Children's infections, their features. Chickenpox and herpes zoster, cytomegaly.Etiology, epidemiology, patho- and morphogenesis, clinical and morphological characteristics, complications, outcomes.
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
varicella chickenpox vesicular rash skin lesions
Searching Images
cytomegalovirus owl eye inclusion cells histology
Reading File
I now have comprehensive content from the textbooks. Let me compile the full educational response.
Children's Infections: Chickenpox, Herpes Zoster & Cytomegalovirus
PART I — VARICELLA (CHICKENPOX) AND HERPES ZOSTER
Shared Etiology
Both varicella (chickenpox) and herpes zoster (shingles) are caused by varicella-zoster virus (VZV) — a member of the herpesvirus family (Herpesviridae, alpha-herpesvirus subfamily). The two diseases are not caused by different viruses but differ because of the host immune state and disease stage (primary vs. reactivation). The viral genome is double-stranded DNA; virions have the characteristic herpesvirus envelope structure.
I.A. VARICELLA (Chickenpox)
Epidemiology
- Highly contagious; distributed worldwide
- In temperate climates without vaccination, varicella is endemic with seasonal peaks in winter and spring, with periodic epidemics
- In Europe and North America, >90% of people acquire varicella before age 15 in the absence of vaccination
- Transmission occurs via:
- Respiratory droplets/aerosols (most important — airborne transmission)
- Direct contact with vesicular fluid
- Incubation period: 10–21 days (typically 14–16 days)
- Infectious from 1–2 days before the rash appears until all lesions have crusted over
- Secondary household attack rate among susceptible contacts: ~60–90%
- Outbreaks common in schools and daycare centers
- Live-attenuated Oka VZV vaccine has virtually eliminated varicella in countries where deployed
Pathogenesis & Morphogenesis
- Primary inoculation: VZV enters via the upper respiratory tract mucosa
- First viremia: Virus replicates in regional lymph nodes → seeds the reticuloendothelial system (liver, spleen)
- Second viremia (~day 11–13): Major viremic phase disseminates virus hematogenously to skin and mucous membranes, producing the exanthem
- The scattered distribution of lesions reflects viremic seeding (not neural spread)
- At the skin: VZV infects epidermal cells → intranuclear inclusions (Cowdry type A), multinucleated giant cells, intraepidermal vesicle formation by ballooning degeneration of keratinocytes; vesicles contain serous fluid with inflammatory cells
- Latency establishment: After resolution of primary infection, VZV travels retrogradely up sensory axons and establishes latent infection in dorsal root ganglia and cranial nerve ganglia (specifically within neurons, not satellite cells), where it persists for life
Clinical Features
Prodrome (uncommon in young children; present in adults):
- 2–3 days of mild fever, chills, malaise, headache, anorexia, backache, dry cough
Exanthem:
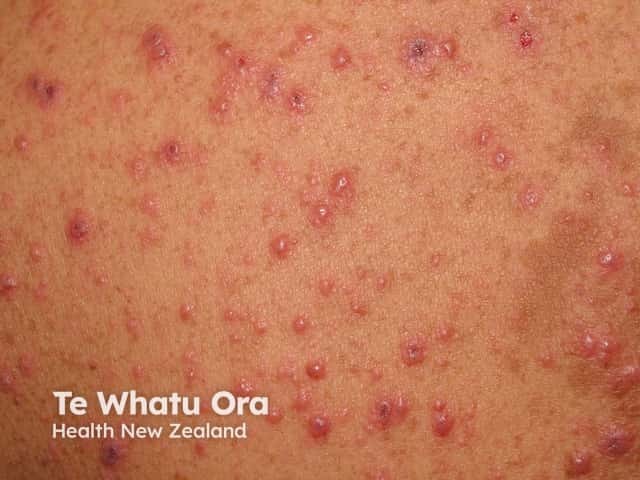
- Begins on face/scalp → rapid centrifugal spread to trunk (denser on back between shoulder blades) → relative sparing of extremities
- Lesions evolve rapidly (within 12 hours): macule → papule → vesicle → pustule → crust
