Vitreous haemorrhage
Reading File
Finding Sources
Searching PubMed
"vitreous hemorrhage" AND management
Reading File
Reading File
Finding Sources
Searching Images
vitreous haemorrhage fundus photograph
Reading File
Loading Image
I now have comprehensive content to deliver a thorough answer. Here is the full clinical overview:
Vitreous Haemorrhage
Definition
Vitreous haemorrhage (VH) is the extravasation of blood into the vitreous cavity. It is one of the most common causes of sudden visual loss in adults.
Causes
| Category | Specific Causes |
|---|---|
| Posterior vitreous detachment (PVD) | With retinal tear or avulsion of a peripheral vessel |
| Proliferative retinopathy | Diabetic retinopathy, retinal vein occlusion, sickle cell disease, Eales disease, vasculitis |
| Miscellaneous retinal disorders | Macroaneurysm, telangiectasia, capillary haemangioma |
| Trauma | Blunt or penetrating injury, often associated with PVD; chorioretinitis sclopetaria |
| Systemic | Bleeding disorders, Terson syndrome |
The most common causes overall are proliferative diabetic retinopathy (PDR) and PVD with a retinal tear.
Clinical Features
Symptoms
- Mild VH: sudden-onset floaters and diffuse visual blurring; visual acuity may be preserved
- Dense VH: severe visual loss (vision may be reduced to light perception)
- Patients often describe red or dark haze, "cobwebs," or a "blood-red" curtain
Signs
- Red reflex is reduced or absent
- Fundus view is partially or completely obscured
- In trauma/PVD context, "tobacco dust" (pigment cells — Shafer's sign) in the anterior vitreous indicates probable retinal break
Investigations
B-scan Ultrasonography ⚠️ Mandatory with dense VH
- Non-clotted VH: uniform appearance on B-scan
- As cellular aggregates form: small particulate echoes
- Critical to exclude retinal tear or detachment and choroidal melanoma when the fundus cannot be visualised
Tip: Always perform B-scan ultrasonography when a dense vitreous haemorrhage prevents fundal view — to exclude retinal detachment or choroidal melanoma. — Kanski's Clinical Ophthalmology, 10th ed.
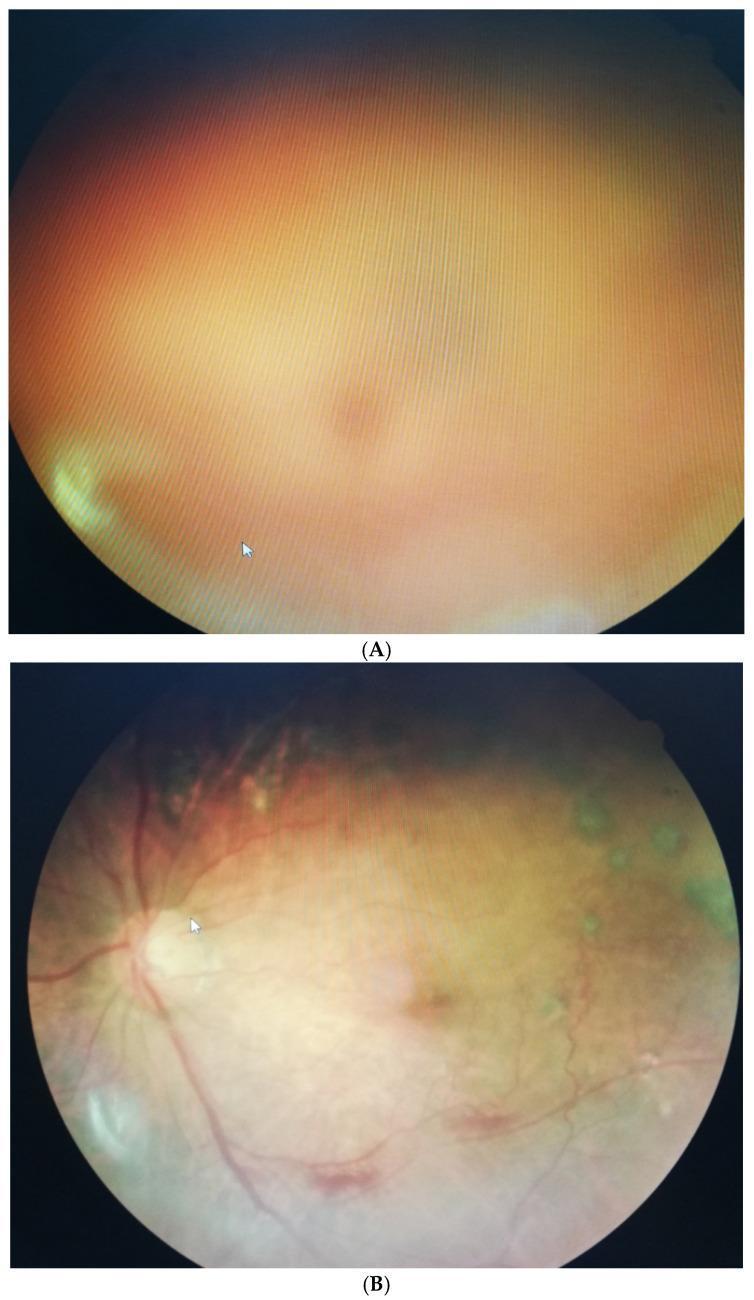
Fundus Appearance
Dense vitreous haemorrhage obscuring the fundus (A), and the same eye 3 months later after clearance (B):
Management
Management is dictated by severity and underlying cause.
