Hanta virus all details and management with drugs dose
Reading File
Finding Sources
Finding Sources
Searching PubMed
hantavirus treatment management ribavirin
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Searching the Web
hantavirus HPS HFRS treatment ribavirin dosing guidelines 2024 2025
Reading File
Reading Web Page
hantavirus ribavirin dosing treatment HFRS HPS management
Loading Image
Finding Sources
I have comprehensive information from multiple authoritative sources. Now I'll compile the full, detailed answer.
Hantavirus — Complete Review
1. Virology & Classification
| Feature | Detail |
|---|---|
| Family | Hantaviridae (formerly Bunyaviridae) |
| Type | Negative-sense, single-stranded RNA, enveloped, helical |
| Genome | Tripartite (S, M, L segments) |
| Transmission | Non-arthropod-borne zoonosis — unique among bunyaviruses |
| Notable | Only bunyavirus transmitted WITHOUT an arthropod vector |
- Sherris & Ryan's Medical Microbiology, 8th Ed.
2. Clinically Important Strains
Old World (Hemorrhagic Fever with Renal Syndrome — HFRS)
| Virus | Region | Host Rodent | Severity |
|---|---|---|---|
| Hantaan | Eastern Asia (China, Korea, Russia) | Apodemus spp. | Severe (CFR 5–15%) |
| Dobrava | Balkans, Eastern Europe | Apodemus flavicollis | Severe |
| Seoul | Worldwide (Norway rats) | Rattus norvegicus | Moderate |
| Puumala | Scandinavia, W. Europe | Bank vole | Mild–moderate |
| Saaremaa | Central Europe | Apodemus agrarius | Mild |
