Is carbon dioxide a simple asphyxiant or chemical asphyxiant as increase in PaCO2 can also decrease the affinity of HB for O2
Reading File
Finding Sources
Finding Sources
Searching the Web
carbon dioxide simple asphyxiant vs chemical asphyxiant classification toxicology
Reading File
Finding Sources
Reading File
Reading File
Reading File
Now I have comprehensive information from multiple authoritative sources. Here is the complete answer:
CO₂: Simple Asphyxiant — With Important Caveats
The Standard Classification
CO₂ is classified as a simple asphyxiant, not a chemical asphyxiant. This is the consensus across toxicology references:
- Rosen's Emergency Medicine explicitly categorizes CO₂ as a "Simple asphyxiant; systemic effects" in its table of inhaled toxins, in direct contrast to CO (chemical asphyxiant).
- Simple asphyxiants (N₂, He, Ar, propane, CO₂) act by displacing O₂ in ambient air, reducing FiO₂ below the threshold needed to sustain life — they exert no direct toxic action on cellular respiration or hemoglobin at standard concentrations.
- Chemical asphyxiants (CO, cyanide, H₂S) are toxic even in the presence of adequate ambient O₂ because they directly interfere with O₂ transport or mitochondrial utilization.
Why Your Question Is Genuinely Nuanced
You are absolutely correct that CO₂ is not a purely inert gas like nitrogen or helium. It has direct physiological effects that blur the line:
1. The Bohr Effect (CO₂ reduces Hb–O₂ affinity)
Increasing PaCO₂ reduces hemoglobin's affinity for O₂ through two mechanisms — together constituting the Bohr effect:
Mechanism A — Indirect (pH-Bohr effect, dominant ~80% of total Bohr effect):
CO₂ + H₂O → H₂CO₃ → HCO₃⁻ + H⁺ (via carbonic anhydrase in RBCs)
The resulting drop in intracellular pH causes H⁺ to bind specific histidine residues on deoxyhemoglobin. These groups have a higher pKa in deoxyhemoglobin than in oxyhemoglobin, so protonation stabilizes the T (tense/deoxy) conformation via salt bridges, decreasing O₂ affinity and shifting the O₂-dissociation curve to the right.
"The release of O₂ from hemoglobin is enhanced when the pH is lowered or when the hemoglobin is in the presence of an increased pCO₂. Both result in decreased oxygen affinity of hemoglobin... This change in oxygen binding is called the Bohr effect." — Lippincott Biochemistry, 8th ed., p. 103–105
Mechanism B — Direct CO₂-Bohr effect (minor, ~20%):
At fixed pH (isohydric hypercapnia), CO₂ directly reacts with unprotonated terminal amino groups (Hb–NH₂) on the α and β globin chains to form carbamino compounds:
Hb–NH₂ + CO₂ → Hb–NH–COO⁻ + H⁺
This carbamino formation preferentially stabilizes the T form, independently reducing O₂ affinity. However, Medical Physiology (Boron & Boulpaep) notes this direct CO₂-Bohr effect is small compared to the pH-mediated component.
2. Other Systemic Effects of CO₂ (Beyond Simple Asphyxiation)
At high concentrations, CO₂ causes direct toxic effects that go beyond simple O₂ displacement:
- Sympathetic stimulation → tachycardia, increased cardiac output, raised pulmonary artery pressure
- CNS excitation → headache, confusion, narcosis at very high levels
- Respiratory acidosis
This is why the Ann Disaster Med. 2005 review states: "CO₂ closely resembles simple asphyxiants from a toxicological standpoint... however, has direct toxic effects," and Rosen's adds the qualifier "systemic effects" next to its simple asphyxiant label.
