Local antibiotic delivery systems definition types application and disadvantages structures everything for to answer 10 marks question
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
I now have all the content needed. Let me compile the comprehensive 10-mark answer.
Local Antibiotic Delivery Systems
Definition
Local antibiotic delivery (LAD) systems are devices or materials that are placed directly at or near an infection site to release high concentrations of antibiotics locally, without generating significant systemic drug levels. The underlying rationale is that the infected necrotic focus of bone (e.g., osteomyelitis) is commonly surrounded by sclerotic, avascular tissue that is virtually unreachable by systemic antibiotics. LAD systems overcome this barrier by maintaining bactericidal concentrations at the site while minimising systemic toxicity. — Rockwood and Green's Fractures in Adults, 10th ed.
Why Local Delivery is Needed
- Avascular/sclerotic bone around infection resists systemic antibiotic penetration
- Biofilm-producing organisms require concentrations 100–1000× higher than systemic routes can safely achieve
- Dead space after surgical debridement provides a nidus for re-infection unless obliterated
- Systemic routes carry dose-dependent toxicity (nephrotoxicity with aminoglycosides, ototoxicity, etc.)
Types / Classification
1. Non-Biodegradable (Non-Absorbable) Systems
The gold standard carrier is polymethylmethacrylate (PMMA) bone cement, used for over 40 years.
Mechanism of release: Passive diffusion of antibiotic through pores, voids, and micro-cracks in the cement matrix. Release follows a biphasic pattern: high initial burst release followed by a prolonged, lower-level "tail" release.
Forms:
| Form | Description | Key Features |
|---|---|---|
| PMMA Antibiotic Beads | Strings of small cement spheres on wire/suture | Greater surface area → higher initial antibiotic concentration; bead pouch technique used between serial debridements |
| PMMA Block Spacer | Solid shaped spacer filling a bone defect | Used in Masquelet induced membrane technique; prepares defect for bone grafting; structurally fills dead space |
| Antibiotic-Coated IM Nails/Rods | Narrow-diameter interlocked rods coated with antibiotic cement | Used for long-bone infections; provides stability + local delivery simultaneously |
Commonly loaded antibiotics for PMMA: Gentamicin, tobramycin, vancomycin (note: vancomycin gives burst release only; rifampicin and tetracycline are incompatible with PMMA).
Advantages:
- Achieves very high local concentrations with minimal systemic levels
- Excellent structural/space-filling properties
- 40+ years of clinical experience
- Can be combined with implant stabilisation
Disadvantages:
- Must be surgically removed (if left in situ, provides a substratum for bacterial colonisation — "foreign body effect")
- Inefficient release kinetics for some drugs (vancomycin: burst only → subtherapeutic tail)
- Incompatible with certain antibiotics (tetracycline, rifampicin)
- Requires a second operative procedure for removal
- Increased cost and surgical morbidity
2. Biodegradable (Bioabsorbable) Systems
Because of PMMA's limitations, biodegradable systems are increasingly used. They are categorised into three groups:
A. Natural Polymers (Protein-Based)
| Material | Source | Key Properties |
|---|---|---|
| Collagen (most studied) | Connective tissue | Biocompatible; antibiotic released primarily by diffusion — rapid bolus release; not by degradation; limited use in osteomyelitis |
| Chitosan | Polysaccharide biopolymer | Innate antimicrobial activity (polycationic — broad spectrum); promising adjunct |
| Others | Thrombin, autologous blood clot, gelatin | Experimental/limited use |
B. Bone Graft Substitutes / Bioceramic Materials
| Material | Properties | Drawbacks |
|---|---|---|
| Calcium Sulfate | Natural bioceramic; biodegradable; dissolves and releases antibiotic; fills dead space | Persistent wound drainage; seroma formation; uncontrolled antibiotic release |
| Hydroxyapatite (HA) | Stimulates osteoid formation on surface; enhances bone remodelling; osteoconductive | Less controlled release kinetics |
| Bioactive Glass | Combines angiogenic + osteoconductive + antimicrobial properties; integrates into bone and soft tissue; no removal required | Relatively newer with limited long-term data |
C. Synthetic Polymers
| Material | Properties |
|---|---|
| PLA (polylactic acid) / PGA (polyglycolic acid) | First-generation; degradation releases acidic products → limits use to small volumes (e.g., implant coating) |
| PDLLA / PLGA | Amorphous copolymers; better release profiles; primarily bulk erosion |
| PTMC (Polytrimethylene carbonate) | Fully biocompatible; degrades without acidic by-products; yields constant antibiotic release over time — currently the preferred synthetic polymer |
Advantages of biodegradable systems overall:
- Obliterate dead space AND release antibiotic after degradation
- Leave no residual substratum for bacterial colonisation
- Can carry a wider range of antibiotics
- Require fewer subsequent surgeries (no removal needed)
- Osteoconductive materials simultaneously promote bone healing
Applications
| Clinical Setting | System Used |
|---|---|
| Open fractures (grades IIIB/IIIC) | PMMA antibiotic beads / bead pouch in wound; vancomycin powder topically |
| Chronic osteomyelitis | PMMA beads or spacer, biodegradable ceramics post-debridement |
| Infected nonunion | PMMA spacer (Masquelet technique), antibiotic-coated IM nails |
| Periprosthetic joint infection (two-stage revision) | Antibiotic-impregnated PMMA spacer between stages |
| Dead space management post-debridement | Calcium sulfate, bioactive glass, PMMA block |
Clinical data: Adding local antibiotics to systemic therapy reduces infection in grade IIIB/IIIC open fractures from ~31% to ~9%; and grade IIIA from ~14.4% to ~2.4%. — Rockwood and Green's Fractures in Adults, 10th ed.