Conservative
- Many cases (e.g., Terson syndrome, mild PVD-related) resolve spontaneously within weeks to months
- Head elevation / bed rest: promotes gravitational settling of blood inferiorly
- Avoid anticoagulants/aspirin where possible
- Control systemic disease (e.g., blood glucose, blood pressure)
Medical / Laser
- Panretinal photocoagulation (PRP): for underlying proliferative retinopathy once media clears
- Intravitreal anti-VEGF (e.g., aflibercept): an alternative to early vitrectomy for PDR-related VH. DRCR.net Protocol AB showed that vitrectomy + PRP achieves faster visual recovery than initial aflibercept, but 2-year visual outcomes are similar — either is a reasonable first-line approach
- Nd:YAG laser hyaloidotomy: for persistent premacular (retrohyaloid) haemorrhage — disperses blood into the vitreous cavity for faster absorption
Surgical — Pars Plana Vitrectomy (PPV)
Indications include:
- Severe persistent VH that precludes adequate PRP (most common indication)
- In type 1 diabetics (absent rubeosis): traditionally considered after 3 months; earlier surgery may improve outcomes
- Bilateral VH
- VH with tractional retinal detachment threatening the macula — treat urgently
- Combined tractional + rhegmatogenous RD — treat urgently
- Dense premacular retrohyaloid haemorrhage (risk of ERM / tractional macular detachment)
- VH with concurrent retinal detachment
Special Scenarios
Terson Syndrome
- Intraocular haemorrhage + subarachnoid haemorrhage (usually ruptured anterior communicating artery aneurysm)
- Haemorrhage is typically bilateral, usually intraretinal/preretinal; may break into vitreous
- Mechanism: retinal venous stasis from elevated cavernous sinus pressure
- Most resolve spontaneously; early vitrectomy in selected cases
Traumatic VH
- Often associated with PVD; pigment cells in anterior vitreous (tobacco dust) warrant urgent peripheral retinal examination
- Chorioretinitis sclopetaria (high-velocity projectile near globe) may cause VH ± retinal detachment
Proliferative Diabetic Retinopathy (PDR)
- Most common cause of VH requiring vitrectomy
- Vitrectomy combined with extensive endolaser PRP
- Anti-VEGF pre-treatment can reduce intraoperative bleeding
Prognosis
- Depends heavily on the underlying cause and presence of complications (retinal detachment, neovascular glaucoma)
- Spontaneous absorption is common in non-proliferative causes
- PDR-related VH carries a guarded prognosis without treatment; good outcomes with timely vitrectomy + PRP
- Terson syndrome: long-term visual prognosis is generally good
— Kanski's Clinical Ophthalmology: A Systematic Approach, 10th Edition
For 20 marks
Checking Date & Time
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive material from three authoritative sources (Kanski's, Wills Eye Manual, Tintinalli's) to compose a detailed 20-mark answer. Here it is:
Vitreous Haemorrhage — 20-Mark Answer
Definition (1 mark)
Vitreous haemorrhage (VH) is the extravasation of blood into the vitreous cavity. It is one of the most common causes of sudden painless visual loss in adults.
