Enteric fever
Reading File
Finding Sources
Finding Sources
Searching PubMed
"enteric fever" OR "typhoid fever"
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
typhoid fever rose spots rash clinical
Reading File
Reading File
I now have comprehensive content. Let me compile the full answer.
Enteric Fever (Typhoid Fever)
Definition
Enteric (typhoid) fever is a systemic illness caused by dissemination of Salmonella enterica serotype Typhi (S. Typhi) or Paratyphi (A, B, or C). The term "enteric fever" was proposed in 1869 to distinguish it from typhus based on its anatomic site of infection (the small intestine). The two names are used interchangeably today.
— Harrison's Principles of Internal Medicine, 22E
Etiology & Microbiology
| Organism | Disease |
|---|---|
| S. Typhi | Typhoid fever (majority of cases in endemic areas) |
| S. Paratyphi A | Paratyphoid fever (more common among travelers; milder) |
| S. Paratyphi B, C | Paratyphoid fever (less common) |
Unlike nontyphoidal Salmonella, humans are the sole reservoir for S. Typhi and S. Paratyphi. Transmission is fecal-oral — via contaminated food or water, or from chronic carriers.
S. Typhi is resistant to gastric acid and initially invades via M cells of small intestinal Peyer's patches, then disseminates via lymphatic and blood vessels — in contrast to S. enteritidis, which remains confined to the gut.
Epidemiology
- 9.2–21 million cases of typhoid fever and 5 million cases of paratyphoid fever annually; 110,000–280,000 deaths/year
- Highest burden: Indian subcontinent (India, Pakistan, Bangladesh, Nepal), Eastern Mediterranean, Africa — >1000 cases/100,000 children in some urban areas
- Risk factors: contaminated drinking water, flooding, street food, raw vegetables grown in sewage-fertilized fields, lack of hand washing, H. pylori infection (reduced gastric acidity)
- In the US: ~5700 cases/year; 78% travel-associated, predominantly from South Asia
Drug Resistance — Critical Update
- Multidrug-resistant (MDR) strains emerged in the 1980s: resistance to chloramphenicol, ampicillin, and trimethoprim (plasmid-mediated)
- Decreased susceptibility to ciprofloxacin (DSC) and full fluoroquinolone-resistant strains (clone H58) spread from the Indian subcontinent to Africa
