Transient elastography
Reading File
Finding Sources
Finding Sources
Searching PubMed
transient elastography liver fibrosis
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Searching Images
transient elastography FibroScan liver stiffness measurement
I have all the information needed for a comprehensive answer.
Transient Elastography
Transient elastography (TE) — commercially marketed as FibroScan (vibration-controlled TE, or VCTE) — is a non-invasive, ultrasound-based technique that quantifies liver stiffness as a surrogate for hepatic fibrosis. It has largely replaced liver biopsy for this purpose in clinical practice.
Principle of Operation
TE uses a low-amplitude, low-frequency (50 Hz) shear wave generated by a probe placed in an intercostal space. A pulse-echo ultrasound system tracks the propagation velocity of this shear wave through a standardized window of liver tissue (1 cm × 4 cm — approximately 100 times the volume of a typical liver biopsy specimen). The underlying physics:
Shear waves propagate faster through stiffer tissue.
The velocity is converted to an elastic modulus expressed in kilopascals (kPa). Higher kPa = stiffer liver = more fibrosis.
— Sleisenger and Fordtran's Gastrointestinal and Liver Disease
Clinical Interpretation (kPa Cutoffs)
| Liver Stiffness | Interpretation |
|---|---|
| < 6 kPa | Normal / no significant fibrosis |
| 6–8 kPa | Mild fibrosis (F1–F2) |
| > 8 kPa | ≥ F3 (advanced fibrosis) |
| > 9.9 kPa | Advanced fibrosis (AUROC 0.93 in NAFLD studies) |
| > 10 kPa | Correlates with cirrhosis in HBV patients |
| > 12.5 kPa | F4 cirrhosis (alcohol-associated liver disease) |
Note: Exact cutoffs vary by etiology (HCV, HBV, NAFLD/MASH, alcohol). Disease-specific cutoffs should be applied.
— Harrison's Principles of Internal Medicine 22E; Sleisenger and Fordtran's
Additional Measurement: Controlled Attenuation Parameter (CAP)
FibroScan devices also provide the CAP score (dB/m), which quantifies ultrasound signal attenuation to estimate hepatic steatosis degree — a useful companion metric, particularly in NAFLD/MASH evaluation.
Diagnostic Performance
- Best at: Distinguishing cirrhosis from no cirrhosis (high sensitivity and specificity)
- Less accurate for: Intermediate fibrosis stages (F1–F2)
- A meta-analysis confirmed high performance for cirrhosis vs. no cirrhosis, with reduced accuracy for lesser degrees of fibrosis
- Approved by the FDA in 2013 for use in patients with liver disease
- Validated across: chronic hepatitis C, chronic hepatitis B, NAFLD, primary biliary cholangitis (PBC), hemochromatosis, post-liver transplant recurrent hepatitis, alcohol-associated liver disease
— Sleisenger and Fordtran's Gastrointestinal and Liver Disease
Probes
| Probe | Use |
|---|---|
| M probe (standard) | Standard patients; ~27% unreliable results in obese patients |
| XL probe | Obesity; improves reliability by sampling deeper tissue |
Limitations and Confounders
Several factors can cause falsely elevated liver stiffness readings (overestimation of fibrosis):
- Obesity / increased skin-to-liver distance → reduced signal reliability
- Ascites → shear waves dispersed; TE unreliable
- Extrahepatic cholestasis → stiffness elevated independently of fibrosis
- Hepatic inflammation (active hepatitis flare) → transient stiffness elevation
- Hepatic steatosis → can adversely affect performance
- Food intake within 2 hours → portal blood flow increases stiffness transiently
- Right heart failure → hepatic congestion elevates stiffness
MR elastography (MRE) is preferred when TE is unreliable (ascites, obesity) — it images a larger liver volume, reduces sampling error, and accurately assesses fatty livers (unlike shear-wave TE). However, MRE is more expensive and less widely available.