Anatomy & Pathophysiological Basis (2 marks)
The vitreous is an avascular gel attached firmly to:
- The ora serrata anteriorly
- The optic nerve head posteriorly
- Along the major retinal vessels
Blood enters the vitreous from adjacent vascular structures — either through rupture of abnormal new vessels (neovascularisation), tearing of normal vessels during PVD, or direct trauma. Because the vitreous is avascular, blood is not cleared quickly and can persist for weeks to months.
Aetiology (3 marks)
| Category | Examples |
|---|---|
| Posterior vitreous detachment (PVD) | Avulsion of a peripheral vessel or an associated retinal tear — most common in elderly |
| Proliferative retinopathy | Diabetic retinopathy (PDR) — most common overall cause; retinal vein occlusion; sickle cell disease; Eales disease; vasculitis |
| Miscellaneous retinal | Arterial macroaneurysm; telangiectasia; capillary haemangioma; Coats disease; ROP; von Hippel–Lindau syndrome |
| Trauma | Blunt/penetrating injury; shaken baby syndrome; chorioretinitis sclopetaria |
| Systemic | Bleeding diathesis; haematological malignancy; anticoagulation (warfarin) |
| Terson syndrome | Subarachnoid/subdural haemorrhage → raised intracranial pressure → retinal/vitreous bleed |
| Intraocular tumour | Choroidal melanoma, retinoblastoma |
Paediatric note: In infancy and childhood, consider birth trauma, non-accidental injury (shaken baby), congenital X-linked retinoschisis, pars planitis, and bleeding dyscrasias.
Clinical Features (3 marks)
Symptoms
- Sudden, painless loss of vision
- Sudden appearance of floaters, cobwebs, black spots, or a red/dark haze
- Dense VH: severe visual loss down to light perception
Signs
Critical:
- Reduced or absent red fundus reflex
- Partial/total obscuration of fundus view
- Red blood cells visible in anterior vitreous on slit-lamp
- Chronic VH: yellow-ochre appearance (haemoglobin breakdown)
Other:
- Mild RAPD may be present with dense haemorrhage
- "Tobacco dust" (Shafer's sign) — pigment cells in anterior vitreous — indicates likely retinal break
- Dependent layering of blood inferiorly
- Iris neovascularisation (rubeosis) in PDR
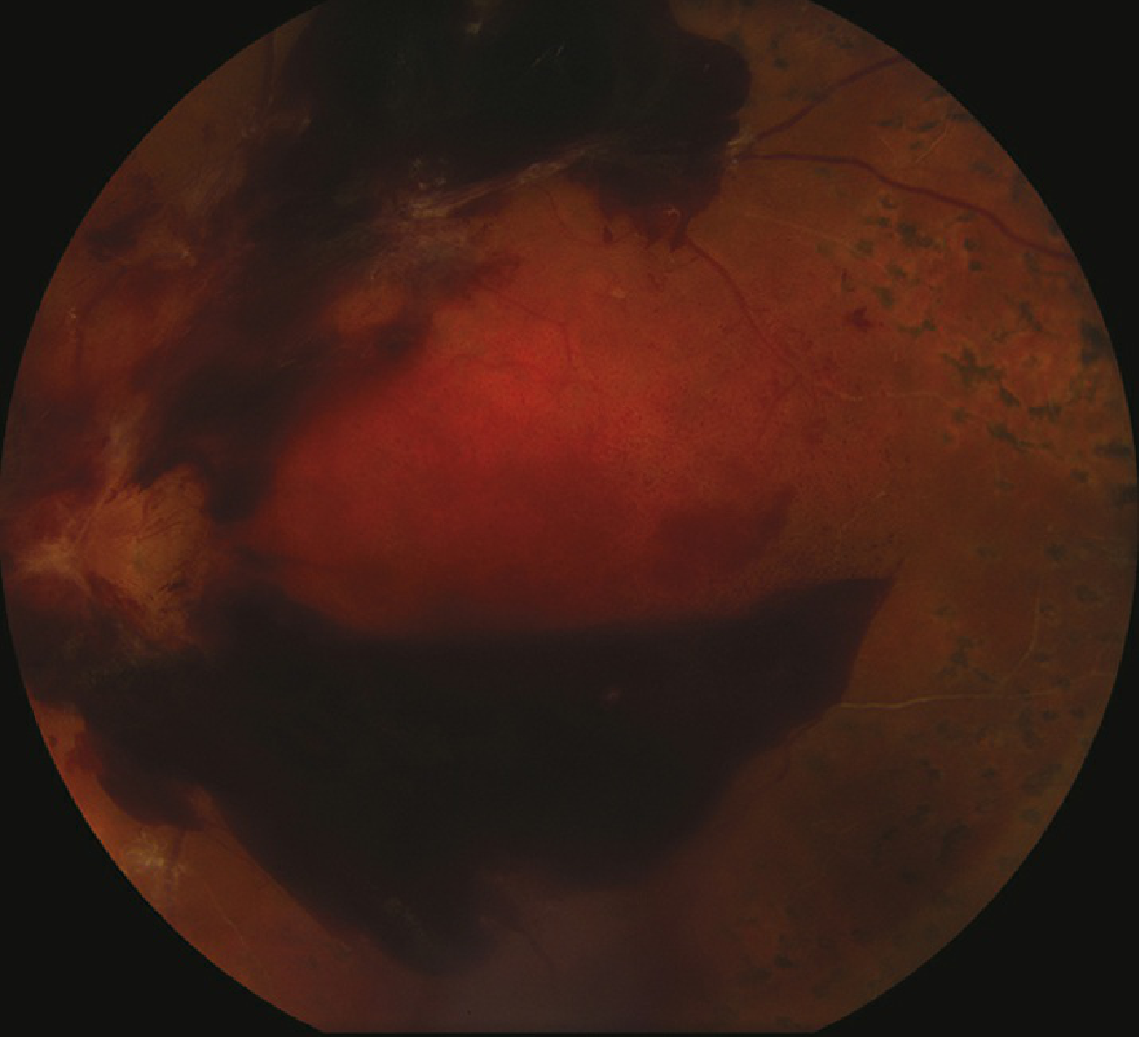
Differential Diagnosis (1 mark)
- Vitritis (uveitis): WBCs, not RBCs; not sudden onset
- Retinal detachment without VH: similar visual symptoms