- Extensively drug-resistant (XDR) S. Typhi emerged in Sindh, Pakistan in 2016 — resistant to MDR antibiotics plus fluoroquinolones and third-generation cephalosporins; susceptible only to azithromycin and carbapenems
Pathogenesis & Morphology
- Ingestion → gastric acid resistance → invasion of small intestinal M cells
- Uptake by macrophages → spread to mesenteric lymph nodes → bacteremia
- Systemic dissemination: liver, spleen, bone marrow, gallbladder
Gross pathology:
- Peyer's patches in terminal ileum enlarge into sharply delineated, plateau-like elevations up to 8 cm
- Mesenteric lymph nodes enlarged
- Oval ulcers oriented longitudinally along ileal axis → risk of perforation
- Spleen: enlarged, soft, pale red pulp; prominent phagocyte hyperplasia
- Liver: small foci of parenchymal necrosis — "typhoid nodules" (macrophage aggregates replacing hepatocytes); also found in bone marrow and lymph nodes
— Robbins & Kumar Pathologic Basis of Disease
Clinical Course
Incubation period: 5–21 days (mean 10–14 days; depends on inoculum size, host immunity, vaccination status)
Week-by-week progression (untreated):
| Week | Features |
|---|---|
| Week 1 | Stepwise rising fever → sustained 39.4–40.5°C; headache (80%), chills, dry cough, malaise; relative bradycardia in up to 50%; rose spots appear (~30%) |
| Week 2 | Sustained high fever; abdominal distension, pain, diarrhea or constipation; hepatosplenomegaly; apathy, confusion ("muttering delirium") |
| Week 3–4 | Defervescence OR life-threatening complications: intestinal hemorrhage (~6%), intestinal perforation (~1%) |
Key clinical signs:
- Relative bradycardia (Faget's sign): pulse inappropriately slow for the degree of fever — seen in <50%
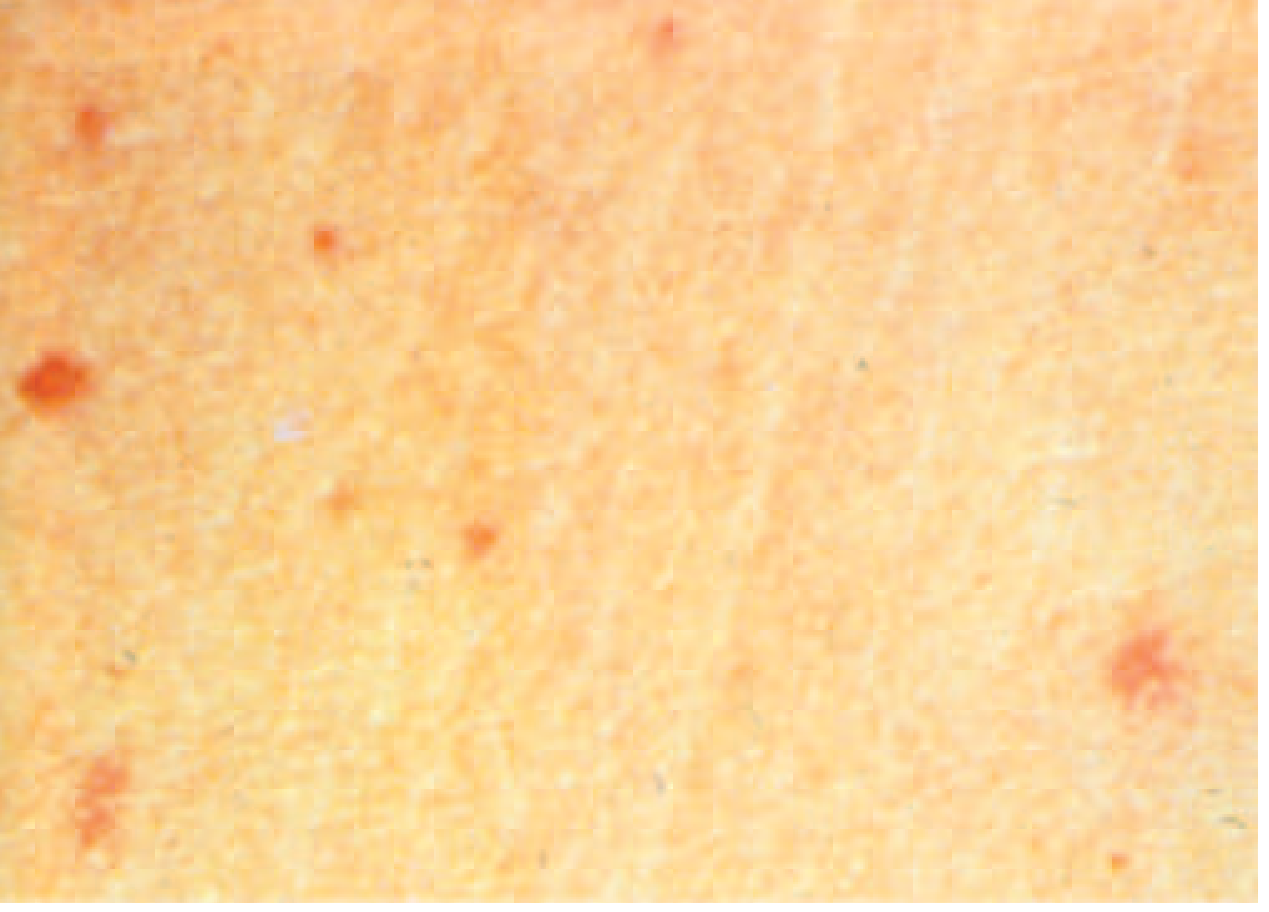
- Rose spots: faint, salmon-colored, blanching maculopapular rash on trunk and chest; present in ~30% of patients; may show 2–3 crops; bacteria can be cultured from punch biopsies; difficult to detect in dark-skinned patients
- Hepatosplenomegaly: ~50% of patients
- Coated tongue: 51–56%
Complications (~27% of hospitalized patients)
Gastrointestinal (weeks 3–4):
- Intestinal hemorrhage (~6%) — from necrosis of ileocecal Peyer's patches
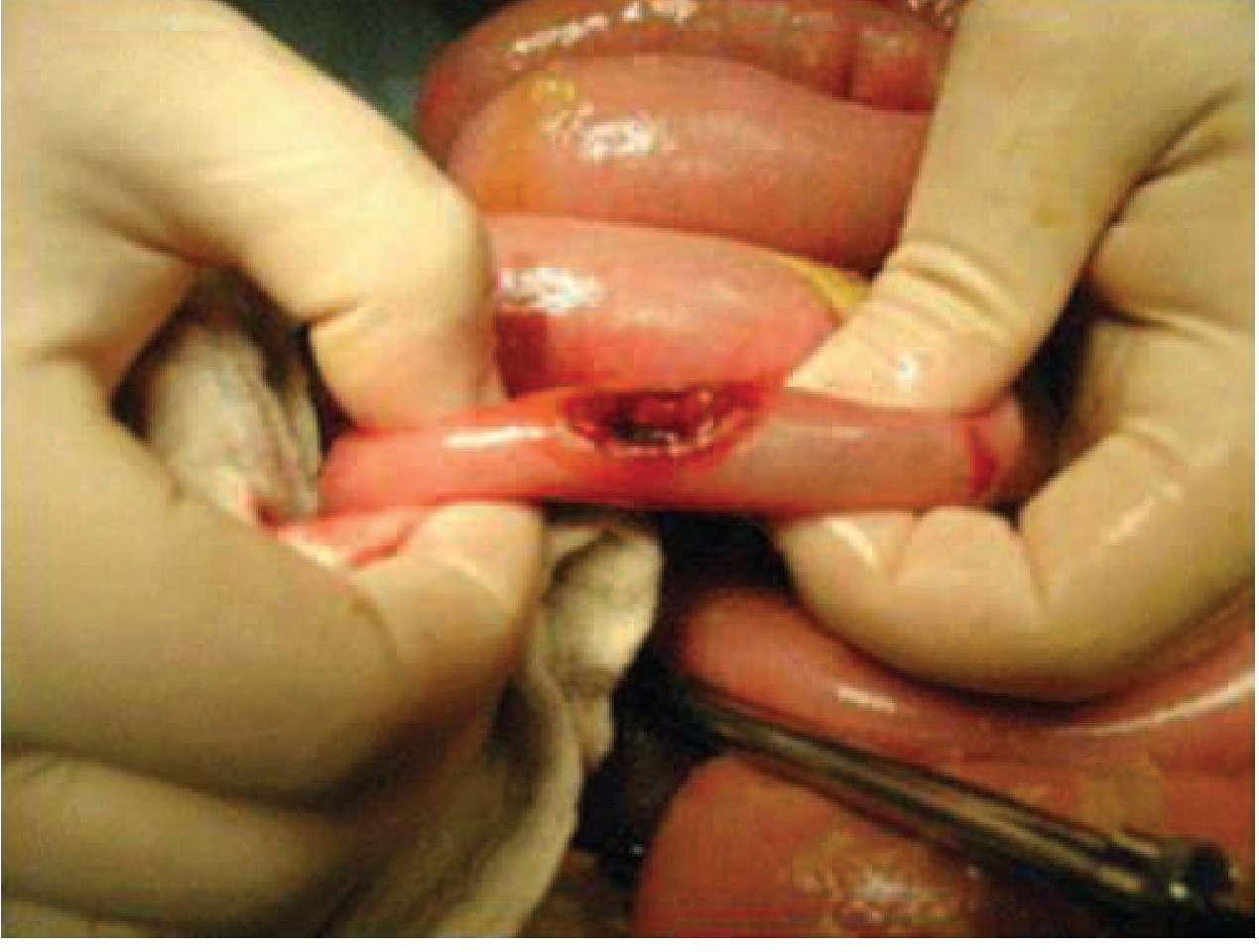
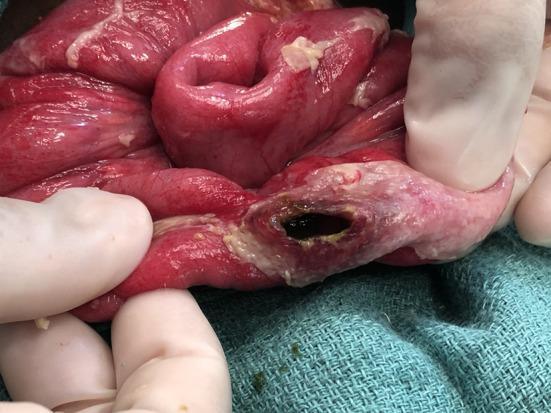
- Intestinal perforation (~1%) → polymicrobial peritonitis, requires emergency surgery
Neurologic (2–40%):
