Rejection in lung transplant and it's treatment full details for md exam
Reading File
search_medical_text
lung transplant rejection types hyperacute acute chronic pathophysiology classification
search_medical_text
lung transplant rejection treatment management immunosuppression
search_medical_image
lung transplant rejection histology bronchiolitis obliterans imaging
GradeResponse
GradeResponse
GradeResponse
I have strong foundational results. Let me compile a comprehensive MD exam-level answer, supplementing the retrieved content with detailed clinical knowledge.
Lung Transplant Rejection — Complete MD Exam Review
Overview
Rejection is the leading cause of morbidity and mortality after lung transplantation. ~50% of recipients experience at least one acute rejection episode in the first year, and by 5 years post-transplant, ~50% develop chronic rejection (Harrison's, p. 8150). Lung allografts are uniquely susceptible due to continuous exposure to the external environment, high immunogenicity, and the lung's rich lymphatic network.
Classification of Rejection
| Type | Timing | Mechanism | Key Feature |
|---|---|---|---|
| Hyperacute | Minutes–hours | Preformed antibodies (AMR) | Fulminant graft failure |
| Acute Cellular (ACR) | Days–weeks (most common in year 1) | T-cell mediated | Lymphocytic perivascular infiltrate |
| Antibody-Mediated (AMR) | Variable | Donor-specific antibodies (DSA) | Neutrophilic vasculitis + C4d deposition |
| Chronic (CLAD) | Months–years | Mixed T-cell + antibody | BOS or RAS phenotype |
1. Hyperacute Rejection
Pathophysiology
- Caused by preformed donor-specific antibodies (DSA) against HLA or ABO antigens
- Antibodies bind donor endothelium → activate complement → neutrophil influx → microvascular thrombosis and hemorrhagic infarction
Clinical Features
- Occurs within minutes to hours of reperfusion
- Severe hypoxemia, white-out on CXR, rapid graft failure
- Rare due to pre-transplant crossmatch testing
Treatment
- Largely preventable by prospective crossmatch and ABO compatibility
- Once established: plasmapheresis, IVIG, retransplantation (prognosis very poor)
2. Acute Cellular Rejection (ACR)
Pathophysiology
- T-lymphocyte mediated (CD4+ and CD8+)
- Donor MHC molecules recognized by recipient T-cells (direct or indirect allorecognition)
- Lymphocytic infiltration of perivascular and peribronchial spaces
Timing & Risk Factors
- Most common in the first post-transplant year, but can occur anytime
- Triggered/augmented by: CMV infection, other viral infections, tapering immunosuppression
- ACR is a major risk factor for CLAD (Harrison's, p. 8150)
ISHLT Grading (A-grade: Vascular; B-grade: Airway)
A-grade (Perivascular rejection):
| Grade | Description |
|---|---|
| A0 | No rejection |
| A1 | Minimal — scattered mononuclear cells around vessels |
| A2 | Mild — more prominent perivascular cuffing |
| A3 | Moderate — perivascular + alveolar involvement |
| A4 | Severe — diffuse perivascular, interstitial, alveolar infiltrates with necrosis |
B-grade (Lymphocytic bronchiolitis):
| Grade | Description |
|---|---|
| B0 | No airway inflammation |
| B1R | Low-grade lymphocytic bronchiolitis |
| B2R | High-grade lymphocytic bronchiolitis |
| BX | Ungradeable |
Clinical Features
- Fever, malaise, dyspnea, decreased exercise tolerance
- Decline in FEV1 / FVC (≥10–15% drop from baseline)
- Hypoxemia, new infiltrates on CXR/CT
- May be asymptomatic (detected on surveillance bronchoscopy)
Diagnosis
- Transbronchial biopsy (TBBx) via bronchoscopy — gold standard
- BAL to exclude infection (always rule out before treating rejection)
- PFTs: obstructive pattern
- CT: ground-glass opacities, septal thickening
Treatment of ACR
First-line:
- IV methylprednisolone 500–1000 mg/day × 3 days (pulse corticosteroids)
- Response rate ~80–90%
Maintenance optimization after ACR:
- Optimize baseline immunosuppression (ensure therapeutic tacrolimus levels)
- Add/increase mycophenolate mofetil (MMF)
Steroid-refractory ACR:
- Antithymocyte globulin (ATG) — polyclonal T-cell depleting agent
- Total lymphoid irradiation (TLI)
- Photopheresis (extracorporeal photochemotherapy)
3. Antibody-Mediated Rejection (AMR)
Pathophysiology
- Mediated by donor-specific antibodies (DSA) against HLA Class I and II
- DSA bind endothelium → complement activation → C4d deposition → neutrophilic capillaritis
- Can be acute (early) or chronic
Histological Hallmarks (ISHLT 2016 Criteria)
- Neutrophilic capillaritis
- C4d deposition in alveolar capillaries (by immunofluorescence or immunohistochemistry)
- Circulating DSA (detected by Luminex single-antigen bead assay)
Clinical Features
- Acute: rapidly progressive respiratory failure, diffuse alveolar damage
- Chronic: contributes to CLAD
Treatment of AMR
Treatment is multimodal and targets multiple points of antibody production/action:
| Therapy | Mechanism | Role |
|---|---|---|
| Plasmapheresis | Removes circulating DSA | First-line, rapid DSA reduction |
| IVIG (1–2 g/kg) | Fc receptor blockade, anti-idiotype antibodies | First-line, combined with plasmapheresis |
| Rituximab (anti-CD20) | Depletes B-cells | Prevents re-synthesis of DSA |
| Bortezomib | Proteasome inhibitor, depletes plasma cells | Refractory AMR |
| Eculizumab | Anti-C5, inhibits complement | Investigational/refractory |
| Carfilzomib | Next-generation proteasome inhibitor | Refractory cases |
4. Chronic Lung Allograft Dysfunction (CLAD)
The umbrella term for chronic rejection. Affects ~50% of patients by 5 years (Harrison's, p. 8150).