- Vitreous amyloidosis: glass-wool opacities, bilateral, systemic history
- Asteroid hyalosis: calcium pyrophosphate deposits — not true haemorrhage; moves with vitreous but does not sediment
Investigations / Workup (3 marks)
1. History
- Systemic disease: diabetes, hypertension, sickle cell, bleeding disorder
- Trauma, recent surgery, headache (Terson)
- Prior episodes, anticoagulant use
2. Ocular Examination
- Slit-lamp (undilated): look for iris neovascularisation, anterior chamber red cells
- IOP measurement (ghost cell glaucoma risk)
- Dilated fundus examination (indirect ophthalmoscopy ± scleral depression)
- Examine contralateral eye — may give clues to the diagnosis (e.g., diabetic changes, AMD, sickle cell "sea fan" neovascularisation)
3. B-scan Ultrasonography ⚠️ Mandatory when fundus is obscured
- Acute/mild VH: scattered low-amplitude mobile opacities (small dots)
- Mature/organised VH: thick mobile membranes, multiple large echoes; blood layers inferiorly
- Key clinical use: exclude retinal detachment, retinal tear (elevated flap), or choroidal melanoma
- Differentiating VH from RD on B-scan:
- RD — hyperechoic membrane moves with eye movements and is fixed at the optic disc
- VH — echoes remain horizontal (gravitational), found in the middle of the posterior chamber
4. Fluorescein Angiography (IVFA)
- Aids in defining aetiology once media clarity permits
- Useful to define extent of neovascularisation or ischaemia
5. Systemic Investigations
- Blood glucose, HbA1c (diabetes)
- FBC, clotting screen, INR (if on warfarin, haematological disease)
- Sickle cell screen in appropriate demographics
- CT/MRI head if Terson syndrome suspected
Management (5 marks)
A. Conservative (Observation)
- Head of bed elevated — promotes inferior settling of blood, clearing visual axis superiorly (also the most common site for retinal breaks)
- Restrict: no heavy lifting, straining, or bending
- Discontinue aspirin, NSAIDs, and anticoagulants unless medically necessary
- Treat the underlying cause as soon as possible:
- Retinal break → laser photocoagulation or cryotherapy
- Proliferative retinopathy → anti-VEGF or PRP
- Detached retina → surgery
Follow up: daily for first 2–3 days if observation elected; if dense VH persists with unknown aetiology, proceed to vitrectomy.