- Meningitis, Guillain-Barré syndrome, neuritis, neuropsychiatric symptoms ("muttering delirium," "coma vigil")
Other uncommon complications:
- Hemophagocytic syndrome, DIC, pancreatitis, hepatitis, endocarditis, myocarditis, orchitis, glomerulonephritis, pneumonia, osteomyelitis, parotitis
Chronic carriage:
- 2–5% of untreated patients develop chronic carriage (>1 year excretion in urine or stool)
- More common in women, infants, persons with biliary abnormalities or S. haematobium co-infection
- S. Typhi survives in gallbladder by forming biofilms on gallstones
- Associated with increased risk of gallbladder carcinoma
- Relapse: occurs in up to 10%, usually 2–3 weeks after fever resolution
Diagnosis
Definitive diagnosis = culture isolation of S. Typhi or S. Paratyphi
| Specimen | Sensitivity | Timing |
|---|---|---|
| Bone marrow culture | ~80% (highest) | Even after antibiotics |
| Blood culture | ~60–80% | First 1–2 weeks (bacteremia phase) |
| Stool culture | Positive later | Weeks 2–3 |
| Urine culture | Lower | Variable |
| Rose spot biopsy | Positive | When rash present |
Serologic tests:
- Widal test (agglutination of S. Typhi H and O antigens): Poor sensitivity and specificity; false positives from prior vaccination, other infections; not reliable in endemic areas — should not be used as the sole basis for diagnosis
- Modern rapid tests (Typhidot, TPTest) have variable performance
- PCR assays: increasingly used in research/reference settings
Non-specific lab findings:
- Leukopenia and neutropenia (15–25% of cases)
- Leukocytosis more common in children, first 10 days, or with perforation/secondary infection
- Moderately elevated liver enzymes
Treatment
Antibiotic Selection (guided by resistance pattern):
| Susceptibility | First-line Treatment | Notes |
|---|---|---|
| Fully susceptible | Fluoroquinolone (ciprofloxacin) | 5–7 days; high cure rate |
| MDR (resistant to chloramphenicol, ampicillin, TMP-SMX) | Fluoroquinolone or 3rd-gen cephalosporin (ceftriaxone) | |
| DSC/Fluoroquinolone-resistant | Azithromycin (oral, uncomplicated) or Ceftriaxone (IV, severe) | High-dose ciprofloxacin has reduced efficacy |
| XDR (resistant to above + cephalosporins) | Azithromycin (uncomplicated) or Carbapenem (meropenem, severe) | Emerging challenge; Pakistan outbreak strains |
Duration: Typically 7–14 days. Severe/complicated disease requires parenteral therapy.