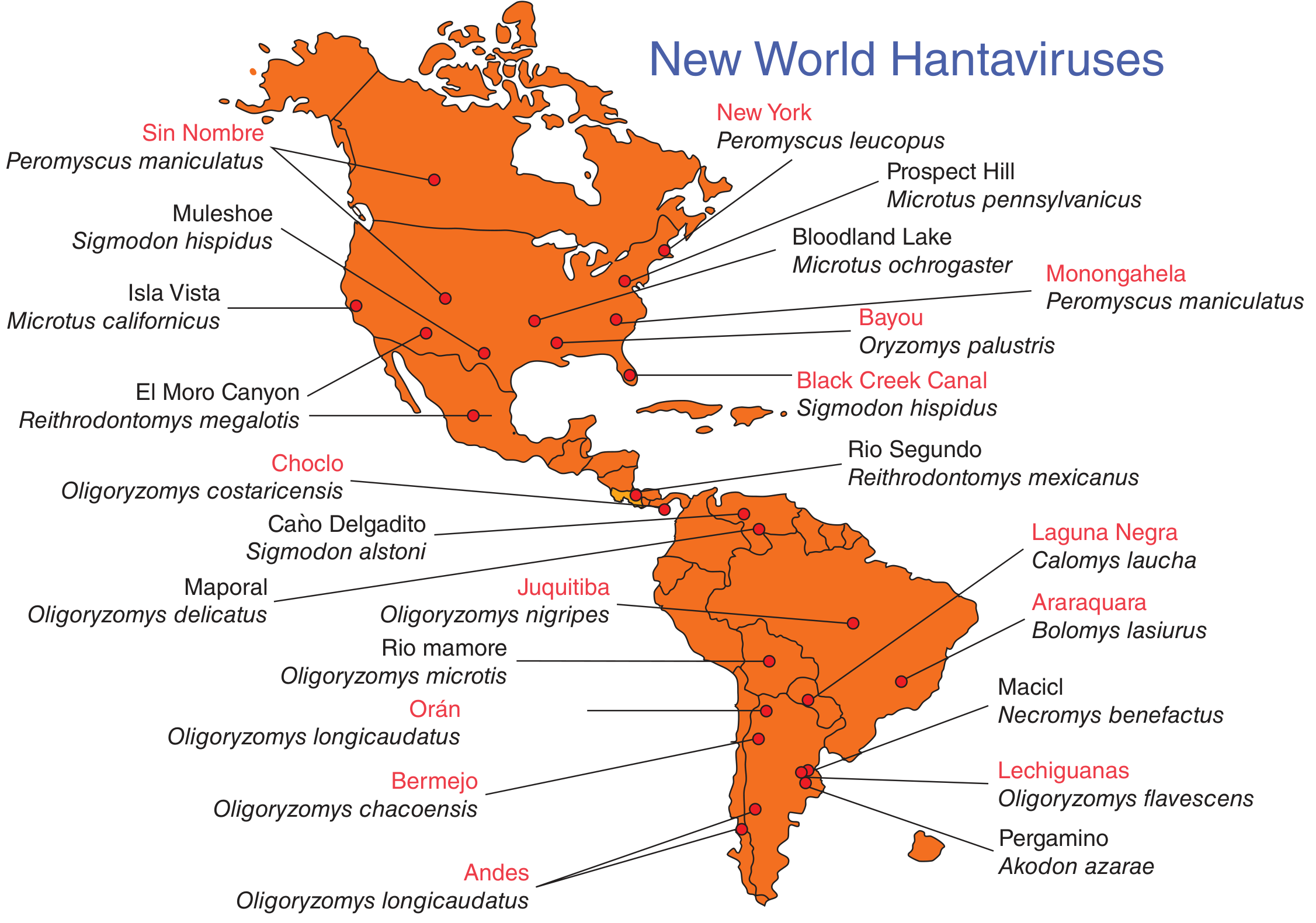
New World (Hantavirus Pulmonary Syndrome — HPS / HCPS)
| Virus | Region | Host Rodent | CFR |
|---|---|---|---|
| Sin Nombre | Western/Central USA | Deer mouse (P. maniculatus) | 30–40% |
| Andes | Argentina, Chile | Oligoryzomys longicaudatus | ~25–35% |
| Bayou | Louisiana, Texas | Rice rat (Oryzomys palustris) | Moderate |
| Black Creek Canal | Florida | Cotton rat (Sigmodon hispidus) | Moderate |
| New York / Monongahela | Eastern USA | White-footed mouse (P. leucopus) | Lower |
| Choclo | Panama | Oligoryzomys costaricensis | — |
Key fact: Andes virus is the ONLY hantavirus with confirmed person-to-person transmission (reported in Chile/Argentina). All others transmit exclusively from rodent to human.
- Red Book 2021; Sherris & Ryan's 8th Ed.
3. Epidemiology & Transmission
Routes of human infection:
- Inhalation of aerosolized rodent urine, feces, or saliva (primary route)
- Direct contact with infected rodents or their excreta
- Rodent bite (rare)
- Contamination of broken skin
High-risk activities:
- Cleaning/entering closed, rodent-infested structures
- Agricultural work (hand plowing, harvesting)
- Camping/backpacking in rodent-infested areas (e.g., 2012 Yosemite National Park outbreak)
- Livestock/feed storage work
USA epidemiology: ~20–40 HPS cases/year; >95% west of Mississippi River; >800 total cases since 1993; 32 states affected. Disproportionate burden in American Indian/Alaska Native populations (CFR 46% vs. general population).