B. Laser
- Panretinal photocoagulation (PRP): for underlying PDR once adequate view obtained; reduces neovascularisation
- Nd:YAG laser hyaloidotomy: for persistent premacular retrohyaloid haemorrhage — disperses blood into vitreous cavity for faster absorption; reduces risk of ERM/tractional macular detachment
C. Intravitreal Anti-VEGF
- Agents such as aflibercept, bevacizumab, ranibizumab used for PDR-related VH
- DRCR.net Protocol AB: eyes treated with early vitrectomy + PRP regained vision faster than those given initial aflibercept; however, 2-year visual outcomes were similar — both are acceptable first-line options for VH from PDR
- Pre-operative anti-VEGF reduces intraoperative bleeding in complex vitrectomy
D. Pars Plana Vitrectomy (PPV) — Surgical
Indications:
- Severe persistent VH precluding adequate PRP (most common indication)
- Type 1 diabetics without rubeosis — traditionally within 3 months; earlier surgery may improve outcomes
- Bilateral VH (most cases)
- VH with tractional retinal detachment threatening the macula → treat without delay
- Combined tractional + rhegmatogenous RD → treat urgently
- Dense premacular retrohyaloid haemorrhage (risk of fibrovascular scaffold formation)
- Non-clearing VH of unknown aetiology — two-thirds of such cases will have retinal tears or detachment
- VH with iris neovascularisation (rubeosis)
- Hemolytic or ghost cell glaucoma
Procedure: PPV is typically combined with extensive endolaser PRP; visual results depend on the specific indication and severity of underlying disease.
Tip: Persistent VH or progressive tractional RD threatening the macula should be treated as soon as possible with pars plana vitrectomy. — Kanski's Clinical Ophthalmology, 10th ed.
Complications of VH (1 mark)
- Ghost cell glaucoma: degenerated erythrocytes (ghost cells) obstruct the trabecular meshwork
- Hemolytic glaucoma: macrophages laden with haemoglobin products obstruct outflow
- Tractional retinal detachment: fibrovascular proliferation along the posterior hyaloid
- Epiretinal membrane (ERM)
- Amblyopia in children
- Persistent severe visual loss
Prognosis (1 mark)
- Depends on underlying cause and presence of complications
- PVD-related and Terson syndrome VH: generally good prognosis with spontaneous resolution
- PDR-related VH: guarded without treatment; good functional outcomes with timely vitrectomy + PRP
- VH + retinal detachment or neovascular glaucoma: worse prognosis
Sources: Kanski's Clinical Ophthalmology (10th ed.) · Wills Eye Manual · Tintinalli's Emergency Medicine
Pan retinal photo coagulation in detail for 20 marks
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching Images
panretinal photocoagulation retinal burns fundus photograph
Loading Image
Loading Image
I now have rich, detailed content from Kanski's across all key domains. Here is the comprehensive 20-mark answer:
Panretinal Photocoagulation (PRP) — 20-Mark Answer
1. Definition (1 mark)
Panretinal photocoagulation (PRP), also called scatter laser photocoagulation, is a laser treatment in which 1,500–7,000 burns are applied to the peripheral retina (outside the temporal vascular arcades), deliberately destroying ischaemic peripheral retinal tissue to cause regression of retinal and iris neovascularisation. It remains the mainstay of treatment for proliferative retinopathy in most healthcare systems.
2. Mechanism of Action (2 marks)
Thermal laser energy is absorbed by melanin in the retinal pigment epithelium (RPE) and haemoglobin in retinal vessels, converting it to heat that destroys the outer retinal layers (photoreceptors + RPE).
This produces its beneficial effect through:
- Destruction of ischaemic peripheral photoreceptors — the most metabolically demanding cells in the retina — reducing total retinal oxygen demand
- Improved oxygenation of inner retina — by eliminating the oxygen-consuming outer layers, oxygen from the choroid can diffuse further inward, reducing inner retinal hypoxia
- Reduction of VEGF production — the ischaemic peripheral retina is the major source of VEGF; ablating this tissue dramatically reduces VEGF secretion
- Regression of neovascularisation — lowered VEGF leads to closure and fibrosis of new vessels (NVD, NVE, NVI)
- Possible RPE scar-mediated effects — laser scars may directly alter cytokine production from the RPE
The net result: ischaemic drive for neovascularisation is eliminated → existing new vessels regress → risk of vitreous haemorrhage and tractional retinal detachment is reduced.