Dexamethasone: For severe disease with altered consciousness/shock — shown to reduce mortality in a landmark trial.
Surgical management: Required for intestinal perforation — resection and peritoneal lavage.
Chronic carriers: Prolonged oral fluoroquinolone course (6 weeks); cholecystectomy if gallstones present.
⚠️ Recent evidence (2024): A systematic review (PMID 39623850) confirms oral azithromycin is clinically effective vs. other antimicrobials for typhoid across all age groups — relevant given the XDR strain emergence in South Asia.
Prevention
Vaccines:
| Vaccine | Type | Route | Efficacy | Notes |
|---|---|---|---|---|
| Vi polysaccharide (ViPS) | Subunit | IM | ~60–70% | ≥2 years; single dose; repeat every 2–3 years |
| Ty21a (Vivotif) | Live attenuated oral | Oral | ~65–70% | ≥6 years; 3–4 doses |
| Typhoid Conjugate Vaccine (TCV) | Vi-conjugated to tetanus toxoid | IM | ~80–85% | ≥6 months; preferred in children; WHO-recommended for endemic countries |
The TCV is now WHO-recommended for routine childhood immunization in typhoid-endemic countries. Global introduction 2019–2022 has been ongoing (as shown in the incidence map above).
General measures:
- Safe water and sanitation
- Hand hygiene
- Food safety (avoid raw fruits/vegetables, street food, ice in endemic areas)
- Identification and treatment of chronic carriers
Summary
| Feature | Detail |
|---|---|
| Causative organism | S. Typhi, S. Paratyphi A/B/C |
| Reservoir | Humans only |
| Transmission | Fecal-oral (water/food) |
| Incubation | 5–21 days |
| Classic triad | Prolonged fever + relative bradycardia + rose spots |
| Key complication | Intestinal perforation (week 3–4) |
| Diagnosis gold standard | Blood/bone marrow culture |
| Treatment (uncomplicated, sensitive) | Ciprofloxacin 7–14 days |
| Treatment (XDR) | Azithromycin or meropenem |
| Prevention | TCV (preferred), ViPS, Ty21a; WASH |
Sources: Harrison's Principles of Internal Medicine 22E (2025); Goldman-Cecil Medicine; Robbins & Kumar Pathologic Basis of Disease; Medical Microbiology 9e
All
Asking for Preferences
Reading File
Reading File
Reading File
Reading File
Searching PubMed
"typhoid fever" AND treatment
Reading File
Reading File
Reading File
Searching Images
typhoid pathogenesis Salmonella typhi Peyer patches ileum perforation
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I now have comprehensive, in-depth content from multiple authoritative textbooks. Let me compile the full detailed response.
Enteric Fever — Comprehensive Review
1. Definition & Nomenclature
Enteric fever is a severe systemic illness caused by Salmonella enterica serotype Typhi (S. Typhi) or Paratyphi (A, B, C). The name "typhoid" derives from the Greek typhos ("stupor" or "cloudy"), reflecting the neurological state of severely ill patients. The term "enteric fever" was proposed in 1869 to distinguish it from typhus based on its anatomical site (intestinal lymphoid tissue).
Critically, this is not truly an intestinal disease — it is a systemic, bacteremic illness with intestinal entry. S. Typhi and S. Paratyphi A rank as the 2nd and 3rd most common pathogens causing extraintestinal infectious disease worldwide (behind hepatitis A).
