Describe etiology, clinical features and management of bronchial asthma
Reading File
Finding Sources
Finding Sources
Searching PubMed
"asthma"[MeSH Terms] AND "management"[MeSH Terms]
Finding Sources
Searching PubMed
"asthma" AND "treatment" AND "guidelines"
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the complete answer from all the textbook sources gathered.
Bronchial Asthma
Definition
Asthma is a chronic inflammatory disorder of the airways characterized by increased responsiveness to multiple stimuli, causing recurrent episodes of wheezing, breathlessness, chest tightness, and coughing — particularly at night or in the early morning — with widespread but variable airflow obstruction that is usually reversible. — Tintinalli's Emergency Medicine
Etiology and Pathogenesis
Classification
Asthma is broadly divided into two forms:
1. Atopic (Extrinsic/Allergic) Asthma
- Most common type; classic example of type I IgE-mediated hypersensitivity
- Usually begins in childhood; positive family history of atopy/asthma common
- Often preceded by allergic rhinitis, urticaria, or eczema
- Triggers: allergens in dust, pollen, animal dander, food, infections
- Skin test with offending antigen produces immediate wheal-and-flare
2. Nonatopic (Intrinsic) Asthma
- No evidence of allergen sensitization; skin tests usually negative
- Less common family history
- Triggered by viral respiratory infections (rhinovirus, parainfluenza), inhaled pollutants (sulfur dioxide, ozone), cold air, stress, exercise
- Virus-induced mucosal inflammation lowers the threshold of subepithelial vagal receptors
Both types share a final common pathway: mast cell and eosinophil activation causing bronchoconstriction, inflammation, and mucus production. — Robbins Basic Pathology
Triggering Factors (Precipitants)
| Category | Examples |
|---|---|
| Allergens | Dust mites, cockroaches, animal dander, pollen, mold |
| Infections | Viral URIs account for 40–80% of adult exacerbations, 80% in children |
| Exercise | Common, especially in cold/dry air |
| Drugs | Aspirin/NSAIDs, β-blockers |
| Occupational | Metal salts, wood/vegetable dust, industrial chemicals, isocyanates |
| Air pollutants | Cigarette smoke, ozone, SO₂, combustion products |
| Emotional stress | Via vagal and adrenergic pathways |
Immunopathogenesis
Atopic Asthma — Th2-Driven Mechanism:
Inhaled allergens are processed by dendritic cells → activate CD4⁺ Th2 cells → cytokine release:
- IL-4 and IL-13: stimulate B cells to produce IgE
- IL-5: recruits and activates eosinophils
- IL-13: increases mucus production
IgE binds to Fc receptors on submucosal mast cells → on re-exposure, allergens cross-link IgE → mast cell degranulation → two phases:
Early Phase (minutes):
- Bronchoconstriction triggered by histamine, prostaglandin D₂, leukotrienes C₄, D₄, E₄
- Increased mucus production, vasodilation, increased vascular permeability
Late Phase (hours):
- Inflammatory mediators stimulate epithelial cells to release chemokines (including eotaxin) → recruitment of Th2 cells, eosinophils, neutrophils, basophils