— Yamada's Textbook of Gastroenterology; Miller's Anesthesia; Goldman-Cecil Medicine
Clinical Applications
- Staging fibrosis in chronic liver disease (HCV, HBV, NAFLD, alcohol)
- Monitoring fibrosis regression with treatment (e.g., HCV cure, alcohol abstinence, weight loss)
- Screening for varices: A low stiffness score + normal platelet count identifies patients at low risk for variceal bleeding, avoiding unnecessary endoscopy
- Selecting patients for liver biopsy: TE can triage who needs invasive testing vs. who can be managed non-invasively
- Hemochromatosis: Evaluating degree of hepatic fibrosis alongside serum ferritin and hyaluronic acid
Comparison with Other Elastography Modalities
| Modality | Method | Notes |
|---|---|---|
| VCTE / FibroScan | 1D shear wave (50 Hz) | Most widely used, FDA-approved |
| Point SWE (ARFI) | Acoustic radiation force | Integrated into standard US machines |
| 2D SWE | Acoustic radiation force | Color-mapped; larger ROI |
| MR Elastography | 65 Hz wave + MRI | Most accurate; unaffected by obesity/ascites/steatosis; expensive |
Illustrative FibroScan Interfaces
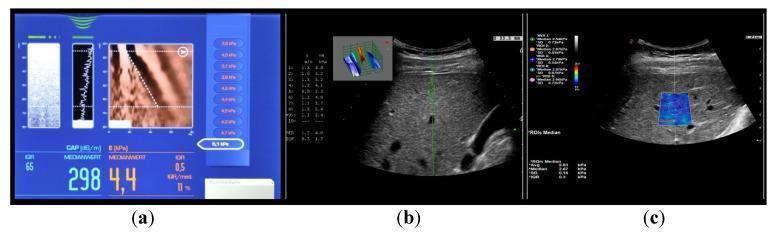
Panel (a): FibroScan display showing LSM 4.4 kPa (normal) and CAP 298 dB/m (steatosis). Panels (b) and (c) show point SWE and 2D SWE comparisons.

FibroScan result: LSM 35.3 kPa (F4/cirrhosis on the METAVIR scale, shown in red zone) with low steatosis (CAP 211 dB/m).
Recent Evidence
Recent systematic reviews support TE's role:
- AASLD-supporting systematic review (Hepatology 2025) — imaging-based noninvasive liver disease assessment for staging fibrosis in chronic liver disease [PMID: 38489521]
- WHO 2024 guidelines meta-analysis (Lancet Gastroenterol Hepatol 2025) — non-invasive tests including TE for staging fibrosis/cirrhosis in chronic hepatitis B [PMID: 39983746]
- Individual participant data meta-analysis (Liver Int 2024) — diagnostic accuracy of non-invasive tests including TE for at-risk MASH [PMID: 38573034]
NAFLD
Finding Sources
Searching PubMed
nonalcoholic fatty liver disease NAFLD MAFLD
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Searching Images
NAFLD NASH spectrum steatosis steatohepatitis fibrosis cirrhosis pathology
Reading File
Reading File
I now have comprehensive content. Let me compile the full answer.
NAFLD / MASLD (Nonalcoholic Fatty Liver Disease)
Nomenclature Update
The field has recently undergone a significant renaming. Under the umbrella term Steatotic Liver Disease (SLD):
| Old Term | New Term |
|---|---|
| NAFLD | MASLD (Metabolic dysfunction-Associated Steatotic Liver Disease) |
| NASH | MASH (Metabolic dysfunction-Associated SteatoHepatitis) |
| — | MetALD (metabolic + alcohol, 20–50 g/d women; 30–60 g/d men) |
The new nomenclature emphasizes the metabolic underpinning and reduces stigma from the word "fatty." Epidemiologic overlap between NAFLD/NAFLD and MASLD/MASH criteria is ≥98%.