2. Microbiology
Organism: Salmonella enterica subsp. enterica
- Full name: S. enterica subsp. enterica serovar Typhi = Salmonella Typhi
- Gram-negative, non-spore-forming, facultative anaerobic bacillus
- Humans are the ONLY natural reservoir — no animal reservoir
- Vi (virulence) capsular polysaccharide antigen is S. Typhi's most important virulence factor — unique to this serovar; protects against complement-mediated killing and phagocytosis; basis of Vi polysaccharide vaccines
- Possesses H (flagellar) and O (somatic) antigens — basis of Widal test
Paratyphoid serotypes:
- S. Paratyphi A → most common, milder disease, no animal reservoir
- S. Schotmuelleri (formerly S. Paratyphi B)
- S. Hirschfeldii (formerly S. Paratyphi C)
3. Epidemiology
| Parameter | Data |
|---|---|
| Annual typhoid cases | 9.2–21 million |
| Annual paratyphoid cases | ~5 million |
| Annual deaths | 110,000–280,000 |
| Highest burden | Indian subcontinent, Eastern Mediterranean, sub-Saharan Africa |
| Endemic threshold | >1000 cases/100,000 children in some urban areas (Pakistan, Bangladesh) |
Risk factors:
- Fecally contaminated drinking water or ice
- Street food/beverages; raw fruits and vegetables grown in sewage-fertilized fields
- Flooding; lack of hand washing and toilet access
- Ill household contacts
- H. pylori infection (reduced gastric acid → lower infectious dose needed)
- Visiting friends and family in endemic regions
Transmission routes:
- Fecal-oral (food/water contamination by carriers or active cases)
- Sexual transmission (male-to-male — described)
- Healthcare worker occupational exposure (laboratory specimens)
4. Pathogenesis
Step-by-step mechanism:
Ingestion of contaminated food/water
↓
Gastric acid barrier overcome (Vi antigen helps resistance)
↓
Attachment to M cells overlying Peyer's patches (terminal ileum)
↓
Transcytosis across epithelium → taken up by subepithelial macrophages
↓
Intracellular survival in macrophages (resist phagolysosomal killing)
↓
Transport via lymphatics to mesenteric lymph nodes
↓
PRIMARY BACTEREMIA (clinically silent, ~Week 1)
↓
Dissemination to liver, spleen, bone marrow, gallbladder
↓
Replication in reticuloendothelial system (RES)
↓
SECONDARY BACTEREMIA (symptomatic — fever, clinical illness)
↓
Gallbladder colonization → reinfection of intestinal Peyer's patches
↓
Weeks 3–4: Hyperplasia → ulceration → NECROSIS of Peyer's patches
↓
Intestinal hemorrhage or PERFORATION
Key virulence mechanisms:
- Vi antigen (polysaccharide capsule): Inhibits complement activation and phagocytosis; unique to S. Typhi; confers resistance to serum killing
- Type III secretion systems (T3SS): Two distinct systems (SPI-1 and SPI-2) mediate invasion of epithelial cells and intracellular survival in macrophages respectively
- Intracellular survival: S. Typhi replicates within a Salmonella-containing vacuole (SCV), evading lysosomal fusion
Pathologic anatomy (Robbins):
- Peyer's patches: Enlarge to plateau-like elevations up to 8 cm → neutrophilic infiltrate + macrophages containing bacteria, red cells, nuclear debris → oval ulcers oriented longitudinally along the ileal axis → perforation risk
- Mesenteric lymph nodes: Enlarged
- Spleen: Enlarged, soft, pale red pulp, obliterated follicular markings, prominent phagocyte hyperplasia
- Liver: Small, randomly scattered foci of parenchymal necrosis — "typhoid nodules" (macrophage aggregates replacing hepatocytes); also in bone marrow and lymph nodes
5. Clinical Course
Incubation period: 5–21 days (mean 10–14 days); longer with smaller inoculum, vaccination, or prior immunity
Week-by-Week Progression (Untreated)
| Week | Pathology | Clinical Features |
|---|---|---|
| Week 1 | Primary bacteremia; initial bacterial seeding | Stepwise rising fever; headache (80%); myalgias, malaise; dry cough (30%); relative bradycardia (<50%); constipation more than diarrhea |
| Week 2 | Secondary bacteremia; Peyer's patch hyperplasia | Sustained fever 39.4–40.5°C; rose spots (~30%); hepatosplenomegaly; abdominal distension; diarrhea or constipation; apathy, confusion ("muttering delirium"); coated tongue (51–56%) |
| Week 3 | Necrosis of Peyer's patches begins | High fever persists; abdominal pain worsens; risk of intestinal hemorrhage (~6%) |
| Week 4 | Ulcers deepen | Risk of perforation (~1%); neuropsychiatric manifestations; resolution begins in uncomplicated cases |
Symptoms by Frequency (Prospective study, Kathmandu)
| Symptom | Frequency |
|---|---|
| Headache | 80% |
| Anorexia | 55% |
| Coated tongue | 51–56% |
| Chills | 35–45% |
| Abdominal pain | 30–40% |
| Cough | 30% |
| Diarrhea | 22–28% |
| Nausea | 18–24% |
| Vomiting | 18% |
| Sweating | 20–25% |
| Constipation | 13–16% |
| Myalgias | 20% |
Hallmark Clinical Signs
1. Fever
- Sustained, 38.8–40.5°C (101.8–104.9°F)
- Documented at presentation in >75% of cases
- Can last 4 weeks untreated
2. Relative Bradycardia (Faget's Sign)
- Pulse inappropriately slow relative to fever height
- Present in <50% of patients
- Mechanism: direct effect of bacteremia/endotoxemia on cardiac conduction
3. Rose Spots
- Faint, salmon-colored, blanching maculopapular lesions, 1–4 mm
- Located primarily on trunk and chest
- Present in ~30% of patients (end of Week 1 – Week 2)
- May show 2–3 crops; resolve without scarring in 2–5 days
- Salmonella can be cultured from punch biopsy
- Difficult to detect in dark-skinned patients

4. Hepatosplenomegaly
- ~50% of patients
- Splenomegaly develops by Week 2
5. "Typhoidal State" (Typhoid Encephalopathy)
- Apathy, confusion, delirium ("muttering delirium"), "coma vigil" (patient appears unconscious but eyes remain open with picking at bedclothes/imaginary objects)
- Neuropsychiatric manifestations in 2–40% of patients
6. Complications
Occur in ~27% of hospitalized patients. Risk correlates with duration before hospitalization, host factors (immunosuppression, acid suppression), strain virulence, and antibiotic choice.