Phenotypes
A. Bronchiolitis Obliterans Syndrome (BOS) — Most Common (~70%)
- Obstructive phenotype
- Fibro-inflammatory obliteration of small airways (bronchioles)
- FEV1 decline, air trapping on CT
B. Restrictive Allograft Syndrome (RAS) — ~30%
- Restrictive phenotype
- TLC and FVC decline
- Peripheral/upper lobe fibrosis on CT
- Worse prognosis than BOS
Pathophysiology of CLAD
- Repeated immunological injury → epithelial damage → aberrant repair
- Transforming growth factor-β (TGF-β) drives fibrogenesis
- Lymphocytic bronchiolitis (B2R ACR) and AMR are key precursors
- Non-immunological triggers: GERD, infections (CMV, Pseudomonas), primary graft dysfunction
BOS Staging (ISHLT)
| Stage | FEV1 (% of baseline) |
|---|---|
| BOS 0 | >90% |
| BOS 0-p (potential) | 81–90% and/or decline in FEF25–75 >25% |
| BOS 1 | 66–80% |
| BOS 2 | 51–65% |
| BOS 3 | ≤50% |
CT Findings in CLAD/BOS
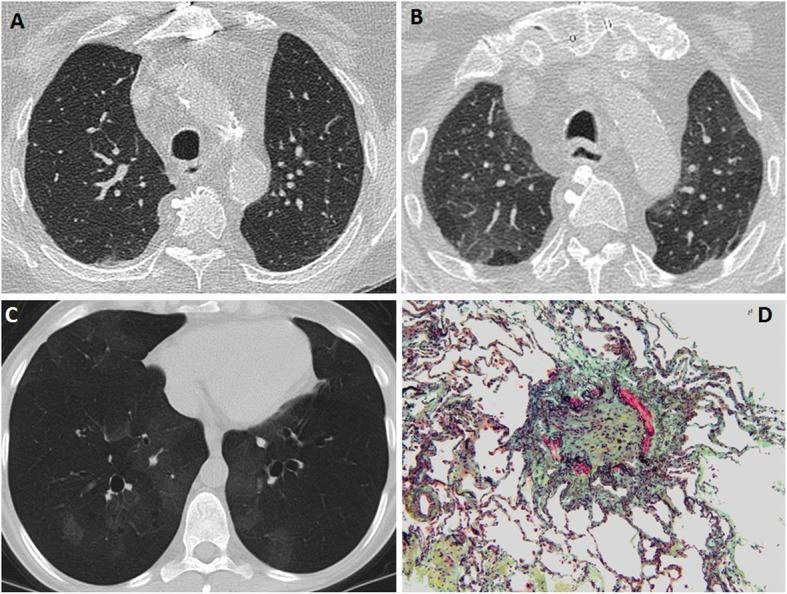
CT findings in BOS: (A) Inspiratory phase — relatively normal; (B) Expiratory phase — geographic air-trapping (pathognomonic); (C) MinIP — mosaic attenuation + bronchiectasis; (D) Histology (Movat pentachrome, 100×) — bronchiole lumen completely obliterated by dense fibrous tissue (green/yellow), confirming obliterative bronchiolitis.
Treatment of CLAD/BOS
| Treatment | Details |
|---|---|
| Azithromycin | Anti-inflammatory, anti-fibrotic; macrolide immunomodulation; may stabilize/improve ~30% of BOS cases |
| Augmented immunosuppression | Pulse steroids, optimize tacrolimus/MMF |
| Photopheresis (ECP) | Effective in steroid-dependent/refractory CLAD |
| Antifungal/antiviral prophylaxis | Treat underlying triggers (CMV, Aspergillus) |
| GERD treatment | Fundoplication if aspiration is contributing |
| Montelukast | Adjunct — modest benefit |
| Pirfenidone | Investigated for RAS (anti-fibrotic) |
| Retransplantation | Only definitive option; controversial due to resource allocation and outcomes |
Maintenance Immunosuppression — The Triple Regimen
All lung transplant recipients receive triple immunosuppression:
| Drug Class | Agent | Mechanism |
|---|---|---|
| Calcineurin inhibitor (CNI) | Tacrolimus (preferred) or Cyclosporine | Inhibits IL-2 transcription (blocks T-cell activation) |
| Antiproliferative | Mycophenolate mofetil (MMF) or Azathioprine | Inhibits purine synthesis → blocks lymphocyte proliferation |
| Corticosteroid | Prednisone (low-dose maintenance) | Broad anti-inflammatory and immunosuppressive |
mTOR inhibitors (Sirolimus, Everolimus): Used in selected patients; some evidence for reducing chronic rejection, but can impair wound healing.