- Red Book 2021
4. Pathophysiology
The central mechanism is increased vascular permeability via functional impairment of vascular endothelium:
- Hantavirus infects capillary endothelial cells (detected by immunohistochemistry in lungs, heart, spleen, lymph nodes)
- CD8+ T-cell immune response → cytokine storm → capillary leak
- HPS: Pulmonary capillary leak → noncardiogenic pulmonary edema; also severe myocardial depression with low cardiac output (distinct from septic shock)
- HFRS: Vascular leak in kidneys + hemorrhagic manifestations → acute renal failure
- Jawetz Medical Microbiology, 28th Ed.; Murray & Nadel's Respiratory Medicine
5. Clinical Syndromes
5A. Hantavirus Pulmonary Syndrome (HPS / HCPS)
Incubation period: 1–6 weeks (typical 2–3 weeks)
Phase 1 — Prodromal (3–7 days)
- Fever, chills, severe myalgias, headache
- Nausea, vomiting, diarrhea, dizziness
- Absence of cough (distinguishing feature early)
- Abdominal pain common
Phase 2 — Cardiopulmonary (hours to days after prodrome)
- Abrupt onset of nonproductive cough + dyspnea → rapid respiratory failure
- Noncardiogenic pulmonary edema
- Bilateral interstitial/alveolar infiltrates + pleural effusions
- Severe hypoxemia requiring mechanical ventilation
- Myocardial depression: ↓ cardiac index, ↓ stroke volume, ↑ systemic vascular resistance, normal pulmonary wedge pressure
- Hypotension, tachycardia, lactic acidosis, metabolic acidosis
Poor prognostic indicators:
- Persistent hypotension
- Marked hemoconcentration
- Cardiac index < 2 L/min/m²
- Serum lactate > 4 mmol/L (36 mg/dL)
- Abrupt lactic acidosis onset
Mortality: 30–40% (initially ~60% in 1993 outbreak, improved with aggressive ICU care)
- Red Book 2021; Murray & Nadel's Respiratory Medicine
5B. Hemorrhagic Fever with Renal Syndrome (HFRS)
Incubation: 1–2 weeks (up to 8 weeks)
Classic 5 clinical phases (particularly Hantaan/Dobrava):
| Phase | Duration | Features |
|---|---|---|
| Febrile | 3–7 days | Fever, headache, back/abdominal pain, facial flushing, conjunctival injection, petechiae |
| Hypotensive | Hours–2 days | Hypotension, shock, vascular leakage |
| Oliguric | 3–7 days | Acute renal failure, hypertension, fluid overload, hemorrhage |
| Diuretic | Days–weeks | Massive diuresis, electrolyte imbalances |
| Convalescent | Weeks–months | Gradual recovery |
Severity by strain: Hantaan/Dobrava (severe, CFR 5–15%) > Seoul/Puumala (moderate–mild, CFR <1%)
- Sherris & Ryan's Medical Microbiology, 8th Ed.
6. Laboratory Findings
Characteristic Blood Picture (HPS)
The 5-point peripheral blood screen (positive predictive value >90% if 4/5 met):
- Hemoglobin elevated for age/sex (hemoconcentration)
- Left shift of granulocytic series
- Absence of toxic changes
- Thrombocytopenia
- Immunoblasts/plasma cells >10% of lymphocytes (basophilic cytoplasm, prominent nucleoli)
Additional labs:
- Neutrophilic leukocytosis with immature granulocytes
- Mildly elevated LFTs
- Elevated hematocrit
- Decreased serum bicarbonate
- Mild acute kidney injury (HPS); severe renal failure (HFRS)
Diagnosis (Confirmatory)
| Test | Notes |
|---|---|
| Serology (IgM + IgG) | Method of choice; IgM + IgG usually positive at symptom onset; IgG may be negative in rapidly fatal cases |
| RT-PCR (viral RNA) | Detectable in blood first 10 days; in peripheral blood mononuclear cells |
| Immunohistochemistry | Capillary endothelial cells — useful at autopsy |
| Viral culture | NOT useful (slow, requires BSL-3/4 containment) |
Commercial labs offer Hantavirus serology, but IgM positives are referred to CDC Viral Special Pathogens Branch for confirmation. Clinicians can call 470-312-0094 for assistance.
- Red Book 2021; Murray & Nadel's Respiratory Medicine
7. Management
7A. General Approach
All patients with suspected HPS should be immediately transferred to a tertiary care/ICU facility.
| Priority | Action |
|---|---|
| Airway | Early intubation if deteriorating oxygenation |
| Circulation | Inotropes/vasopressors for myocardial depression |
| Fluids | Careful fluid management — maintain perfusion WITHOUT exacerbating pulmonary edema |
| ECMO | Consider early when pulmonary wedge pressure and cardiac indices have deteriorated |
7B. Respiratory Support
- Mechanical ventilation usually required for only 2–4 days in survivors
- Recovery heralded by onset of diuresis (watch for this)
- Low-tidal volume ventilation (lung-protective) principles apply
- ECMO has been successful in severe cases and should be considered early (Harrison's 22E; Red Book 2021)
7C. Hemodynamic Support
- Vasopressors/inotropes for cardiovascular collapse
- High-dose methylprednisolone — randomized trial showed NO benefit in HPS
- Cytokine-blocking agents — theoretical role, not systematically evaluated
7D. Antimicrobials
- Broad-spectrum antibiotics are commonly administered while awaiting confirmatory diagnosis (bacterial septic shock is far more common)
- No specific antimicrobial therapy proven effective for HPS
- Red Book 2021
7E. HFRS-Specific Supportive Care
- Fluid and electrolyte balance
- Dialysis if required for fluid overload/renal failure
- Close monitoring of diuretic phase (electrolyte losses)
8. Antiviral Drug Therapy — Ribavirin
Ribavirin in HFRS ✅ (Benefit shown)
IV Ribavirin Protocol (HFRS — China RCT, 242 patients):
| Phase | Dose | Duration |
|---|---|---|
| Loading dose | 33 mg/kg IV (single dose) | Day 1 |
| Maintenance 1 | 16 mg/kg IV every 6 hours | Days 1–4 (4 days) |
| Maintenance 2 | 8 mg/kg IV every 8 hours | Days 5–7 (3 days) |
| Total duration | 7 days |
Outcome: Mortality reduced 7-fold among ribavirin-treated HFRS patients.