Summary of Advantages vs. Disadvantages
| Non-Biodegradable (PMMA) | Biodegradable | |
|---|---|---|
| Advantages | High local concentrations; structural spacer; proven track record | No second surgery; wider antibiotic range; promotes bone healing; no colonisation risk |
| Disadvantages | Requires removal; foreign body risk; inefficient kinetics for some drugs; antibiotic incompatibilities | Less mechanical strength; variable/uncontrolled release (calcium sulfate); newer agents with limited long-term data |
Key Structural Principle
All LAD systems must satisfy three criteria:
- Biocompatibility — tolerated by host tissues without reaction
- Adequate release kinetics — maintain antibiotic concentrations above MIC (minimum inhibitory concentration) at the infection site throughout the therapeutic window
- Dead space management — physically fill the void left by debridement to prevent fluid accumulation and re-infection
Source: Rockwood and Green's Fractures in Adults, 10th ed. 2025 — Chapters 29 & 30 (Osteomyelitis, Infected Nonunion)
Bone brain axis definition types application and disadvantages structures everything for to answer 10 marks question with appropriate illustrative diagrams
Checking Date & Time
Finding Sources
Finding Sources
Searching PubMed
bone brain axis osteocalcin
Reading Article
Reading Article
Reading Article
Reading Article
Reading Article
Reading Article
Searching Images
bone brain axis osteocalcin bidirectional signaling diagram
Loading Image
Loading Image
Reading Article
Searching Images
osteocalcin GPR158 brain neurotransmitter serotonin dopamine signaling pathway
Loading Image
I now have all the material needed — three excellent diagrams and comprehensive 2024–2026 review data. Let me compile the full 10-mark answer.
The Bone–Brain Axis
1. Definition
The bone–brain axis refers to the bidirectional communication network between the skeletal system and the central nervous system (CNS). Bone, long regarded solely as a structural and mineral-storage organ, is now recognised as an endocrine organ that secretes hormones and cytokines influencing brain function, cognition, mood, and neurodegeneration. Reciprocally, the brain — through neuroendocrine and autonomic pathways — regulates bone remodelling and mineral homeostasis.
This crosstalk is mediated by:
- Bone-derived hormones/factors entering the bloodstream and crossing the blood–brain barrier (BBB)
- Hypothalamic neuropeptides, neurotransmitters, and sympathetic/parasympathetic signals acting on osteoblasts and osteoclasts
- Extracellular vesicles (EVs) carrying miRNAs and proteins between the two systems
"The bone and brain, though distinct in structure and function, share remarkable molecular and developmental similarities." — Hansda & Das, Biology 2025 [PMID 41007423]
2. Illustrative Diagrams
Diagram 1 — Bone–Organ Axes (including Bone–Brain Axis)
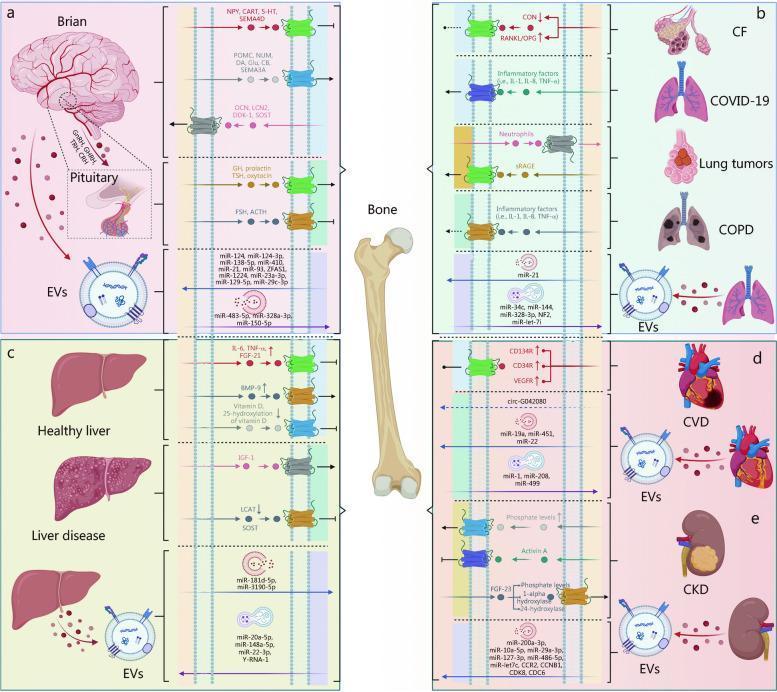
Panel (a) shows the bone–brain axis: bone sends OCN, LCN2, DKK-1, and SOST; brain/pituitary sends NPY, CART, 5-HT, SEMA4D, and POMC-derived peptides. Extracellular vesicles (EVs) with miRNAs serve as molecular couriers in both directions.