- Classic description: vesicle appears as a "dew drop on a rose petal" — thin-walled, 2–3 mm, elliptical, clear vesicle on an erythematous base
- Hallmark: Lesions in all stages simultaneously present in any given area (due to crops)
- Vesicles become umbilicated pustules then crust; crusts fall in 1–3 weeks
- Oral/mucosal lesions (enanthem) common — shallow painful ulcers on the palate, buccal mucosa
Complications
| Complication | Notes |
|---|---|
| Secondary bacterial superinfection (most common) | S. aureus, S. pyogenes; impetigo, cellulitis, necrotizing fasciitis |
| Varicella pneumonia | Serious in adults; interstitial pneumonitis; also in immunocompromised children |
| CNS complications | Acute cerebellar ataxia (most common CNS complication in children; usually benign), encephalitis (rare but serious), Reye syndrome (associated with aspirin use) |
| Hemorrhagic varicella | Rare; thrombocytopenia + hemorrhagic vesicles; more in immunocompromised |
| Visceral dissemination | Hepatitis, myocarditis — in immunocompromised |
| Congenital varicella syndrome | Maternal primary infection in first 20 weeks → limb hypoplasia, cicatricial skin scarring, neurological defects, eye abnormalities |
| Neonatal varicella | If mother develops varicella 5 days before to 2 days after delivery: severe, potentially fatal (no passive immunity transmitted) |
Outcomes
- In immunocompetent children: self-limited; full recovery in 1–2 weeks
- In adults, immunocompromised, neonates, pregnant women: higher morbidity and mortality
- Lifelong immunity follows natural infection (but latent VZV persists in ganglia)
I.B. HERPES ZOSTER (Shingles)
Etiology & Pathogenesis (Reactivation)
- Herpes zoster results from reactivation of latent VZV in sensory/autonomic ganglia following a prior episode of varicella (which may have been subclinical)
- Trigger: decline in VZV-specific cell-mediated immunity — occurring with aging, immunosuppression (HIV, malignancy, corticosteroids, organ transplant), psychological stress
- Reactivated VZV travels antegradely down sensory nerve axons to the skin of the corresponding dermatome → circumscribed dermatomal eruption
- Lesion distribution is neural (not hematogenous) → explains clustering in a single dermatome (vs. scattered lesions of varicella)
- Most common ganglia involved: thoracic (50–60%), trigeminal, cervical, lumbosacral
Epidemiology
- Most common in older adults (incidence rises sharply after age 50) and immunocompromised individuals
- Incidence of PHN: 13% in ages 60–64, rising to 21% in ages 80–84
- A meta-analysis of 18 cohort studies found PHN incidence increased by 1.22–3.11% per 10-year age increase
- Less contagious than varicella; ~1/3 the secondary attack rate; requires direct contact with open vesicles (or airborne in disseminated zoster)
- Susceptible contacts exposed to herpes zoster can develop primary varicella (not zoster)
Clinical Features
Prodrome (days before rash):
- Localized burning, aching, stabbing pain in the affected dermatome; may mimic pleurisy, cardiac disease, appendicitis before rash appears
- Hyperesthesia, allodynia, dysesthesia, pruritus
Eruption:
- Unilateral, dermatomal cluster of erythematous macules and papules → vesicles (closely grouped, on confluent erythematous base — contrast with varicella's scattered lesions)