Key requirement: Must be initiated EARLY in the course of infection for benefit.
One open-label Russian trial showed insufficient efficacy for Puumala-caused HFRS — benefit may be strain-dependent.
Ribavirin in HPS ❌ (No proven benefit)
- Ribavirin is active in vitro against hantaviruses including Sin Nombre virus
- Two clinical trials (1 open-label + 1 randomized double-blind placebo-controlled) showed NO survival benefit in the cardiopulmonary phase of HPS
- Ribavirin is NOT the standard of care for HPS
- Animal models (hamsters, Andes virus) showed protection — but this did not translate to human HPS trials
- Red Book 2021; Murray & Nadel's Respiratory Medicine; PMC7594967
Summary Table
| Syndrome | Ribavirin Use | Evidence |
|---|---|---|
| HFRS | Recommended if early | RCT positive (China); 7-fold mortality reduction |
| HPS (cardiopulmonary stage) | NOT recommended | Two RCTs/trials: no benefit |
9. Isolation Precautions
- Standard precautions for hospitalized HPS patients (USA)
- Contact + Droplet precautions for Andes virus infection (person-to-person transmission possible)
- HPS and nonpulmonary hantavirus infections are nationally notifiable diseases in the USA (reportable to state/local public health)
10. Prevention & Control
| Measure | Details |
|---|---|
| Rodent control | Primary prevention — reduce rodent density near homes/work |
| Safe cleaning | Wet mop/spray rodent droppings with disinfectant before disturbing; avoid dry sweeping |
| Ventilate enclosed spaces | Open windows/doors 30 min before cleaning rodent-infested areas |
| PPE | N95 mask, gloves when cleaning infested areas |
| Camping precautions | Do not sleep on bare ground; use tents with floors |
| Surveillance | Avoid areas with unusually high rodent populations |
| Vaccine | No approved vaccine currently available (Old World vaccines used in China/Korea, experimental only elsewhere) |
11. Quick Comparison: HPS vs HFRS
| Feature | HPS (New World) | HFRS (Old World) |
|---|---|---|
| Main viruses | Sin Nombre, Andes | Hantaan, Dobrava, Seoul, Puumala |
| Geographic region | Americas | Asia, Europe |
| Primary organ involved | Lungs | Kidneys |
| Hemorrhage | Absent | Present |
| Renal failure | Mild/absent | Prominent |
| Person-to-person | Only Andes virus | No |
| Mortality | 30–40% (severe) | 5–15% (Hantaan); <1% (Seoul/Puumala) |
| Ribavirin benefit | No | Yes (if early) |
| Diagnosis | IgM/IgG + RT-PCR | IgM/IgG + RT-PCR |
Sources
- Harrison's Principles of Internal Medicine, 22nd Ed. (2025) — McGraw Hill
- Red Book 2021: Report of the Committee on Infectious Diseases — AAP
- Sherris & Ryan's Medical Microbiology, 8th Ed.
- Jawetz, Melnick & Adelberg's Medical Microbiology, 28th Ed.
- Murray & Nadel's Textbook of Respiratory Medicine
- Tintinalli's Emergency Medicine: A Comprehensive Study
- Huggins JW et al. — IV Ribavirin RCT in HFRS (China, 242 patients), PMC7594967
This is a shared conversation. Sign in to Orris to start your own chat.