Diagram 2 — Bone-Derived Proteins Crossing the Blood–Brain Barrier
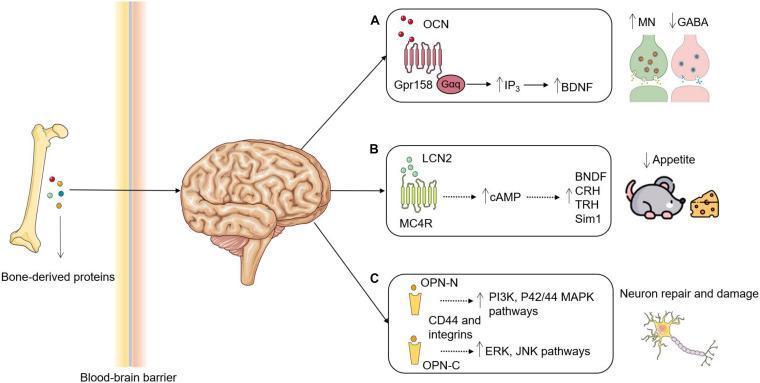
Three key bone-to-brain signalling pathways: A) OCN → GPR158 → Gαq/IP₃/BDNF → ↑monoamines, ↓GABA; B) LCN2 → MC4R → ↑cAMP → appetite suppression; C) OPN-N → neuron repair; OPN-C → neuronal damage.
Diagram 3 — Brain–Bone Neural Regulation
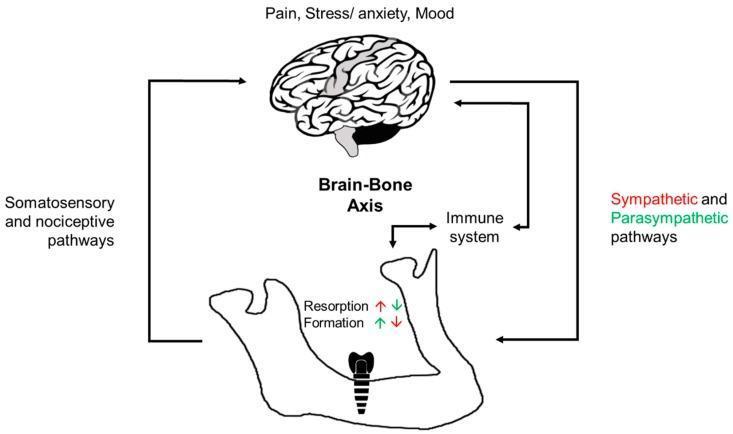
The brain modulates bone via: sympathetic NS (promotes resorption, inhibits formation), parasympathetic NS (promotes formation, inhibits resorption), immune system mediators, and somatosensory/nociceptive feedback.