Why CO₂ Is Still NOT a Classical Chemical Asphyxiant
| Feature | Simple Asphyxiant (N₂) | CO₂ | Chemical Asphyxiant (CO/CN⁻) |
|---|---|---|---|
| Toxic at normal O₂ levels? | No | No | Yes |
| Direct cellular toxicity? | No | Minimal | Yes (blocks cytochrome c oxidase or Hb binding) |
| IDLH concentration | No standard | 40,000 ppm | Very low (CO: 1,200 ppm) |
| Reduces Hb-O₂ affinity? | No | Yes (Bohr effect) | CO increases affinity (left shift) |
| Mechanism of harm | O₂ dilution | O₂ dilution + acidosis + Bohr effect | Disrupts O₂ transport/utilization |
Crucially, the Bohr effect from hypercapnia actually helps O₂ delivery to tissues physiologically — it is a right-shift, which means hemoglobin offloads O₂ more readily at the tissues. This is the opposite of pathological O₂ trapping (as seen with CO, which causes a left shift and prevents O₂ release). So the Bohr effect of CO₂ is not itself a mechanism of tissue hypoxia — it is a homeostatic adaptive response.
Conclusion
CO₂ is classified as a simple asphyxiant because it does not cause toxicity at ambient O₂ concentrations. However, it is not a "pure" simple asphyxiant like argon or nitrogen — it has additional systemic and biochemical effects (the Bohr effect via H⁺ and carbamino formation, sympathetic stimulation, CNS narcosis). Some authors describe it as a simple asphyxiant with chemical properties, and Rosen's explicitly flags this with the annotation "systemic effects."
The Bohr effect it induces is a physiologically beneficial rightward shift in the O₂-dissociation curve (better O₂ unloading at tissues), not a mechanism of hypoxic injury — so it does not qualify CO₂ as a "chemical asphyxiant" in the toxicological sense.
Sources:
- Rosen's Emergency Medicine, Table 148.1
- Lippincott Illustrated Reviews: Biochemistry, 8th ed., pp. 101–106
- Medical Physiology (Boron & Boulpaep), pp. 955–957
- Murray & Nadel's Textbook of Respiratory Medicine
Explain right shift and left shift to me
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
I have everything needed for a clear, comprehensive explanation with diagrams from the textbooks.
The Oxygen-Haemoglobin Dissociation Curve (ODC) — Right & Left Shifts
The Baseline Curve
The ODC is a sigmoidal (S-shaped) curve that plots haemoglobin saturation (%) on the Y-axis against partial pressure of O₂ (PO₂, in mmHg) on the X-axis. Its sigmoid shape arises from cooperative binding — once Hb binds the first O₂ molecule, its affinity for subsequent O₂ molecules increases.
Two key reference points on the normal curve:
- PO₂ ~100 mmHg → arterial blood in lungs → Hb is ~97–98% saturated (loading)
- PO₂ ~40 mmHg → venous blood in tissues → Hb is ~75% saturated (unloading)
The P50 is the PO₂ at which Hb is exactly 50% saturated. Normal P50 = ~26–27 mmHg. It is the key indicator of whether the curve has shifted.
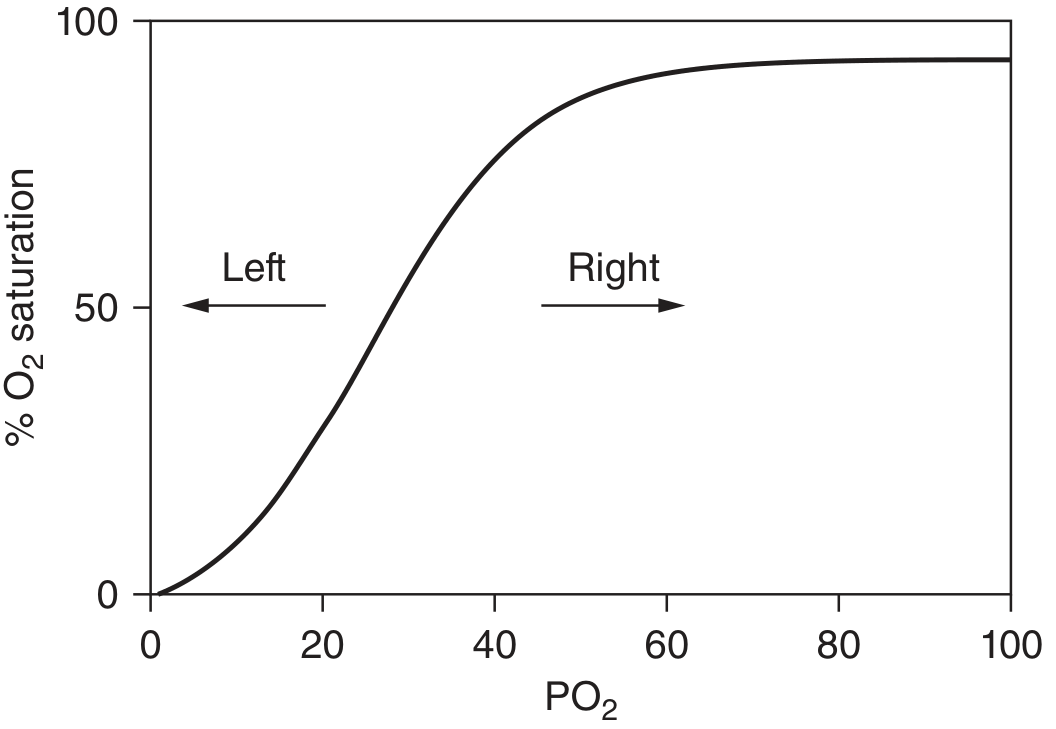
Right Shift
Definition: The curve moves to the right → P50 increases → Hb needs a higher PO₂ to achieve the same saturation.
