Potassium handling in alpha intercalated cells
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Loading Image
Searching PubMed
"alpha intercalated cell" AND "potassium" AND "H-K-ATPase"
I now have comprehensive information to provide a detailed answer. Here is a synthesis from multiple authoritative nephrology textbooks:
Potassium Handling in Alpha-Intercalated (Type A) Cells
Alpha-intercalated (α-IC) cells reside in the cortical collecting duct (CCD), outer medullary collecting duct (OMCD), and inner medullary collecting duct (IMCD). In the CCD, they exist alongside principal cells and β-intercalated cells. Their primary role with respect to potassium is active K⁺ reabsorption — a critical counterbalance to principal cell K⁺ secretion.
Core Transport Mechanism: Apical H⁺-K⁺-ATPase
The defining potassium transporter in α-IC cells is the apical H⁺-K⁺-ATPase (a P-type ATPase). This pump:
- Secretes H⁺ into the tubular lumen (contributing to urinary acidification)
- Reabsorbs K⁺ from the lumen into the cell in a coupled, electroneutral exchange
Two H⁺-K⁺-ATPase α-isoforms are expressed:
- HKα1 — homologous to the gastric isoform
- HKα2 — homologous to the colonic isoform
Once K⁺ enters the cell via this pump, it has two fates depending on potassium balance:
- Recycle back across the apical membrane (via apical K⁺ channels, particularly during K⁺ replete states)
- Exit across the basolateral membrane into the interstitium (during K⁺ depletion, promoting net K⁺ reabsorption)
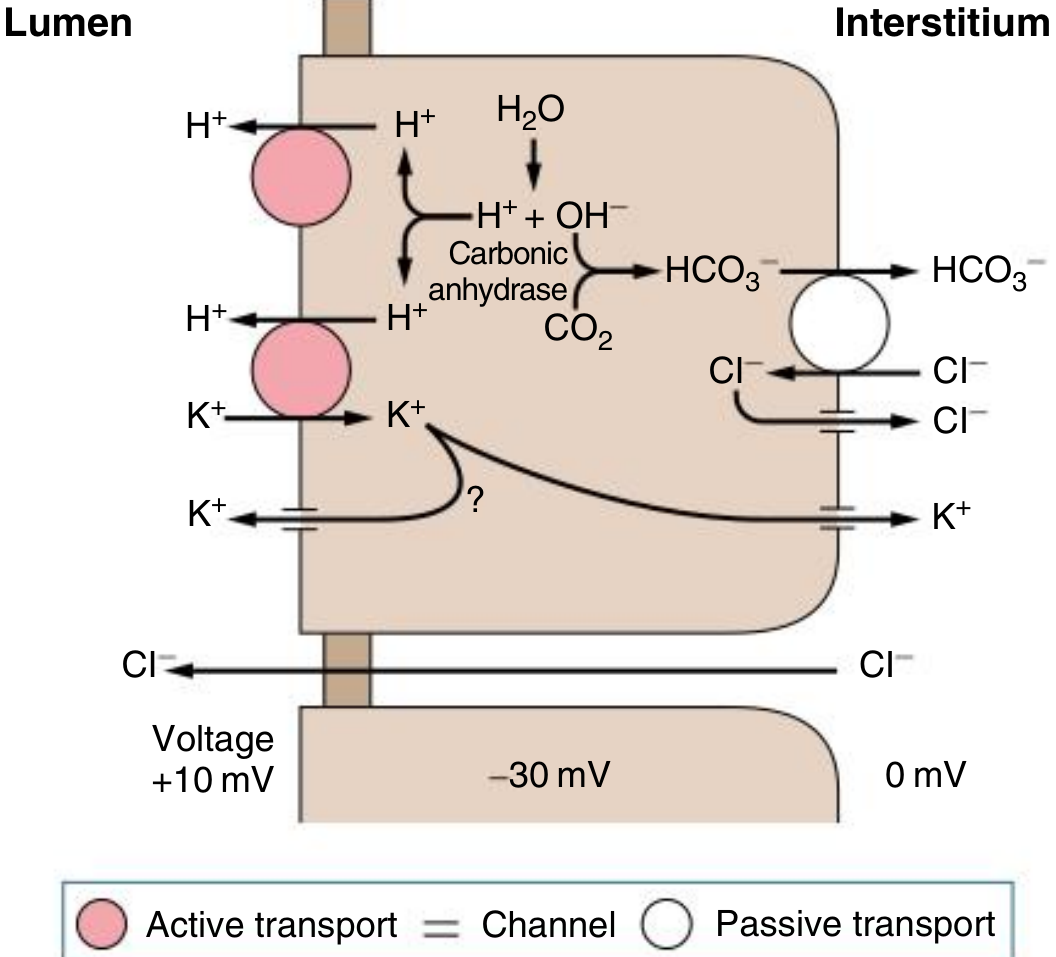
Fig. 12.4 — α-Intercalated cell of the cortical collecting duct. H⁺ is secreted into the lumen via H⁺-ATPase and H⁺-K⁺-ATPase (active transport, pink circles). K⁺ reabsorbed by H⁺-K⁺-ATPase can either recycle apically or exit basolaterally. HCO₃⁻ exits basolaterally via kAE1 (Cl⁻/HCO₃⁻ exchanger). — Comprehensive Clinical Nephrology, 7th ed.
Secondary K⁺ Role: BK Channel-Mediated K⁺ Secretion