Major Life-Threatening Complications
| Complication | Frequency | Timing | Management |
|---|---|---|---|
| Intestinal hemorrhage | ~6% | Weeks 3–4 | Fluid resuscitation, blood transfusion, broadened antibiotics; surgical bowel resection if severe |
| Intestinal perforation | ~1% | Weeks 3–4 | Emergency surgery: resection + peritoneal lavage + broad-spectrum antibiotics for polymicrobial peritonitis |
| Typhoid encephalopathy/shock | 2–40% (neuro) | Weeks 2–4 | IV dexamethasone (3 mg/kg loading, then 1 mg/kg q6h × 48h) + antibiotics |
Other Complications
| System | Complications |
|---|---|
| Neurological | Meningitis, Guillain-Barré syndrome, neuritis, seizures, deafness, psychosis, ataxia |
| Cardiovascular | Myocarditis, endocarditis, pericarditis, mycotic aneurysm |
| Hepatobiliary | Hepatitis, cholecystitis (gallbladder perforation — rare), hepatic abscess |
| Pulmonary | Pneumonia (dry cough common early; rarely cultures positive) |
| Hematological | DIC, hemophagocytic lymphohistiocytosis (HLH), hemolytic anemia |
| Renal | Glomerulonephritis, pyelonephritis, hemolytic-uremic syndrome |
| Musculoskeletal | Osteomyelitis (especially in sickle cell disease), septic arthritis |
| Other | Orchitis, parotitis, pancreatitis, endophthalmitis, splenic abscess |
⚠️ HLH as a complication: A 2024 systematic review (PMID 38579699) characterizes enteric fever-associated HLH — a rare but life-threatening complication with fever, cytopenia, splenomegaly, and hyperferritinemia. Early recognition is critical.
Relapse
- Up to 10% of patients; typically 2–3 weeks after fever resolution
- Same organism and susceptibility profile
- Usually milder than primary episode
- Relapse rates lower with azithromycin than with fluoroquinolones or ceftriaxone
Chronic Carrier State
- 2–5% of untreated patients → chronic carriage (>1 year excretion in stool or urine)
- Gallbladder is the reservoir (biofilm formation on gallstones; invasion of gallbladder epithelium)
- Up to 10% excrete S. Typhi in feces for up to 3 months after acute illness
- Risk factors for chronic carriage: Women, infants, biliary abnormalities, Schistosoma haematobium co-infection
- Associated with gallbladder carcinoma (much higher incidence where S. Typhi is endemic)
7. Diagnosis
Gold Standard: Culture
| Specimen | Sensitivity | Optimal Timing | Notes |
|---|---|---|---|
| Bone marrow culture | ~80–95% (highest) | Any time, even post-antibiotics | Invasive; remains positive despite prior antibiotics |
| Blood culture | 60–80% | Weeks 1–2 (bacteremic phase) | Most practical; yield declines with antibiotics or after Week 2 |
| Stool culture | 30–40% | From Week 2 onwards | Negative early; positive in carriers |
| Urine culture | 25–30% | Weeks 2–3 | Lower yield |
| Rose spot biopsy | Variable | When rash present | Culturable when rose spots visible |
| Intestinal secretions (string test) | ~60–70% | Any time | Via duodenal fluid; useful but rarely performed |
Lab comparison of Salmonella clinical syndromes (Jawetz):
| Feature | Enteric Fever | Bacteremia | Enterocolitis |
|---|---|---|---|
| Incubation | 7–20 days | Variable | 8–48 hours |
| Onset | Insidious | Abrupt | Abrupt |
| Fever pattern | Stepwise then plateau | Spiking septic | Usually low |
| Duration | Several weeks | Variable | 2–5 days |
| Blood culture | Positive Weeks 1–2 | Positive during fever | Negative |
| Stool culture | Positive from Week 2 | Infrequently positive | Positive early |
Non-Specific Laboratory Findings
- Leukopenia and neutropenia (15–25%) — contrasts with expected leukocytosis in sepsis
- Leukocytosis — more common in children, first 10 days, or with perforation/secondary infection
- Moderately elevated liver enzymes (ALT, AST)
- Mild thrombocytopenia
- Elevated CRP/ESR (nonspecific)
- Anemia (from GI losses or hemolysis)
Serological Tests
Widal Test (Widal agglutination):
- Detects antibodies to S. Typhi H (flagellar) and O (somatic) antigens
- Diagnostic titer: O agglutinin ≥1:160 or H agglutinin ≥1:160 (varies by region and baseline titers)
- Problems:
- Cross-reaction with other Salmonella strains → false positives
- False negatives if collected too early (first week)
- High background titers in endemic populations reduce specificity
- Prior vaccination raises H titers → false positive
- No standardization between laboratories
- Conclusion: Widal test alone is unreliable and should NOT be the sole basis for diagnosis. Culture remains essential.