3. Components / Key Players
A. Bone → Brain Signals (Bone-Derived Factors)
| Factor | Source Cell | Brain Receptor | Effect on CNS |
|---|---|---|---|
| Osteocalcin (OCN) | Osteoblasts | GPR158, GPRC6A, GPR37 | ↑ Monoamine synthesis (serotonin, dopamine, norepinephrine); ↓ GABA; ↑ BDNF; ↑ cognition; stress response regulation |
| Lipocalin-2 (LCN2) | Osteoblasts | MC4R (hypothalamus) | ↑ cAMP → appetite suppression; ↑ BDNF, CRH, TRH |
| Osteopontin (OPN) | Osteoblasts/osteocytes | CD44, integrins | OPN-N → neuron repair (PI3K/MAPK); OPN-C → neuronal damage (ERK/JNK) |
| FGF-23 | Osteocytes | FGFR1 in brain | Phosphate/Vit D homeostasis; influences cognitive function |
| Sclerostin (SOST) | Osteocytes | Crosses BBB | Wnt pathway modulation in neurons |
| RANKL/OPG | Osteoblasts | Brain microglia | Neuroinflammation modulation |
| Extracellular Vesicles (EVs) | Bone marrow | Multiple | Transport miRNAs (miR-21, miR-124-3p etc.) modulating neuroplasticity |
B. Brain → Bone Signals (Neural/Neuroendocrine Factors)
| Factor | Source | Receptor on Bone | Effect on Bone |
|---|---|---|---|
| Sympathetic NS (norepinephrine) | Hypothalamus → SNS | β2-adrenergic receptor (osteoblasts) | ↑ Bone resorption, ↓ bone formation |
| Parasympathetic NS (acetylcholine) | PNS | Muscarinic receptors | ↑ Bone formation, ↓ resorption |
| Leptin | Hypothalamus (indirect) | Osteoblast leptin receptors | Inhibits bone formation (via hypothalamic relay); also has direct anabolic effects |
| NPY (Neuropeptide Y) | Hypothalamus, sympathetic neurons | Y1/Y2 receptors on osteoblasts | Y2 activation → ↓ bone formation |
| Serotonin (5-HT) | Raphe nuclei (central) | 5-HT receptors on osteoblasts | Central 5-HT → ↓ bone mass (via sympathetic outflow); gut serotonin has opposite effect |
| POMC-derived peptides (ACTH, α-MSH) | Pituitary | MC2R, MC5R on bone cells | Modulate osteoclast/osteoblast activity |
| FSH | Pituitary | FSH receptors on osteoclast precursors | ↑ Bone resorption (especially post-menopause) |
| Glucocorticoids (stress axis — HPA) | Adrenal cortex | GR on osteoblasts | ↑ Osteoblast apoptosis, ↓ bone formation |
4. Types of Bone–Brain Axis Communication
Type 1: Endocrine (Hormonal) Communication
- Bone-derived hormones (OCN, LCN2, FGF-23) enter systemic circulation → cross BBB → bind CNS receptors
- Most studied pathway; OCN is the principal "bone hormone" acting on the brain
Type 2: Neural (Autonomic) Communication
- Sympathetic: hypothalamus → lateral hypothalamic area → spinal cord → sympathetic chain → β2-adrenergic receptors on osteoblasts → ↑ RANKL, ↓ OPG → net resorption
- Parasympathetic: vagal and local cholinergic innervation → muscarinic receptors → ↑ bone formation
- Sensory/Nociceptive: sensory nerves in bone (substance P, CGRP) send pain/stress signals to the brain; important in fracture pain and bone disease
Type 3: Immune-Mediated (Osteoimmune) Communication
- Bone marrow is a primary immune organ; immune cells (macrophages, T/B cells) regulate osteoclast differentiation via RANKL/OPG system
- Brain neuroinflammation (microglia activation, IL-1β, TNF-α, IL-6) affects bone remodelling via HPA axis and direct circulating cytokines
Type 4: Extracellular Vesicle (EV) Communication
- Bone marrow mesenchymal stem cells and osteoblasts release EVs containing miRNAs (miR-21, miR-124-3p, miR-138-5p) that cross the BBB and regulate neuroplasticity, neuroinflammation, and glial function
- Brain-derived EVs also carry miRNAs that influence osteogenesis
Type 5: Shared Molecular Pathway Communication
- Wnt/β-catenin: active in both osteoblasts and neurons; sclerostin (a Wnt inhibitor from bone) can cross BBB
- RANKL/NF-κB: regulates both osteoclastogenesis and microglial activation
- Leptin–melanocortin system: integrates energy metabolism, bone mass, and brain reward circuits
5. Applications / Clinical Significance
| Domain | Application |
|---|---|
| Neurodegenerative disease | OCN levels inversely correlate with Alzheimer's disease (AD) and Parkinson's disease (PD) severity; exogenous OCN may be neuroprotective |
| Osteoporosis ↔ Dementia comorbidity | Bidirectional vicious cycle: ↓ bone density → ↓ OCN → worsens AD pathology; AD neurodegeneration → sympathetic overactivation → ↑ bone loss |