— Harrison's Principles of Internal Medicine 22E (2025)
Disease Spectrum
NAFLD/MASLD is not a single entity but a spectrum:
Hepatic Steatosis (MASL) → Steatohepatitis (MASH) → Fibrosis (F1→F4) → Cirrhosis → HCC
↑ ↑
Clinically benign Primary driver of fibrosis
- Steatosis alone (≥5% hepatocytes): Most clinically benign; may remain stable for years or transition to MASH
- MASH: Steatosis + hepatocyte ballooning + lobular inflammation ± fibrosis; drives progressive fibrosis
- Fibrosis stages: F0 (none) → F1 (minimal) → F2 (significant) → F3 (bridging/advanced) → F4 (cirrhosis)
- Chronic steatohepatitis is the primary predictor of hepatic fibrosis in MASLD
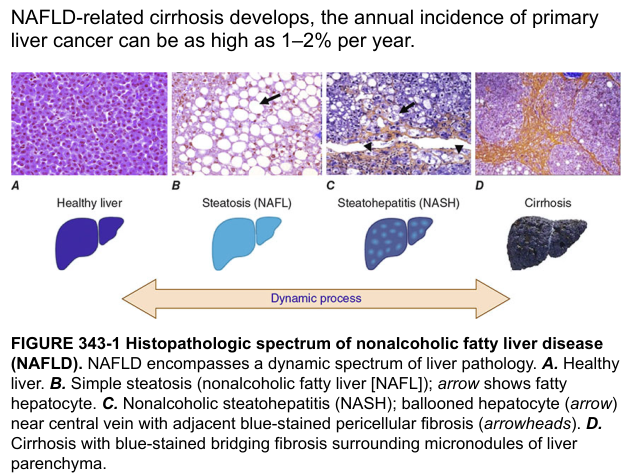
Harrison's Fig. 354-1: Histopathologic spectrum of NAFLD — healthy liver, steatosis (NAFL), steatohepatitis (NASH), and cirrhosis
Epidemiology
- Most common chronic liver disease in the US and worldwide
- Prevalence in US adults: 25–30% have MASLD; 3–6% of those have MASH
- MASH identified in 14% of asymptomatic patients ≥50 undergoing colon cancer screening
- Hepatic fibrosis ≥F2 has more than doubled in the past two decades
- Currently a leading indication for liver transplantation in the US
- Ethnic variation: Lowest prevalence in African Americans (~25%), highest in Hispanic and Asian-Indians (~50%)
- Increasing in children and adolescents in parallel with the obesity epidemic
Risk Factors / Associations
Core metabolic risk factors (one or more required for MASLD diagnosis):
- Overweight / obesity (especially central/truncal obesity)
- Insulin resistance / Type 2 diabetes
- Dyslipidemia (hypertriglyceridemia, low HDL, high LDL)
- Hypertension
- Metabolic syndrome
Also associated with: PCOS, OSA, hypothyroidism, chronic fatigue, mood disorders
Lean MASLD: Occurs in normal-BMI individuals, especially with lipodystrophy and truncal obesity
Pathogenesis
The central initiating events are obesity and insulin resistance:
- ↑ Lipolysis in adipose tissue → excess free fatty acids (FFAs) delivered to liver
- ↓ Adiponectin → reduced FFA β-oxidation in skeletal muscle → increased hepatocyte FFA uptake → triglyceride accumulation (steatosis)
- Hepatotoxic lipid metabolites → ER stress, mitochondrial dysfunction, increased reactive oxygen species (ROS)
- Inflammasome activation in hepatocytes → IL-1 release → local inflammation
- Gut microbiome dysbiosis → increased gut-derived endotoxin → liver inflammation
- Hepatocyte injury → stellate cell activation → collagen deposition → fibrosis
— Robbins & Kumar Basic Pathology
Clinical Features
- Often asymptomatic (steatosis or even MASH may cause no symptoms)
- Most common cause of incidental elevated serum transaminases
- AST:ALT ratio typically < 1 (vs. alcohol-related liver disease where ratio > 2)
- Symptoms when present: fatigue, malaise, right upper quadrant discomfort
- Advanced disease: signs of portal hypertension (ascites, splenomegaly, varices)
- Cardiovascular disease risk is elevated (shared metabolic risk factors) — a leading cause of death in MASLD patients
Diagnosis
Imaging:
- Ultrasound: bright/echogenic liver (sensitive if >20% fat); insensitive for inflammation or fibrosis
- CT: lower density than spleen in steatosis
- MRI-PDFF: most accurate quantification of hepatic fat
- CAP (Controlled Attenuation Parameter): ultrasound-based fat quantification on FibroScan
Non-invasive fibrosis scoring:
| Score | Components | Use |
|---|---|---|
| FIB-4 | Age, AST, ALT, platelets | Exclude advanced fibrosis (<1.3 = low risk; >3.25 = high risk) |
| NFS (NAFLD Fibrosis Score) | Age, BMI, IFG/DM, AST/ALT, platelets, albumin | Stage fibrosis |
| APRI | AST, platelet count | Simple screening |
| ELF (Enhanced Liver Fibrosis) | TIMP-1, PIIINP, hyaluronic acid | Blood-based, predicts liver outcomes |
| VCTE (FibroScan, kPa) | Liver stiffness | AUROC 0.93 at 9.9 kPa for advanced fibrosis |
Liver biopsy: Still required to definitively distinguish MASH from simple steatosis (hepatocyte ballooning, lobular inflammation). The NAFLD Activity Score (NAS) grades steatosis, lobular inflammation, and ballooning.