Newer rapid tests:
- Typhidot (IgM/IgG against 50-kDa outer membrane protein): Better sensitivity in early disease
- TPTest, Tubex: Detect anti-O9 IgM
- Variable and suboptimal performance in endemic areas
- PCR assays: Increasing use in reference laboratories; not widely available
8. Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Malaria | Must exclude first in any febrile traveler; thick/thin smear, RDT |
| Viral hepatitis | Jaundice prominent; elevated bilirubin; serologies |
| Dengue fever | Thrombocytopenia, myalgia, rash (different morphology), arthralgia |
| Rickettsial infections | Eschar, different rash distribution, serology |
| Leptospirosis | Conjunctival suffusion, jaundice, renal failure; exposure history |
| Amebic liver abscess | Focal liver lesion on imaging; Entamoeba serology/PCR |
| Brucellosis | Animal exposure; night sweats; arthralgia/spondylitis |
| Acute HIV | Pharyngitis, lymphadenopathy; HIV serology |
| Bacterial endocarditis | Murmur, embolic phenomena; echocardiography |
| Appendicitis | Right iliac fossa pain; no high fever at onset; CT |
9. Treatment
Overall mortality: 2.5% overall; 4.5% among hospitalized patients; 10–30% if untreated → drops to <1% with prompt appropriate antibiotics.
Antibiotic Therapy (Harrison's 22E, Table 171-1)
| Indication | Agent | Dose & Route | Duration |
|---|---|---|---|
| Empirical (pending susceptibility) | Ceftriaxone | 2 g/day IV | 10–14 days |
| Ciprofloxacin | 500 mg BD PO or 400 mg q12h IV | 5–7 days | |
| Azithromycin | 1 g/day PO | 10 days | |
| Fully susceptible | Ceftriaxone | 2 g/day IV | 10–14 days |
| (optimal) | Ciprofloxacin | 500 mg BD PO | 5–7 days |
| Fully susceptible | Azithromycin | 1 g/day PO | 5 days |
| (alternative) | Amoxicillin | 1 g TID PO or 2 g q6h IV | 14 days |
| Chloramphenicol | 25 mg/kg TID PO/IV | 14–21 days | |
| Trimethoprim-sulfamethoxazole | 160/800 mg BD PO | 7–14 days | |
| MDR (resistant to chloramphenicol, ampicillin, TMP-SMX) | Ceftriaxone | 2 g/day IV | 10–14 days |
| Ciprofloxacin | 500 mg BD PO | 5–7 days | |
| Azithromycin | 1 g/day PO | 5 days | |
| Ceftriaxone-resistant / XDR | Azithromycin | 1 g/day PO | 5–7 days |
| Meropenem | Standard dosing IV | 10–14 days |
⚠️ Fluoroquinolone caution: Due to high prevalence of decreased susceptibility to ciprofloxacin (DSC; MIC ≥0.125 µg/mL) on the Indian subcontinent and parts of Africa, fluoroquinolones should no longer be used as empirical first-line therapy for travel-acquired enteric fever from these regions. Use ceftriaxone or azithromycin empirically.
⚠️ XDR typhoid (Pakistan, 2016–present): Resistant to chloramphenicol, ampicillin, TMP-SMX, fluoroquinolones, AND 3rd-generation cephalosporins. Only susceptible to azithromycin (uncomplicated) and carbapenems (severe/complicated). >5000 cases documented; exported to UK, US, and multiple countries.
📌 2024 Evidence (PMID 39623850): Systematic review of RCTs confirms oral azithromycin is clinically equivalent to other antimicrobials for typhoid fever across all age groups, validating its central role especially for XDR typhoid.