- Eosinophils release major basic protein and eosinophil cationic protein → further epithelial damage and bronchoconstriction
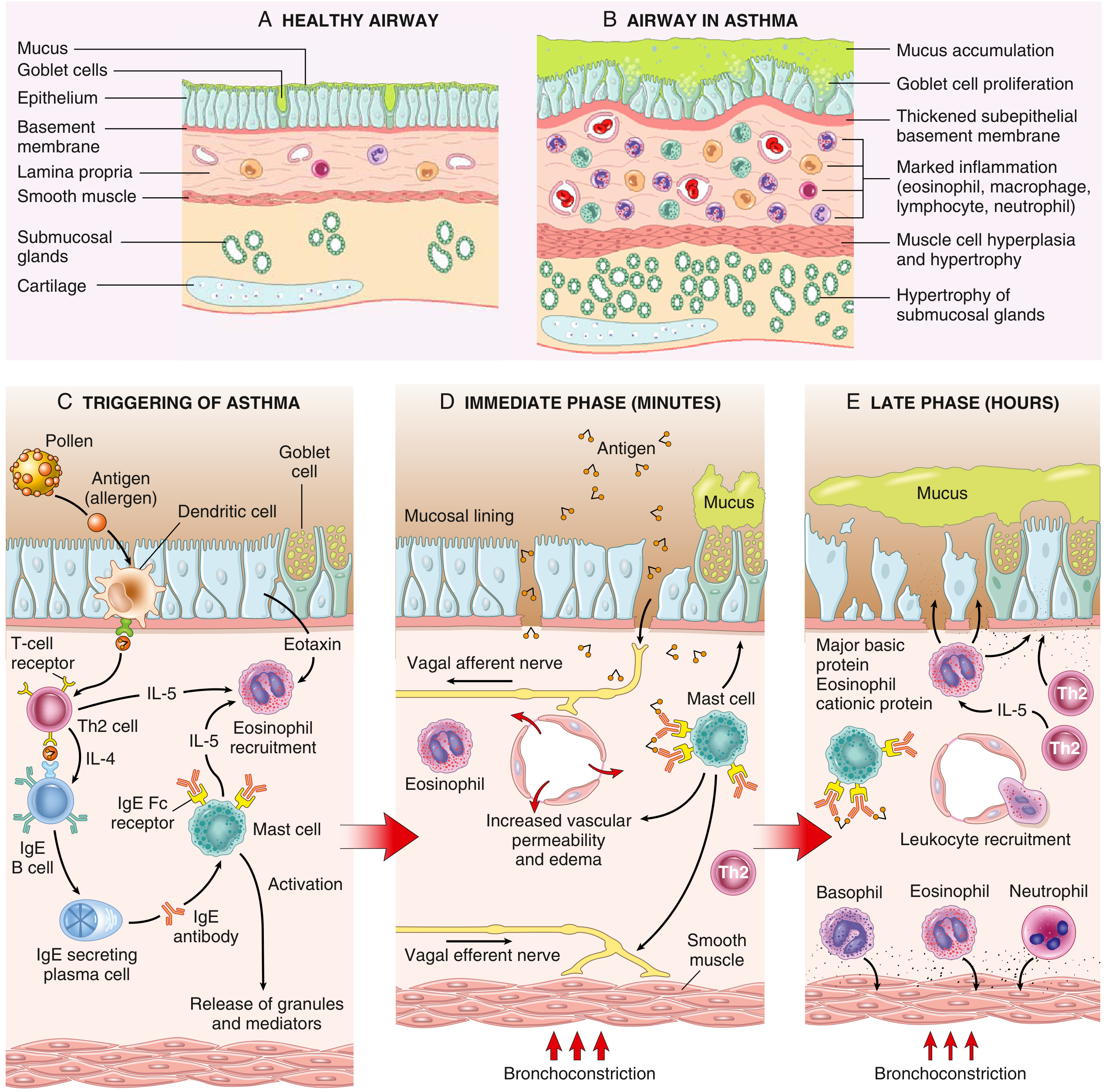
Airway Remodeling
Repeated bouts of inflammation lead to permanent structural changes:
- Hypertrophy and hyperplasia of bronchial smooth muscle
- Subepithelial fibrosis and basement membrane thickening
- Mucus gland hyperplasia and hypersecretion (goblet cell metaplasia)
- Angiogenesis
- Deposition of sub-epithelial collagen (may begin years before symptoms)
These changes contribute to non-reversible loss of lung function in chronic disease. — Tintinalli's Emergency Medicine
Genetic Factors
- Asthma shows familial clustering but genetics are complex
- GWAS studies have identified variants in genes such as the IL-4 receptor and others clearly linked to asthma pathogenesis
- Genetic predisposition to atopy (type I hypersensitivity) is key
- The hygiene hypothesis proposes that lack of microbial exposure in early childhood results in later immunological hyperreactivity — Robbins Basic Pathology
Morphology (Pathological Changes)
Macroscopically in fatal asthma:
- Lungs over-distended with patchy atelectasis
- Airway lumens contain thick mucus plugs (composed of mucus, serum proteins, inflammatory cells, cellular debris)
Microscopically:
- Goblet cell metaplasia of airway epithelium; increased mucus
- Charcot-Leyden crystals (from eosinophil-derived galectin-10) in mucus
- Thickened basement membrane (subepithelial fibrosis)
- Marked inflammatory infiltrate: eosinophils, macrophages, lymphocytes, neutrophils
- Hypertrophy and hyperplasia of bronchial smooth muscle
- Hypertrophy of submucosal glands
Clinical Features
Symptoms
- Episodic dyspnea (breathlessness), classically worse at night and early morning
- Wheezing (expiratory > inspiratory)
- Chest tightness
- Cough (may be the only symptom — "cough-variant asthma")
- Symptoms have wide-spread and variable airflow obstruction, characteristically reversible between episodes
Signs During an Attack
- Tachypnea, tachycardia
- Use of accessory muscles of respiration
- Prolonged expiration; expiratory polyphonic wheeze
- Hyperinflated (barrel) chest
- Pulsus paradoxus (>10 mmHg drop in systolic BP during inspiration) in severe attacks
- Cyanosis in severe/life-threatening attacks
Features of Severe/Life-Threatening Attack
- Silent chest (no wheeze despite severe obstruction — ominous sign)
- Inability to speak in sentences
- Altered consciousness
- SpO₂ < 92%; PaCO₂ rising (indicates fatigue and impending respiratory failure)
- Respiratory muscle fatigue with ventilatory failure
Investigations
Pulmonary Function Tests:
- Spirometry: reduced FEV₁, reduced FEV₁/FVC ratio (obstructive pattern), with ≥12% (and ≥200 mL) reversibility after bronchodilator
- Peak Expiratory Flow Rate (PEFR): reduced; diurnal variation >20% is diagnostic
- Bronchoprovocation testing (methacholine/PC20): used when spirometry is normal
Blood Tests:
- Eosinophilia (peripheral blood)