Drug-Induced Steatosis (Secondary NAFLD)
Important causes to exclude:
| Drug | Pattern |
|---|---|
| Amiodarone, tamoxifen, methotrexate | Macrovesicular steatosis / steatohepatitis / fibrosis |
| Glucocorticoids | Macrovesicular steatosis |
| NRTIs (e.g., zidovudine) | Microvesicular steatosis (mitochondrial dysfunction) |
| Valproic acid, tetracyclines | Microvesicular steatosis |
— Harrison's Principles of Internal Medicine 22E
Treatment
1. Lifestyle Modification (Foundation for all patients)
- Weight loss 3–5%: Improves hepatic steatosis
- Weight loss >10%: Improves MASH and fibrosis
- Mediterranean diet preferred (long-term adherence, cardiovascular benefit)
- Avoid: saturated fats, refined carbohydrates, sugar-sweetened beverages, excess fructose
- Coffee (≥3 cups/day): associated with reduced fibrosis risk and lower HCC risk
- Exercise: ≥150 min/week moderate, or ≥60 min/week intensive — improves insulin sensitivity and MASH histology independent of weight loss
2. Pharmacologic Therapies
| Agent | Mechanism | Evidence | Notes |
|---|---|---|---|
| Resmetirom (Rezdiffra) | THR-β agonist | FDA accelerated approval (2024) for at-risk MASH (F2–F3 without cirrhosis) | MAESTRO-NASH: NASH resolution 30% vs 10% placebo; fibrosis improvement 26% vs 14% |
| Pioglitazone | PPARγ agonist | PIVENS trial: NASH resolution 47% vs 21% placebo | Weight gain, bone loss, possible bladder cancer risk |
| GLP-1 RAs (semaglutide, liraglutide) | Incretin mimetics | Improve steatosis, inflammation; emerging fibrosis data | Weight loss, cardiovascular benefit; broadly used |
| SGLT-2 inhibitors | Glycosuria → weight loss | Improve LFTs and steatosis; histologic endpoints pending | Cardioprotective |
| Vitamin E (800 IU/d) | Antioxidant | PIVENS: NASH resolution 43% vs 19%; meta-analysis supportive | Concerns: hemorrhagic stroke, cardiac mortality, prostate cancer; no antifibrotic effect proven |
| Statins | HMG-CoA reductase inhibition | Safe across MASLD spectrum including compensated cirrhosis; reduce CV mortality; may decrease HCC risk | Underutilized; do NOT worsen liver disease in MASLD |
Not recommended for MASH: ursodeoxycholic acid, betaine, DPP-4 inhibitors, silymarin — no meaningful histologic benefit.
3. Bariatric / Metabolic Surgery
- Indicated in selected patients with obesity; can improve or reverse MASH and fibrosis
4. Liver Transplantation
- MASLD/MASH-cirrhosis: currently a leading indication in the US
- Post-transplant survival comparable to other indications
- Important: screen for cardiovascular/renal disease before listing (major comorbidities increase waitlist mortality)
- Donor livers with >30% macrovesicular steatosis carry increased graft failure risk
Complications
- Cirrhosis and hepatic decompensation (ascites, SBP, variceal bleeding, HE, HRS)
- Hepatocellular carcinoma (HCC): Rising as HCV declines; can occur in non-cirrhotic MASH livers
- Cardiovascular disease: Leading cause of death in MASLD (not liver disease for most patients)
- Extrahepatic malignancies: MASLD is an independent risk factor
Pediatric NAFLD
- Prevalence increasing with childhood obesity
- Histology differs: inflammation/scarring more prominent in portal tracts/periportal regions (zone 1 vs. zone 3 in adults); mononuclear rather than neutrophilic infiltrates predominate
— Robbins & Kumar Basic Pathology; Harrison's Principles of Internal Medicine 22E; Sleisenger and Fordtran's Gastrointestinal and Liver Disease; Yamada's Textbook of Gastroenterology
Recent Evidence
- AASLD Practice Guideline systematic review (Hepatology 2025) — imaging-based noninvasive liver disease assessment for fibrosis staging [PMID: 38489521]
- MASLD-HCC global prevalence meta-analysis (Clin Mol Hepatol 2024) — systematic review of MASLD-related HCC global prevalence [PMID: 38623613]
- APASL Clinical Practice Guidelines (Hepatol Int 2025) — Asia-Pacific guidelines for diagnosis and management of MAFLD [PMID: 40016576]
This is a shared conversation. Sign in to Orris to start your own chat.