- Vesicles: 3–5 days, then pustulate and crust over in 7–10 days; full resolution in 2–4 weeks
- Does not cross midline in immunocompetent patients
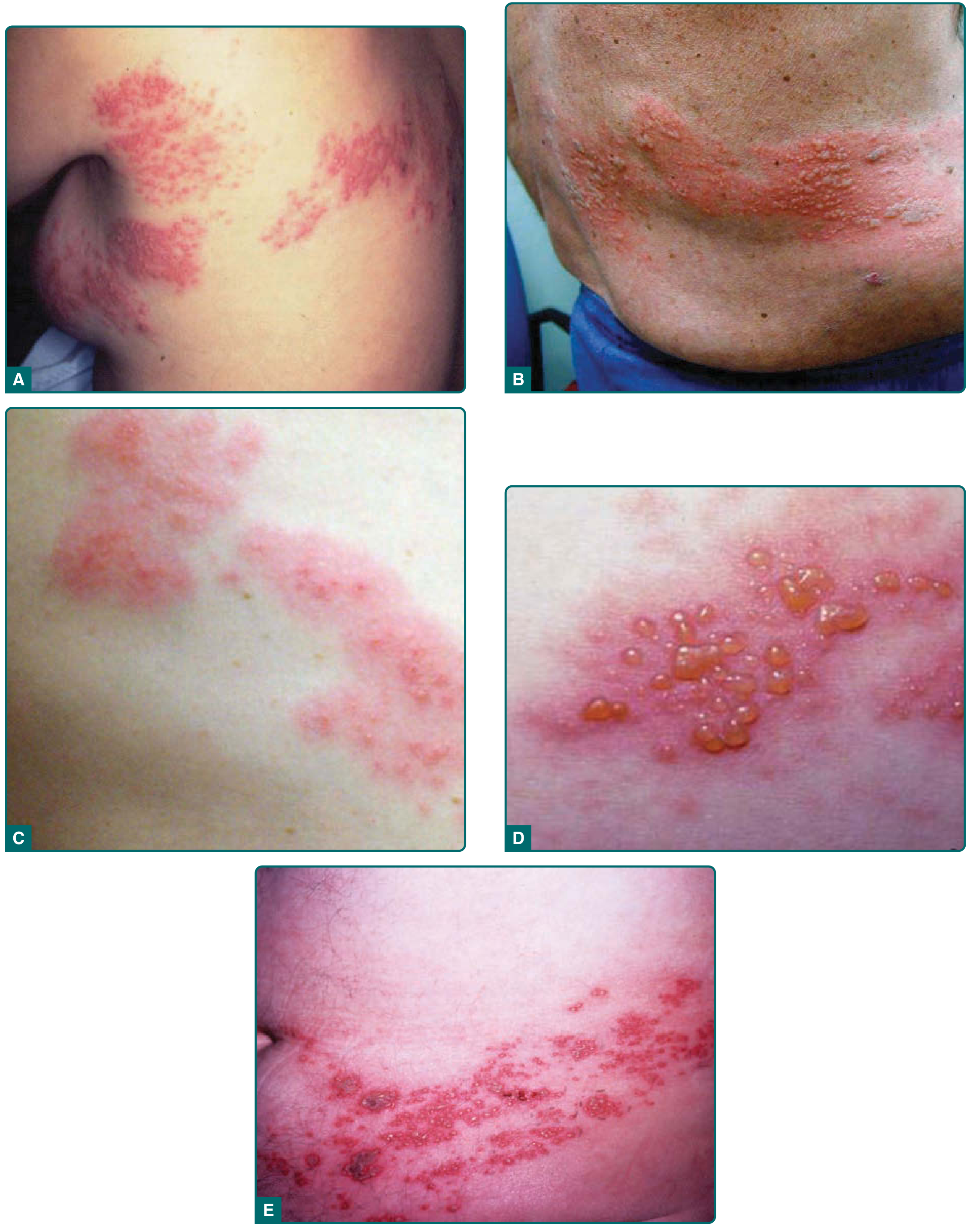
Special patterns:
- Ophthalmic zoster (V1 branch of CN V): Risk of keratitis, uveitis, corneal scarring; Hutchinson sign (vesicles on tip/side of nose = nasociliary branch involvement) predicts eye involvement
- Ramsay Hunt syndrome: VZV reactivation in geniculate ganglion of CN VII → painful auricular vesicles + ipsilateral facial palsy + sensorineural hearing loss; ± vertigo
- Disseminated zoster: >20 vesicles outside primary/adjacent dermatomes; occurs in immunocompromised; risk of visceral involvement (pneumonia, hepatitis, encephalitis)
Complications
| Complication | Features |
|---|---|
| Postherpetic neuralgia (PHN) | Most common debilitating complication; chronic neuropathic pain >90 days after rash; burning/aching/lancinating; allodynia in ~90% of PHN patients |
| Ophthalmic complications | Keratitis, uveitis, corneal scarring, vision loss |
| Ramsay Hunt syndrome | Facial palsy, hearing loss |
| Stroke/granulomatous angiitis | VZV-infected neurons invade cerebral vessels → segmental granulomatous angiitis → contralateral hemiplegia (average 7–8 weeks after ophthalmic zoster) |
| Visceral zoster | Esophagitis, hemicystitis, pleuritis, peritonitis, ileus — via visceral afferent neurons corresponding to the infected dermatome |
| Myelitis | Rare; ascending or transverse myelitis |
| Motor zoster | Myositis; rare LMN weakness in affected segment |
Morphology of VZV Lesions (Histology)
- Intraepidermal vesicles by ballooning degeneration and reticular degeneration of keratinocytes
- Cowdry type A intranuclear inclusions (eosinophilic inclusions surrounded by a halo) in epidermal cells and neurons
- Multinucleated giant cells (syncytia) — characteristic of herpesviruses
- In dorsal root ganglia: lymphocytic infiltration, neuronal necrosis, hemorrhage
PART II — CYTOMEGALOVIRUS (CMV) / CYTOMEGALY
Etiology
- Human CMV (Human Herpesvirus 5, HHV-5) — member of Herpesviridae, beta-herpesvirus subfamily (Betaherpesvirinae), genus Cytomegalovirus
- Largest human herpesvirus genome: double-stranded DNA, 196,000–240,000 bp, encoding ≥166 proteins
- Characterized by slow replication cycle ("cytomegalovirus" = cell-enlarging virus)
- Highly species-specific: only human CMV infects humans
Epidemiology
- Ubiquitous; widespread genetic diversity among strains
- No seasonal predilection
- Three peaks of acquisition:
- Early childhood (especially daycare/childcare settings)
- Adolescence
- Child-bearing years
Routes of Transmission:
- Horizontal: Direct person-to-person contact with virus-containing body fluids — saliva, urine, genital secretions, tears; sexual transmission (CMV is an STI)
- Vertical:
- Transplacental (in utero) — most important for congenital disease
- Intrapartum (passage through infected cervix)
- Postnatal (via breast milk — usually benign in term infants, but can cause sepsis-like syndrome in preterm infants)
- Iatrogenic: Blood transfusions, organ/stem cell transplantation
- Childcare centers: Excretion rates from urine/saliva in children age 1–3 years = 30–70%; children shed large quantities for prolonged periods and frequently transmit to parents and caregivers