What this means physiologically:
- Haemoglobin has lower affinity for O₂
- O₂ is more easily released ("unloaded") to tissues
- At any given PO₂, Hb holds less O₂
Think of it this way: Hb becomes a "generous giver" — it lets go of O₂ more readily.
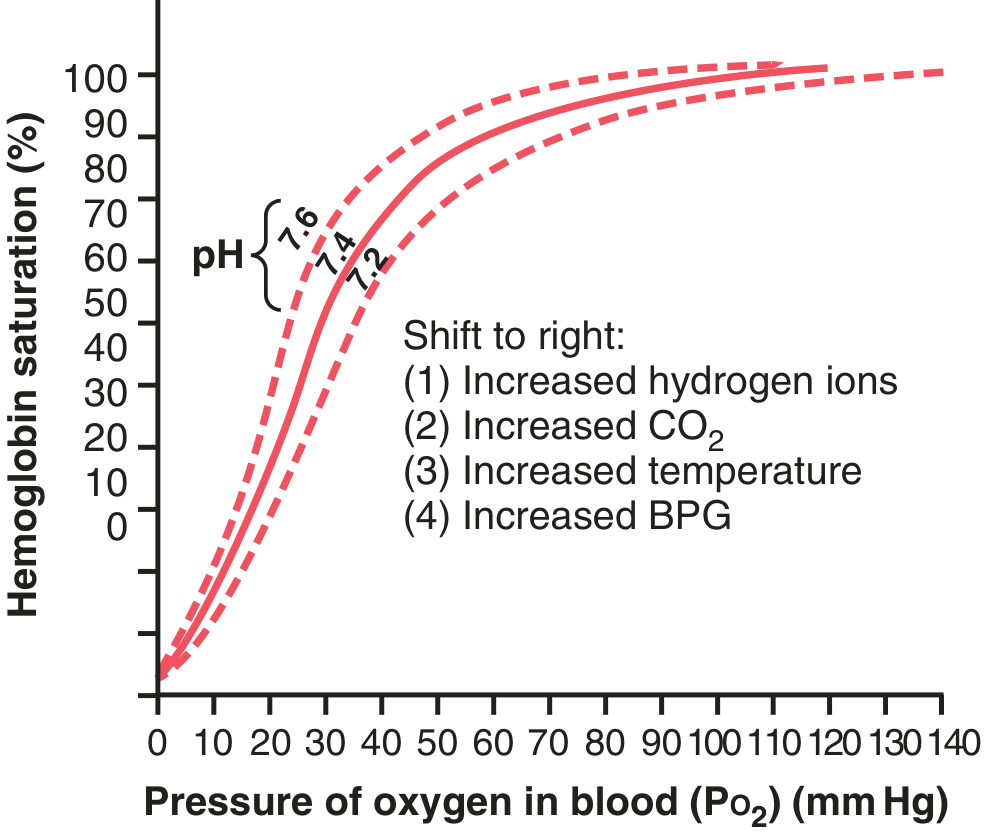
Causes — mnemonic: "CADET, face Right!"