Under conditions of high tubular flow, α-IC cells also secrete K⁺ via large-conductance, Ca²⁺- and stretch-activated Big Potassium (BK) channels on their luminal surface. Flow-mediated deflection of luminal cilia on both principal cells and α-IC cells activates these BK channels. This flow-induced K⁺ secretion contributes significantly to urinary K⁺ excretion during:
- Volume expansion
- Diuretic use (loop or thiazide)
- Osmotic diuresis
Basolateral Machinery
The basolateral membrane of the α-IC cell contains:
- kAE1 (truncated erythrocyte anion exchanger, SLC4A1) — mediates Cl⁻/HCO₃⁻ exchange, allowing bicarbonate reabsorption into the bloodstream
- KCC4 (KCl cotransporter) — exports Cl⁻ that entered via kAE1
- ClC-Kb/ClC-K2 Cl⁻ channel — additional Cl⁻ recycling
- H⁺-ATPase (vacuolar type) — maintains intracellular electronegativity and cell volume (functionally substitutes for Na⁺-K⁺-ATPase in this cell type)
Regulation of H⁺-K⁺-ATPase (K⁺ Reabsorption)
| Stimulus | Effect on H⁺-K⁺-ATPase | Net Renal K⁺ Effect |
|---|---|---|
| K⁺ depletion / hypokalemia | ↑ Expression & activity | ↑ K⁺ reabsorption, ↓ K⁺ excretion |
| Aldosterone | ↑ Expression & activity | Minimizes hypokalemia during hyperaldosteronism |
| Metabolic acidosis | ↑ Activity (direct + via ↑ ammonia) | ↑ K⁺ reabsorption |
| K⁺ excess / hyperkalemia | ↓ Expression | ↓ K⁺ reabsorption |
Importantly, aldosterone acts on both principal cells (↑ K⁺ secretion via ENaC/ROMK) and α-IC cells (↑ H⁺-K⁺-ATPase), so its net renal effect on K⁺ balance depends on the balance between these opposing forces. During chronic aldosterone excess, upregulation of H⁺-K⁺-ATPase may partially blunt the degree of urinary K⁺ wasting.
Interplay with Principal Cells
α-IC cell K⁺ reabsorption occurs in parallel with principal cell K⁺ secretion. The net urinary K⁺ excretion reflects the sum of:
- Principal cell secretion (via ROMK and BK channels, driven by ENaC-mediated Na⁺ reabsorption and the resulting lumen-negative potential)
- α-IC cell reabsorption (via H⁺-K⁺-ATPase)
This bidirectional arrangement allows rapid, fine-tuned regulation of renal K⁺ excretion across a wide range of dietary K⁺ intakes.