Adjunctive Therapy
Dexamethasone (corticosteroids):
- For severe disease with delirium, obtundation, stupor, coma, or shock
- Regimen: IV dexamethasone 3 mg/kg loading dose, then 1 mg/kg q6h × 48 hours
- Shown to reduce mortality in severe typhoid (landmark RCT, Hoffman et al., NEJM)
- Should be reserved for critically ill patients only
Supportive care:
- IV fluid resuscitation and electrolyte correction
- Blood transfusion if significant GI hemorrhage
- NG tube decompression for ileus
Surgical management (perforation/hemorrhage):
- Emergency laparotomy: bowel resection/repair + peritoneal lavage
- Broadened antibiotics to cover polymicrobial peritonitis
Chronic Carrier Treatment
- Oral fluoroquinolone (ciprofloxacin or norfloxacin) × 4–6 weeks — effective because fluoroquinolones are highly concentrated in bile
- Alternative: High-dose parenteral ampicillin (if susceptible and fluoroquinolone not tolerated)
- Cholecystectomy if antibiotic therapy fails (especially with gallstones serving as biofilm substrate), followed by another antibiotic course
10. Prevention
Vaccines
| Vaccine | Type | Route | Age | Efficacy | Schedule |
|---|---|---|---|---|---|
| Ty21a (Vivotif) | Live attenuated oral | Oral | ≥6 years | ~50% at 2.5–3 years | 4 doses (Days 1, 3, 5, 7); booster every 5 years |
| Vi CPS (Typhim Vi) | Vi polysaccharide | IM | ≥2 years | ~55% at 3 years | Single dose; booster every 2 years |
| Typbar-TCV | Vi conjugated to tetanus toxoid | IM | ≥6 months | 79–95%; antibody persists up to 7 years | Single 0.5-mL IM dose; WHO-recommended |
| TYPHIBEV | Vi conjugated | IM | ≥6 months | ~79–95% | Single dose; WHO-recommended (2020) |
Key vaccine points:
- Unconjugated vaccines (Ty21a, Vi CPS) are poorly immunogenic in children <2 years — cannot elicit T cell-dependent memory
- TCV (conjugate vaccines) are effective from 6 months, elicit T-cell dependent immunity, and are WHO-recommended for routine childhood immunization in high-incidence countries
- Pakistan introduced TCV nationally in November 2019 — first country to do so
- No licensed vaccine exists for paratyphoid fever
- Vaccine protective efficacy can be overcome by high inocula (e.g., large food-borne exposures) — vaccines are an adjunct, not a substitute for food/water precautions
Travel Recommendations
- Vaccination recommended for travel to Southern Asia, Africa, the Caribbean, Central and South America
- Even short travel (<2 weeks) to high-risk areas warrants vaccination
- Laboratory workers exposed to S. Typhi should be vaccinated
- Household contacts of known carriers should be vaccinated
Public Health Measures
- Safe drinking water and sanitation (WASH — Water, Sanitation, Hygiene)
- Food handler screening and exclusion during illness
- Notifiable disease in the US — reporting enables identification of source patients and chronic carriers
- Contact precautions during hospitalization for diapered/incontinent patients (stool precautions until 3 consecutive negative stool cultures post-antibiotics)
- XDR typhoid → full contact precautions as for MDR organisms
WASH Evidence
📌 2023 Meta-analysis (PMID 37644449): Water, sanitation, and hygiene interventions significantly reduce typhoid fever risk, confirming WASH as a cornerstone of prevention alongside vaccination.
11. Drug Resistance Timeline
| Era | Development |
|---|---|
| Pre-antibiotics | Mortality 10–15%; chloramphenicol era began 1948 |
| 1980s | MDR strains (plasmid-mediated): resistant to chloramphenicol, ampicillin, TMP-SMX; emerged in China and Southeast Asia |
| 1990s–2000s | Fluoroquinolone-treated MDR → DSC (decreased susceptibility, MIC ≥0.125) and frank quinolone-resistant strains; Clone H58 dominant |
| 2016–present | XDR S. Typhi in Sindh, Pakistan: adds fluoroquinolone AND ceftriaxone resistance to MDR profile; only azithromycin and carbapenems active |
| 2026 | 📌 (PMID 41990105): Meta-analysis of India isolates 1977–2024 confirms rising burden of AMR, with MDR rates varying from 55% in some periods to declining trends post-TCV introduction |
12. Summary: High-Yield Points
| Feature | Key Fact |
|---|---|
| Causative organisms | S. Typhi (most), S. Paratyphi A/B/C |
| Only reservoir | Humans |
| Entry mechanism | M cells over Peyer's patches (terminal ileum) |
| Incubation period | 5–21 days (mean 10–14 days) |
| Classic triad | Prolonged fever + relative bradycardia + rose spots |
| Rose spots | Faint, salmon-colored, blanching maculopapular on trunk; ~30% of patients |
| Key pathological finding | Enlarged Peyer's patches → longitudinal ulcers → perforation; typhoid nodules in liver |
| Hallmark lab finding | Leukopenia with neutropenia (paradoxical in bacteremia) |
| Gold standard diagnosis | Bone marrow culture (most sensitive); blood culture (most practical) |
| Widal test verdict | Unreliable; not diagnostic alone |
| Worst complications | Intestinal perforation (Weeks 3–4); typhoid encephalopathy |
| Chronic carriage | 2–5%; gallbladder reservoir; associated with gallbladder cancer |
| Treatment empirical (high-resistance area) | Ceftriaxone IV or azithromycin PO |
| XDR treatment | Azithromycin (mild) or meropenem (severe) |
| Severe disease adjunct | Dexamethasone 3 mg/kg loading → 1 mg/kg q6h × 48h |
| Best vaccine | TCV (typhoid conjugate vaccine) — 79–95% efficacy; from age 6 months |
| No vaccine available for | S. Paratyphi (paratyphoid fever) |
Sources: Harrison's Principles of Internal Medicine 22E (2025, McGraw Hill); Goldman-Cecil Medicine; Robbins & Kumar Pathologic Basis of Disease; Sleisenger & Fordtran's GI and Liver Disease; Medical Microbiology 9E; Jawetz Melnick & Adelberg's Medical Microbiology 28E; Tintinalli's Emergency Medicine; Red Book 2021 (AAP); PubMed PMIDs: 39623850, 38579699, 37644449, 41990105
This is a shared conversation. Sign in to Orris to start your own chat.