL — thereby increasing VLDL-C secretion.
- High insulin simultaneously stimulates SREBP-1c, activating glucokinase and acetyl-CoA carboxylase-1 (ACC-1), driving de novo lipogenesis and further VLDL overproduction.
- The resulting hypertriglyceridemia, combined with cholesteryl ester transfer protein (CETP) activity, produces small, dense LDL particles (highly susceptible to oxidation and subendothelial retention) and low HDL-C.
- This triad — elevated TG, low HDL, and small dense LDL — is the classic atherogenic dyslipidemia of the insulin-resistant state. (Fuster and Hurst's The Heart, 15th Ed.; PMC review, 2025)
2. Endothelial Dysfunction
The vascular endothelium is a key early site where hyperinsulinemia exerts damage:
- Insulin normally activates the PI3-kinase/Akt pathway → endothelial nitric oxide synthase (eNOS) → nitric oxide (NO) production → vasodilation and antiatherogenic effects.
- In insulin resistance, this pathway is selectively impaired, reducing NO bioavailability and impairing endothelium-dependent vasodilation.
- Simultaneously, the MAPK/ERK pathway (which is not desensitized in insulin resistance) remains active. High insulin through this pathway stimulates endothelin-1 (ET-1) secretion — a potent vasoconstrictor and pro-inflammatory mediator.
- The net result: vasoconstriction, increased leukocyte adhesion, and reduced fibrinolysis — all early steps in atherogenesis. (Fuster and Hurst's The Heart, 15th Ed., p. 262)
3. Vascular Smooth Muscle Cell (VSMC) Proliferation and Collagen Synthesis
Insulin is a growth factor. Via IGF-1 receptors and the insulin receptor on VSMCs:
- High insulin promotes VSMC proliferation and migration into the intima.
- It stimulates collagen synthesis, contributing to fibrous plaque formation and arterial wall thickening.
- It activates vascular collagen cross-linking, increasing arterial stiffness.
These effects are mediated principally through the MAPK pathway, which — unlike the metabolic PI3K pathway — retains full sensitivity even when insulin resistance is present. (PMC review, PMID 39415589)
4. Systemic Inflammation and Oxidative Stress
The insulin-resistant adipose tissue is a major driver of a chronic pro-inflammatory state:
- Visceral adipose tissue contains abundant M1 macrophages that secrete TNF-α, IL-6, and IL-1β.
- Dysfunctional adipocytes release elevated free fatty acids, leptin, resistin, and angiotensinogen, while adiponectin (anti-inflammatory/anti-atherogenic) is suppressed.
- Elevated lipids and glucose cause lipotoxicity and glucotoxicity, augmenting reactive oxygen species (ROS) production.
- ROS activates NF-κB, amplifying cytokine production and creating a feed-forward inflammatory loop.
- Intracellular diacylglycerols and ceramides activate serine/threonine kinases that further impair insulin signalling.
- Elevated CRP, fibrinogen, and IL-6 are measurable correlates of this state and independent cardiovascular risk markers. (Fuster and Hurst's The Heart, 15th Ed., pp. 262–263)
5. Prothrombotic and Impaired Fibrinolytic State
Hyperinsulinemia shifts haemostasis toward coagulation:
- High insulin increases plasminogen activator inhibitor-1 (PAI-1) production from the liver and endothelium → impaired fibrinolysis → increased thrombus formation on vulnerable plaques.
- Elevated serum fibrinogen concentrations (driven by IL-6 from adipose tissue) further increase clot burden.
- This procoagulant phenotype dramatically amplifies the clinical consequences of even modest plaque rupture. (Current Surgical Therapy 14e, p. 245)
6. Hypertension (Increased Mechanical Stress on Vessel Wall)
Hyperinsulinemia contributes to hypertension — an independent mechanical driver of atherosclerosis — through multiple mechanisms:
- Reduced NO → impaired vasodilation
- Elevated ET-1 → vasoconstriction
- Renal sodium retention via stimulation of Na-K-ATPase and NHE3 in the proximal tubule
- Sympathetic nervous system activation
- Renin-angiotensin-aldosterone system (RAAS) stimulation and increased aldosterone production
- Increased renal afferent arteriolar vasodilation → hyperfiltration and microvascular remodelling. (PMC review, 2025)
Summary Table
| Mechanism | Key Mediators | Atherogenic Effect |
|---|---|---|
| Atherogenic dyslipidemia | ↑VLDL, ↑TG, ↓HDL, small dense LDL | LDL oxidation, foam cell formation |
| Endothelial dysfunction | ↓NO, ↑ET-1 | Impaired vasodilation, leukocyte adhesion |
| VSMC proliferation | MAPK/ERK, IGF-1R | Intimal thickening, plaque growth |
| Inflammation & oxidative stress | TNF-α, IL-6, ROS, NF-κB | Plaque initiation and progression |
| Prothrombotic state | ↑PAI-1, ↑fibrinogen | Thrombosis on ruptured plaques |
| Hypertension | ↓NO, ↑RAAS, Na+ retention | Mechanical arterial wall injury |
Clinical Evidence
The Insulin Resistance Atherosclerosis Study and the Bezafibrate Infarction Prevention Trial demonstrated that insulin resistance was independently associated with incident CVD events and increased carotid intima-media thickness. A meta-analysis of 65 studies by Gast et al. confirmed that the HOMA index of insulin sensitivity significantly predicted relative risk for CVD events. (Fuster and Hurst's The Heart, 15th Ed., p. 261)
A 2025 meta-analysis (PMID 41180178) of cohort studies confirmed that metabolic insulin resistance scores are independently associated with incident cardiovascular disease across populations.
Key takeaway: Hyperinsulinemia does not cause atherosclerosis through a single pathway. It acts simultaneously on lipid metabolism, endothelial function, vascular smooth muscle, coagulation, inflammation, and blood pressure regulation — each of which independently accelerates plaque formation. The convergence of all these pathways explains why cardiometabolic disease driven by the insulin-resistant state accounts for the majority of "residual" cardiovascular risk beyond LDL-C and smoking.
This is a shared conversation. Sign in to Orris to start your own chat.