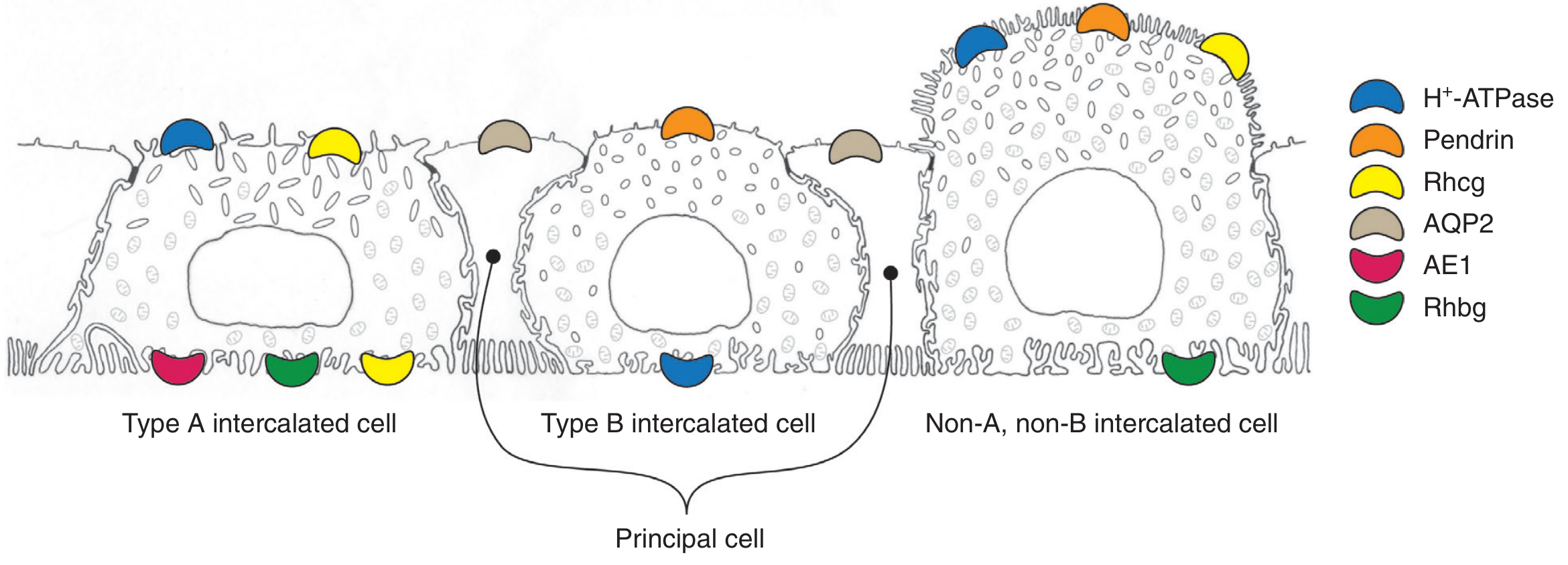
Fig. 9.3 — Three intercalated cell subtypes. Type A (α-IC): apical H⁺-ATPase + basolateral AE1 (kAE1, pink) + basolateral Rhbg (green). H⁺-K⁺-ATPase not depicted here but is co-expressed apically. — Brenner & Rector's The Kidney
Key Summary
| Feature | α-Intercalated Cell |
|---|---|
| Location | CCD, OMCD, IMCD |
| Primary K⁺ role | Active reabsorption via apical H⁺-K⁺-ATPase |
| Secondary K⁺ role | Secretion via BK channels (flow-activated) |
| K⁺ fate intracellularly | Apical recycling or basolateral exit (K⁺-balance dependent) |
| Upregulated by | Hypokalemia, metabolic acidosis, aldosterone |
| Clinical relevance | Hypokalemia → ↑ H⁺-K⁺-ATPase → K⁺ conservation; metabolic alkalosis risk |
Sources: Comprehensive Clinical Nephrology, 7th ed. · Brenner & Rector's The Kidney · NKF Primer on Kidney Diseases, 8th ed. · Harrison's Principles of Internal Medicine, 22nd ed.
This is a shared conversation. Sign in to Orris to start your own chat.