- Elevated total and allergen-specific IgE
Exhaled Nitric Oxide (FeNO):
- Elevated (>35–40 ppb) indicates type 2 airway inflammation; useful to guide ICS therapy
Imaging:
- CXR: hyperinflation, peribronchial thickening; useful to exclude pneumothorax, infection
- CT chest: bronchiectasis, air trapping in refractory cases
Skin Prick Tests / Serum RAST: identify specific allergen sensitivities
Sputum: eosinophils, Charcot-Leyden crystals
Management
Goals of Therapy (GINA)
- Symptoms ≤2 times/week
- Nocturnal awakenings ≤2 times/month
- Reliever use ≤2 times/week (except pre-exercise)
- No more than 1 exacerbation/year
- Optimization of lung function and maintenance of normal daily activities
- Minimal or no treatment side effects
— Harrison's Principles of Internal Medicine, 22e
A. Non-Pharmacological Management
- Allergen/trigger avoidance: remove pets, pest control, dust mite covers, avoid tobacco smoke and cannabis combustion products
- Occupational asthmatics: removal from offending environment may achieve resolution
- Patient education: inhaler technique, PEFR monitoring, action plans
- Treat comorbidities: rhinosinusitis, GERD, obesity, OSA — all worsen asthma control
B. Pharmacological Management — Stepwise (GINA/NAEPP)
Medications are divided into:
- Reliever medications: rapid-onset bronchodilators for acute symptoms
- Controller medications: anti-inflammatory agents for long-term control
GINA Step Therapy (Ages 12+)
| Step | Preferred Regular Controller | As-Needed Reliever |
|---|---|---|
| Step 1 | None | Low-dose ICS/formoterol OR SABA |
| Step 2 | None OR low-dose ICS | Low-dose ICS/formoterol OR SABA |
| Step 3 | Low-dose ICS/formoterol | Low-dose ICS/formoterol |
| Step 4 | Medium-dose ICS/formoterol | Low/medium-dose ICS/formoterol |
| Step 5 | High-dose ICS/LABA + add-on LAMA; consider biologics | As above |
ICS is now recommended at all steps. Note: ICS/formoterol as both controller and reliever (MART = Maintenance And Reliever Therapy) is the preferred approach at Steps 3–4. — Harrison's Principles of Internal Medicine, 22e
Key Drug Classes
1. Inhaled Corticosteroids (ICS) — Cornerstone of controller therapy
- Examples: budesonide, beclomethasone, fluticasone
- Reduce airway inflammation, prevent remodeling
- Side effects at high doses: thrush (reduced by spacer + gargling), hoarseness, bruising, osteoporosis, cataracts, growth suppression in children
2. Short-Acting β₂-Agonists (SABA) — First-line reliever
- Albuterol (salbutamol) 2.5–5 mg via nebulizer q20 min × 3 doses in acute attack; or 4–8 puffs via MDI q20 min
- Levalbuterol: active R-isomer, half the dose equivalent
- Mechanism: relax airway smooth muscle via β₂-receptor activation → rapid bronchodilation
3. Long-Acting β₂-Agonists (LABA)
- Examples: formoterol, salmeterol
- Must always be combined with ICS — never used as monotherapy in asthma
- Formoterol has rapid onset, used as both controller and reliever
4. Anticholinergics
- Short-acting: Ipratropium bromide 0.5 mg q20 min × 3 doses — added to SABA in severe acute asthma; not first-line