Congenital CMV — epidemiology:
- ~5 per 1,000 live births infected in utero = most common congenital viral infection in the United States
- Higher in Black newborns (9.5/1,000) than non-Hispanic white (2.7/1,000) or Hispanic white infants (3.0/1,000)
-
75% of cases in U.S. born to women with nonprimary (reactivation/reinfection) infection
- First-trimester maternal primary infection → most severe fetal sequelae
Latency: CMV persists in leukocytes and tissue cells; intermittent shedding throughout life; reactivates with immunosuppression
Pathogenesis & Morphogenesis
Pathogenesis:
- Primary infection via mucosal surfaces → local replication → viremia (cell-associated, within monocytes/lymphocytes)
- Viral dissemination to multiple organs: lungs, liver, CNS, retina, GI tract, kidneys
- Establishment of latency in monocytes, CD34+ progenitor cells, lymphoid tissue
- Lifelong persistence; reactivation triggered by immunosuppression or inflammation
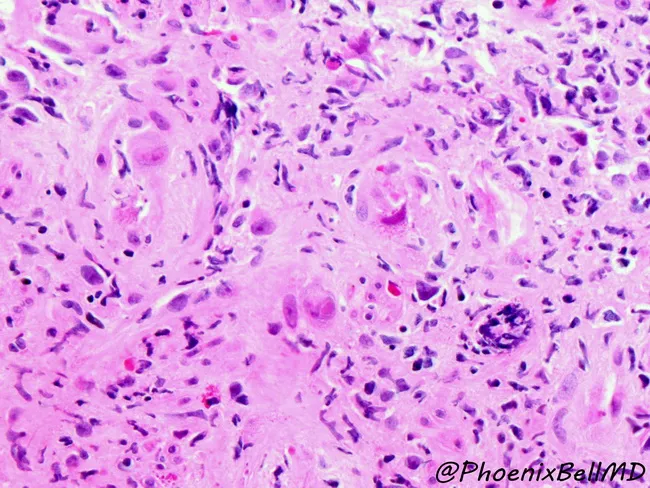
The cytomegalic cell (hallmark of CMV morphology):
- Infected cells are dramatically enlarged (cytomegaly) — up to 25–35 µm in diameter
- Intranuclear inclusion: Large, round, eosinophilic inclusion surrounded by a clear halo ("owl's eye" or "cytomegalic inclusion") with marginated chromatin
- Intracytoplasmic inclusions: Multiple basophilic/amphophilic granular inclusions (PAS- and GMS-positive, representing viral aggregates in cytoplasm)
- Cells affected: epithelial cells, endothelial cells, stromal cells, macrophages, neurons
Clinical Manifestations
1. Acquired CMV Infection (Postnatal, Immunocompetent)
- Majority: Asymptomatic — especially in children
- CMV mononucleosis (adolescents/adults): Infectious mononucleosis-like syndrome — prolonged fever, mild hepatitis, lymphocytosis; heterophile antibody (monospot) NEGATIVE — this distinguishes it from EBV mononucleosis
- Pharyngitis and lymphadenopathy are less prominent than EBV mononucleosis
2. Congenital CMV Infection
- ~90% asymptomatic at birth (but still at risk of late sequelae)
- ~10% symptomatic at birth (congenital CMV disease):
- Jaundice (direct hyperbilirubinemia)
- Petechiae (thrombocytopenia)
- Hepatosplenomegaly
- Microcephaly
- Periventricular calcifications (intracerebral; pathognomonic pattern — distinguishes from toxoplasmosis which is diffuse)
- Retinitis
- Growth restriction, developmental delays
- Mortality: 3–10% of symptomatic infants (0.3–1.0% of all congenital CMV)
Sensorineural Hearing Loss (SNHL) — most important long-term sequela:
- CMV is the leading non-genetic cause of SNHL in children
- Accounts for ~20% of hearing loss at birth and ~25% of all hearing loss at age 4
- Occurs in up to 50% of symptomatic congenital CMV and up to 15% of asymptomatic congenital CMV
- ~40% of CMV-associated SNHL is not detectable in the first month of life (late-onset)
- ~50% show progressive deterioration over time
3. Immunocompromised Hosts (HIV, Transplant, Malignancy)
- CMV retinitis: Most common serious manifestation in HIV/AIDS; "pizza pie" fundus appearance; can cause blindness
- CMV pneumonitis: Interstitial pattern; serious in hematopoietic stem cell transplant (HSCT) recipients