| Factor | Direction | Physiological context |
|---|---|---|
| ↑ CO₂ (hypercapnia) | Right | Active tissues producing CO₂ |
| ↑ H⁺ / ↓ pH (acidosis) | Right | Metabolically active or ischaemic tissue |
| ↑ Temperature | Right | Exercising muscle |
| ↑ 2,3-BPG | Right | Chronic hypoxia, anaemia, high altitude |
| CO (carbon monoxide) | Left (see below) | — |
The Bohr effect (covered in your previous question) is the combination of ↑CO₂ and ↑H⁺ shifting the curve right in peripheral tissues — maximising O₂ delivery exactly where it is most needed.
"A shift of the oxygen-hemoglobin dissociation curve to the right in response to increases in blood CO₂ and H⁺ levels enhances the release of O₂ from the blood in the tissues." — Guyton & Hall, Medical Physiology
Clinical importance of right shift:
- During exercise: working muscles are hot, acidotic, and hypercapnic → all three factors maximise O₂ unloading where demand is highest.
- In anaemia: RBCs upregulate 2,3-BPG to compensate, shifting right to deliver more O₂ per unit of blood.
- At high altitude: chronic hypoxia → ↑2,3-BPG → right shift → tissues extract more O₂ from each Hb molecule.
Left Shift
Definition: The curve moves to the left → P50 decreases → Hb achieves the same saturation at a lower PO₂.
What this means physiologically:
- Haemoglobin has higher affinity for O₂
- O₂ is held more tightly and released less readily to tissues
- Hb "grabs" O₂ well in the lungs but "won't let go" at the tissues
Think of it this way: Hb becomes a "greedy hoarder" — it picks up O₂ easily but doesn't release it.
Causes:
| Factor | Physiological/Clinical context |
|---|---|
| ↓ CO₂ / ↑ pH (alkalosis) | Hyperventilation, respiratory alkalosis |
| ↓ Temperature | Hypothermia |
| ↓ 2,3-BPG | Stored bank blood (BPG degrades within days) |
| Carbon monoxide (CO) | CO binds Hb with 240× greater affinity than O₂; leftward shift + reduces O₂-carrying capacity — double toxicity |
| Fetal haemoglobin (HbF) | HbF has lower affinity for 2,3-BPG → left-shifted → pulls O₂ across placenta from maternal HbA |
| MetHb | Remaining Fe²⁺ subunits shift left (Darling-Roughton effect) |
Clinical importance of left shift:
- Stored blood transfusion: packed RBCs have depleted 2,3-BPG → left-shifted curve → initially poor O₂ delivery to tissues (normalises over 24h as BPG regenerates).
- CO poisoning: left shift explains why SpO₂ on pulse oximetry reads falsely normal (it can't distinguish oxyhaemoglobin from carboxyhaemoglobin), yet tissues are profoundly hypoxic.
- HbF: the left shift is physiologically essential — it lets the fetus extract O₂ from maternal blood across the placenta.
Summary Comparison
| Right Shift | Left Shift | |
|---|---|---|
| P50 | Increased (>27 mmHg) | Decreased (<27 mmHg) |
| Hb–O₂ affinity | ↓ (weaker grip) | ↑ (stronger grip) |
| O₂ loading in lungs | Slightly impaired | Enhanced |
| O₂ unloading in tissues | Enhanced ✓ | Impaired ✗ |
| Key causes | ↑CO₂, ↑H⁺, ↑temp, ↑2,3-BPG | ↓CO₂, ↓H⁺, ↓temp, ↓2,3-BPG, CO, HbF |
| Net clinical effect | Better O₂ delivery | O₂ trapped in blood |
The key insight: a right shift is generally adaptive (tissues get more O₂) while a left shift is generally maladaptive in most clinical settings — except in the specific case of HbF pulling O₂ across the placenta, where the left shift is the whole point.
Sources: Guyton & Hall Medical Physiology; Goldman-Cecil Medicine 22nd ed.; Lippincott Biochemistry 8th ed.
This is a shared conversation. Sign in to Orris to start your own chat.