3. Indications (3 marks)
Primary Indication: Proliferative Diabetic Retinopathy (PDR)
The Diabetic Retinopathy Study (DRS) established PRP's benefit in high-risk PDR:
- Severe NVD (new vessels at disc) without haemorrhage: 26% risk of severe visual loss at 2 years → reduced to 9% with PRP
- NVD with haemorrhage, or NVE > ½ disc area with haemorrhage: high-risk characteristics requiring prompt PRP
High-risk PDR characteristics (DRS criteria):
- NVD ≥ ¼–⅓ disc area
- Any NVD with preretinal or vitreous haemorrhage
- NVE ≥ ½ disc area with preretinal or vitreous haemorrhage
Other Indications:
| Condition | When to Apply PRP |
|---|---|
| Retinal vein occlusion (CRVO, BRVO) | When neovascularisation of iris/angle/retina develops |
| Sickle cell retinopathy | Proliferative sickle cell retinopathy ("sea fan" NV) |
| Eales disease | Peripheral retinal neovascularisation |
| Retinal vasculitis | With neovascularisation |
| Rubeosis iridis (neovascular glaucoma) | Combined with other treatment |
| Before cataract surgery in PDR | To reduce perioperative risk |
| Intraoperative during pars plana vitrectomy | Combined with endolaser during surgery |
4. Pre-Treatment Evaluation (1 mark)
- Dilated fundus examination (indirect ophthalmoscopy + slit-lamp biomicroscopy)
- Fluorescein angiography (FA): delineates extent of NV, identifies areas of capillary non-perfusion, guides treatment boundaries
- Wide-field FA: accurately maps peripheral capillary non-perfusion for targeted/selective treatment
- OCT: assess for co-existing diabetic macular oedema (DMO) before PRP
- IOP measurement; iris neovascularisation check (undilated slit-lamp)
Key principle: If co-existing clinically significant macular oedema (CSMO) or central-involving DMO is present, treat the oedema first (with anti-VEGF or focal/grid laser), then perform PRP — to avoid exacerbation by PRP.
5. Informed Consent (1 mark)
Patients must be counselled about:
- Visual field loss: PRP reduces peripheral field; may be severe enough to legally preclude driving — though most patients with initially good vision maintain the binocular field standard required
- Risk to central vision: PRP can worsen or precipitate macular oedema — reduced by fractionating treatment over 2–3 sessions
- Night vision impairment: destruction of peripheral rod photoreceptors
- Colour vision changes
- Alternative: Anti-VEGF therapy (DRCR.net Protocol S, CLARITY studies) is as effective as PRP at 5 years for PDR; patient may choose this route, but must understand the need for repeated injections and shorter-term follow-up data
6. Equipment and Setup (1 mark)
Laser Sources
- Argon green (514 nm): historically standard
- Diode (810 nm): penetrates media opacity better; absorbed by melanin preferentially
- Frequency-doubled Nd:YAG (532 nm): common in modern systems
- Pattern scan laser (PASCAL): delivers pre-programmed multispot arrays with very short pulses, allowing rapid delivery of large numbers of burns
Contact Lenses
- Panfundoscopic lens (e.g. Volk SuperField, Mainster PRP 165): preferred; provides wide-field view of peripheral retina
- ⚠️ With panfundoscopic lenses: actual retinal burn diameter is twice the interface setting (200 μm selected → 400 μm retinal burn)
- Three-mirror (Goldmann) lens: alternative; requires more repositioning
- High-magnification lens (Mainster, Area Centralis): for posterior component of treatment near arcades
Anaesthesia
- Topical (proxymetacaine drops): adequate in most cases
- Sub-Tenon or peribulbar block: for patients with severe discomfort, especially peripheral/horizontal meridian treatment
7. Laser Parameters (3 marks)
| Parameter | Standard Setting |
|---|---|
| Spot size | 400 μm retinal diameter (200 μm on interface with panfundoscopic lens) |
| Duration | 0.05–0.1 s (argon); 0.01–0.05 s (10–50 ms) with modern/pattern scan lasers |
| Power | Titrated to produce light grey/white burn (mild intensity); just above threshold |
| Spacing | Burns separated by 1–1.5 burn widths |
| Total burns | 1,500 per session initially; 2,500–3,500 total for mild PDR; 4,000 for moderate; 7,000 for severe PDR |
| Sessions | Fractionated over 2–3 sessions to reduce complications |
Burn intensity: only a light (grey-white) burn is desired — heavy/intense burns increase complication risk without additional benefit.