- CMV colitis: Bloody diarrhea, pain, ulceration
- CMV encephalitis/meningoencephalitis
- CMV syndrome: Fever, thrombocytopenia, leukopenia, mild hepatitis
4. Perinatal/Postnatal CMV in Preterm Infants
Acquired via breast milk from CMV-positive mothers → hepatitis, interstitial pneumonia, thrombocytopenia, leukopenia, viral sepsis-like syndrome
Complications & Outcomes
| Population | Major Complications | Outcomes |
|---|---|---|
| Immunocompetent children/adults | Mononucleosis; usually self-limited | Full recovery |
| Congenital (symptomatic) | SNHL, neurological impairment, microcephaly, vision loss | Significant morbidity; 3–10% mortality |
| Congenital (asymptomatic) | Late-onset SNHL (up to 15%); cognitive delay possible | Progressive hearing loss in many |
| HIV/Immunocompromised | Retinitis (blindness), pneumonia, colitis, encephalitis | Life-threatening without treatment |
| HSCT/Solid organ transplant | Pneumonitis, GI disease, graft-versus-host exacerbation | High mortality without prophylaxis/treatment |
| Preterm neonates | Sepsis-like syndrome, hepatitis, pneumonia | Can be severe |
Morphological Summary of Organ Pathology
| Organ | CMV Morphological Finding |
|---|---|
| Lung | Interstitial pneumonitis; cytomegalic cells in alveolar epithelium and macrophages |
| Liver | Hepatocyte and bile duct cell enlargement with inclusions; periportal inflammation |
| Brain | Periventricular necrosis + calcification (congenital); encephalitis; microglial nodules |
| Retina | Full-thickness retinal necrosis with hemorrhage; "pizza pie" appearance |
| GI tract | Mucosal ulceration; endothelial and epithelial owl's-eye cells |
| Kidney | Tubular epithelial cell inclusions; CMV-uria common |
| Salivary glands | Classic site of latency; large cytomegalic ductal epithelial cells |
Comparative Summary Table
| Feature | Varicella | Herpes Zoster | CMV |
|---|---|---|---|
| Virus family | Herpesviridae (α) | Herpesviridae (α) | Herpesviridae (β) |
| Virus | VZV (HHV-3) | VZV (HHV-3) | CMV (HHV-5) |
| Primary/reactivation | Primary infection | Reactivation | Primary or reactivation |
| Transmission | Airborne/contact | Contact (rarely airborne) | Contact, sexual, vertical, transfusion |
| Peak age | Children (school age) | Elderly/immunocompromised | Childcare age; congenital |
| Hallmark rash | Centripetal, scattered, all-stage-simultaneous | Unilateral, dermatomal, clustered | No specific rash (petechiae in congenital) |
| Histology | Ballooning degeneration, multinucleated cells, Cowdry A inclusions, intraepidermal vesicles | Same + ganglionic necrosis | Cytomegalic cells with owl's-eye inclusions |
| Latency site | Sensory ganglia neurons | Sensory ganglia neurons | Monocytes/CD34+ progenitors |
| Key child complication | Cerebellar ataxia, secondary bacterial infection | PHN (adults), Ramsay Hunt | SNHL, microcephaly, developmental delay |
| Vaccine | Live attenuated (Oka) | Recombinant subunit (Shingrix) | None approved |
Sources:
- Fitzpatrick's Dermatology, Chapters on Varicella and Herpes Zoster
- Red Book 2021: Report of the Committee on Infectious Diseases (AAP), CMV chapter
- Harrison's Principles of Internal Medicine, 22nd ed., CMV chapter
This is a shared conversation. Sign in to Orris to start your own chat.