- Long-acting: LAMA (tiotropium) as add-on at Step 5
5. Leukotriene Modifiers
- Montelukast (CysLT1 antagonist): oral, once daily; preferred in children (avoids ICS growth concerns); effective in exercise-induced bronchoconstriction and aspirin-exacerbated respiratory disease
- Zileuton (5-lipoxygenase inhibitor): raises LFTs in 3%; inhibits CYP1A2
- Zafirlukast: oral, twice daily
- Safety note: montelukast carries a black-box warning for neuropsychiatric events (suicidal ideation)
6. Systemic Corticosteroids
- Oral: prednisone 40–60 mg/day with taper over 1–2 weeks for acute exacerbations; lowest possible dose for refractory disease
- IV: methylprednisolone in hospitalized patients; transitioned to oral once stable
- IM: triamcinolone acetonide for poorly adherent patients
- Chronic OCS side effects: diabetes, osteoporosis, cataracts, hypertension, truncal obesity, peptic ulcers, immunosuppression
7. Biologics (Step 5 — Severe Asthma)
- Anti-IgE: omalizumab — for allergic asthma with elevated IgE
- Anti-IL-5: mepolizumab, reslizumab, benralizumab — reduce eosinophils; for eosinophilic asthma
- Anti-IL-4Rα: dupilumab — blocks IL-4 and IL-13 signaling
- Anti-TSLP: tezepelumab — blocks upstream innate immune trigger
8. Other agents
- Cromolyn sodium: mast cell stabilizer; nebulized, 4×/day; used in children, exercise prophylaxis
- Azithromycin: adjunctive in severe non-eosinophilic asthma; reduces exacerbations
C. Management of Acute Severe Asthma / Status Asthmaticus
- High-flow oxygen to maintain SpO₂ 94–98%
- SABA (albuterol): 2.5–5 mg nebulized q20 min × 3, or continuous nebulization 10–15 mg/hr
- Ipratropium bromide 0.5 mg q20 min × 3 (added to SABA)
- Systemic corticosteroids: IV methylprednisolone or oral prednisolone 40–60 mg immediately
- IV magnesium sulfate (2 g over 20 min): bronchodilator effect in severe/refractory cases
- Consider IV salbutamol or IV aminophylline if failing initial therapy
- NIV/intubation and mechanical ventilation for impending respiratory failure
D. Step-Down Therapy
Once asthma is well-controlled for several months, medications are tapered. Reduction starts with the agent of greatest toxicity (e.g., oral steroids first). ICS should generally be maintained as baseline protection. Nonadherence is a common reason for poor control and must be actively assessed. — Fishman's Pulmonary Diseases
Key Comorbidities Worsening Control
- Allergic rhinitis ("united airway disease") — treating upper airway inflammation improves asthma
- GERD — acid reflux triggers bronchoconstriction
- Obesity — associated with a difficult-to-control phenotype
- OSA — worsens asthma outcomes; CPAP improves quality of life and reduces exacerbations
- Anxiety/depression — contribute to nonadherence
Sources: Robbins Basic Pathology; Harrison's Principles of Internal Medicine, 22e; Tintinalli's Emergency Medicine (7e); Fishman's Pulmonary Diseases and Disorders; Murray & Nadel's Textbook of Respiratory Medicine
This is a shared conversation. Sign in to Orris to start your own chat.