8. Treatment Pattern and Extent (2 marks)
Boundaries of Treatment
- Posteriorly: outside the temporal macular vascular arcades (approximately 2 disc diameters from the foveal centre)
- Nasally: usually 2 disc diameters nasal to the optic disc are left untreated (to preserve paracentral field)
- Anteriorly: extends to ora serrata
Technique
- Apply a "barrier" row of laser burns just temporal to the macula early in the procedure — protects against accidental macular damage
- In severe PDR: treat the inferior fundus first — any subsequent vitreous haemorrhage gravitates inferiorly and would otherwise obscure this area
- Avoid areas of vitreoretinal traction — laser over traction sites can precipitate haemorrhage or tractional detachment
- Review at 4–6 weeks after completing adequate burns; additional sessions applied as needed
Multispot / Pattern Scan (PASCAL)
- Short pulses (e.g. 20 ms), very short intervals, pre-programmed arrays
- Delivers large number of burns rapidly
- Shorter pulse duration may require a greater total number of burns for equivalent response
Targeted Retinal Photocoagulation (TRP)
- Wide-field FA guides selective treatment of only the ischaemic/non-perfused peripheral zones
- Achieves NV regression while minimising collateral damage to perfused retina
- Reduces side effects compared to conventional full PRP
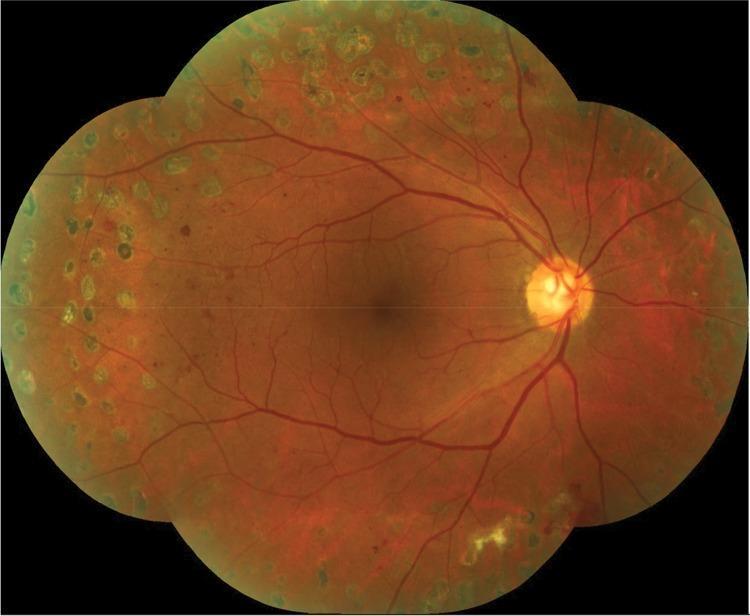
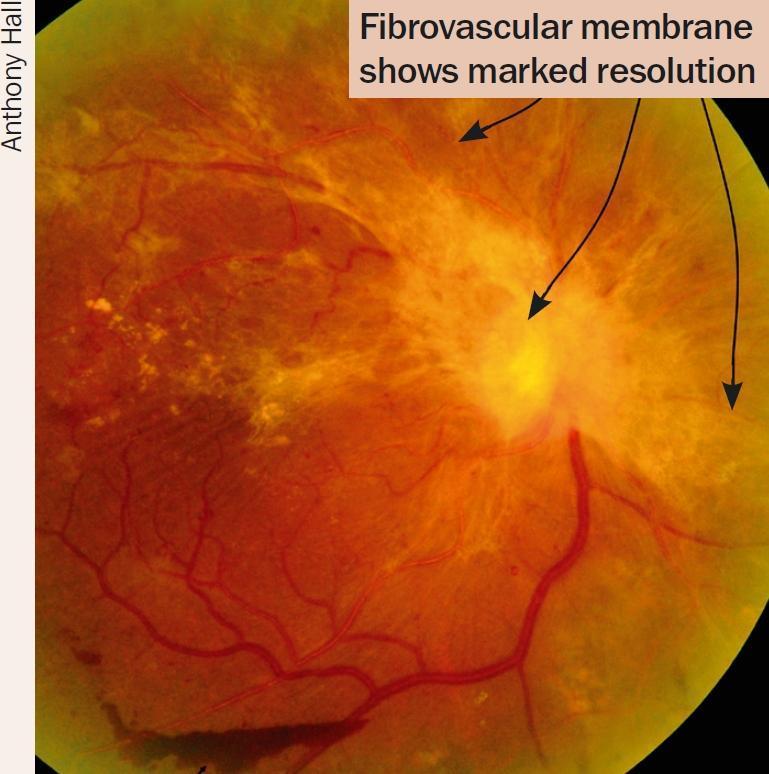
9. Indicators of Successful Regression (1 mark)
- Blunting of neovascular vessel tips
- Shrinking and disappearance of NV — leaving "ghost" vessels or fibrosis
- Regression of intraretinal microvascular abnormalities (IRMA)
- Decreased venous beading and dilation
- Absorption of retinal haemorrhages
- Disc pallor (in some cases)
10. Complications (2 marks)
Immediate / Short-term
| Complication | Mechanism |
|---|---|
| Pain | Especially peripheral/horizontal meridian treatment; limits burns per session |
| Vitreous haemorrhage | Contraction of regressing vessels or vitreous separation induced by treatment |
| Macular oedema (DMO) | Treatment-induced breakdown of blood-retinal barrier; exacerbated by large single sessions — reduced by fractionating |
| Choroidal effusion | Extensive single-session treatment |
| Exudative retinal detachment | With very extensive burns in one session |
| Corneal burn | With improper technique or contact lens coupling |
Long-term / Chronic
| Complication | Notes |
|---|---|