| Depression & anxiety | Bone-derived OCN regulates the acute stress response (fight-or-flight); ↓ OCN → impaired stress resilience; links bone health to psychiatric disorders |
| Cognitive function | Higher OCN in circulation correlates with better memory and learning in both animal models and aging humans |
| Fracture pain & bone healing | Neural sensitisation and sympathetic activity post-fracture influence healing; chronic pain → HPA activation → glucocorticoid-mediated bone loss |
| Therapeutic targeting | Bisphosphonates (anti-osteoporosis drugs) improve cognitive outcomes; lithium (GSK-3β inhibitor) simultaneously protects both bone (Wnt activation in osteoblasts) and brain (neuroprotection) |
| Whole-body vibration therapy | Modulates osteoclast mechanosensing axis → secondary CNS effects |
| Biomarker discovery | Serum OCN, LCN2, FGF-23, and SOST as dual bone-brain biomarkers for early diagnosis of combined neuroskeletal decline |
6. Disadvantages / Limitations / Challenges
| Limitation | Details |
|---|---|
| Mechanistic complexity | Multiple overlapping signalling axes make it difficult to isolate and study single pathways; OCN, LCN2, and OPN all act simultaneously on different CNS targets |
| Translational gap | Most mechanistic data from rodent models; human equivalents not always confirmed — e.g., OCN's role in stress response demonstrated in mice lacks large-scale human RCT evidence |
| Confounding factors | Aging, obesity, metabolic syndrome, and inflammation all affect both bone and brain simultaneously, making it difficult to attribute effects specifically to the bone–brain axis |
| Blood–brain barrier variability | Only certain molecules (like OCN, SOST) cross the BBB; most bone-derived cytokines do not freely enter the CNS, limiting direct action |
| Receptor heterogeneity | GPR158 shows bidirectional effects in different brain regions and pathological conditions (e.g., neuroprotective in some contexts, pro-tumorigenic in others), complicating therapeutic targeting |
| Lack of standardised biomarkers | No validated clinical test currently integrates bone-brain axis status; OCN assays are not standardised across labs |
| Therapeutic dilemma | Drugs targeting one arm of the axis can adversely affect the other — e.g., glucocorticoids needed for neuroinflammation are highly detrimental to bone mass |
| Temporal dynamics | Bone remodelling cycles (weeks–months) operate on very different timescales from neural signalling (milliseconds–hours), making chronobiological integration poorly understood |
7. Key Signalling Pathways Summary
BONE SIDE ←→ BRAIN SIDE
─────────────────────────────────────────────────────────────
Osteocalcin (OCN) ──────────────────▶ GPR158/GPRC6A
↓
Gαq → IP₃ → BDNF
↓
↑ Serotonin, Dopamine
↓ GABA → Anxiolytic + Cognitive ↑
LCN2 ───────────────────────────────▶ MC4R (hypothalamus)
↓ cAMP ↑
↓ Appetite, ↑ BDNF/CRH
FGF-23 ─────────────────────────────▶ FGFR1 in brain
Phosphate/Vit D homeostasis
Sympathetic NS ◀──────────── Lateral hypothalamus (leptin, NPY)
(β2-AR on osteoblasts)
↑ RANKL → ↑ Osteoclastogenesis → Net Bone Loss
Wnt/β-catenin ←────────────────────── Sclerostin (SOST from osteocytes)
(shared pathway in neurons and osteoblasts)
8. Summary Table
| Feature | Bone → Brain | Brain → Bone |
|---|---|---|
| Key molecules | OCN, LCN2, FGF-23, OPN, EVs | Leptin, NPY, serotonin, NE, ACTH |
| Primary pathway | Endocrine (blood → BBB) | Neural (SNS/PNS) + Endocrine (HPA) |
| Main effects | Cognition ↑, mood regulation, stress response, neurogenesis | Bone remodelling (formation/resorption balance) |
| Disease links | Low OCN → AD, PD, depression | SNS overactivation → osteoporosis |
| Therapeutic targets | Recombinant OCN, GPR158 agonists | β-blockers (propranolol reduces fracture risk), lithium |
References (PubMed):
- Shi & Chen, Eur J Med Res 2024 — [PMID 38849920]
- Hansda & Das, Biology 2025 — [PMID 41007423]
- Guan et al., Biology 2025 — [PMID 41007258]
- Chen et al., Neuroscience 2026 — [PMID 41391736]
- Li et al., Front Cell Dev Biol 2025 — [PMID 40337551]
- Lee et al., Curr Osteoporos Rep 2026 — [PMID 41663782]
- Kang et al., Front Pharmacol 2025 — [PMID 40727110]
Tendon transfer definition types application and disadvantages structures everything for to answer 10 marks question with appropriate illustrative diagrams
Checking Date & Time
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.