| Visual field constriction | Tunnel vision in extensive treatment; may affect driving |
| Night blindness | Loss of peripheral rod photoreceptors |
| Colour vision changes | |
| Decreased contrast sensitivity | |
| Tractional retinal detachment | Contraction of residual fibrovascular tissue post-regression |
| Epiretinal membrane (ERM) | Recognised complication of PRP |
| Inadvertent foveal burn | If technique is poor or lens orientation confused (inverted/reversed image) |
| Choroidal neovascular membrane | Rare; if burns extend subfoveal |
| Accommodative paresis | Ciliary body involvement |
11. PRP vs. Anti-VEGF — Current Evidence (2 marks)
| Study | Key Finding |
|---|---|
| DRCR.net Protocol S (5 years) | Intravitreal ranibizumab non-inferior to PRP for PDR at 5 years; fewer VH episodes; better macular oedema profile; slightly better visual field preservation with anti-VEGF |
| CLARITY study (1 year) | Intravitreal aflibercept as effective as PRP in PDR |
| DRCR.net Protocol AB | For VH from PDR: early vitrectomy + PRP gives faster visual recovery than initial aflibercept; 2-year outcomes similar — both acceptable |
| YOSEMITE / RHINE (2 years) | Faricimab (bispecific VEGF/Ang-2 inhibitor) shows promise |
Clinical implications:
- PRP remains preferred when anti-VEGF compliance is uncertain (single treatment course)
- Anti-VEGF preferred when co-existing DMO is present (DRCR Protocol S: 28% vs 42% DMO rate with anti-VEGF vs PRP)
- Anti-VEGF does not replace PRP in patients lost to follow-up — PRP provides more durable protection
- PRP + anti-VEGF combined may be used in severe PDR with co-existing DMO
12. Special Situations (1 mark)
- With co-existing DMO: treat macular oedema first with anti-VEGF (or focal/grid laser for non-central CSMO) before PRP. If focal laser to macula is needed, apply it before PRP in the same session.
- Before cataract surgery in PDR: PRP reduces risk of post-operative rubeosis and NVG
- Intraoperative (endolaser): applied during PPV via intraocular probe, allowing treatment despite media opacity
- Terson syndrome / vitreous haemorrhage: if PRP cannot be completed due to VH → proceed to early vitrectomy + intraoperative endolaser
- Rubeosis/NVG: PRP combined with anti-VEGF injection (intravitreal) for rapid NVI regression; PRP for long-term stability
Sources: Kanski's Clinical Ophthalmology: A Systematic Approach, 10th Edition · Wills Eye Manual, 8th Edition
This is a shared conversation. Sign in to Orris to start your own chat.