Surveillance & Monitoring
| Tool | Purpose | Schedule |
|---|---|---|
| PFTs (spirometry) | Detect FEV1 decline (BOS staging) | Weekly early post-transplant, then monthly |
| Surveillance bronchoscopy + TBBx | Detect subclinical ACR | At 1, 3, 6, 12 months (program-dependent) |
| DSA monitoring | Detect AMR early | Every 3–6 months |
| CMV PCR | Detect and treat CMV-triggered rejection | Per protocol |
| CT chest | Characterize CLAD phenotype, exclude infection | As clinically indicated |
High-Yield Summary for MD Exam
| Feature | ACR | AMR | CLAD/BOS |
|---|---|---|---|
| Timing | Days–months | Variable | Months–years |
| Mechanism | T-cell | Donor-specific antibodies | Mixed/fibroproliferative |
| Histology | Lymphocytic perivascular infiltrate | Neutrophilic capillaritis + C4d | Obliterative bronchiolitis |
| Diagnosis | TBBx | TBBx + DSA + C4d | PFTs + CT (FEV1 <80% of baseline) |
| First-line treatment | IV methylprednisolone pulse | Plasmapheresis + IVIG | Azithromycin + augmented IS |
| Refractory treatment | ATG, photopheresis | Rituximab, bortezomib | ECP, retransplantation |
| Key risk factor | CMV infection | HLA sensitization | Prior ACR, GERD, infections |
Key exam points:
- ACR graded on ISHLT A (vascular) and B (airway) scales — A2 and above requires treatment
- BOS is the most common form of chronic rejection; diagnosed by FEV1 <80% of baseline on two occasions ≥3 weeks apart with no reversible cause
- Always rule out infection before treating rejection (bronchoscopy + BAL)
- Azithromycin is unique — it has immunomodulatory properties and can stabilize or partially reverse BOS
- Lung transplant rejection rates are higher than other solid organs due to constant alloantigen exposure from the environment
- Retransplantation is the only definitive treatment for end-stage CLAD but remains controversial
Rejection in lung transplant and it's treatment full details for md exam
search_medical_text
lung transplant rejection types hyperacute acute chronic pathophysiology classification ISHLT grading
search_medical_text
lung transplant rejection treatment immunosuppression azithromycin photopheresis retransplantation
search_medical_image
lung transplant rejection bronchiolitis obliterans histology CT imaging
GradeResponse
GradeResponse
GradeResponse
search_medical_image
bronchiolitis obliterans syndrome lung transplant air trapping CT scan
GradeResponse
Here is the complete MD exam-level breakdown on lung transplant rejection.
Lung Transplant Rejection — Complete MD Exam Guide
Why Lungs Are Uniquely Prone to Rejection
The lung is the most immunogenic solid organ for several reasons:
- Continuous exposure to inhaled environmental antigens and pathogens
- Rich resident lymphocyte population and dendritic cell network
- Direct airway communication with the outside world
- High MHC expression on bronchial epithelium and vascular endothelium
~50% of recipients have at least one acute rejection episode in year 1; ~50% develop chronic rejection by 5 years (Harrison's 21st ed., p. 8150). Greater immunosuppression is required compared to other solid organs, which significantly increases drug toxicity risk (Bailey & Love's, p. 1666).
Classification of Rejection
| Type | Onset | Mechanism | Key Pathology |
|---|---|---|---|
| Hyperacute | Minutes–hours | Preformed antibodies | Microvascular thrombosis, hemorrhagic infarction |
| Acute Cellular (ACR) | Days–months | T-cell mediated | Lymphocytic perivascular/peribronchial infiltrate |
| Antibody-Mediated (AMR) | Days–months | Donor-specific antibodies (DSA) | Neutrophilic capillaritis + C4d deposition |
| Chronic (CLAD) | Months–years | Mixed immune + fibroproliferative | Obliterative bronchiolitis or pleuroparenchymal fibrosis |
1. Hyperacute Rejection
Mechanism
- Preformed anti-HLA or anti-ABO antibodies in recipient bind donor endothelium
- Immediate complement activation → neutrophil influx → microvascular thrombosis → hemorrhagic infarction
- Essentially massive antibody-mediated destruction
Clinical Features
- Occurs within minutes to hours of reperfusion
- Catastrophic hypoxemia, bilateral white-out on CXR, graft failure on the table
- Essentially irreversible once established
Treatment & Prevention
| Approach | Details |
|---|---|
| Prevention | Prospective crossmatch, ABO compatibility testing before transplant |
| Plasmapheresis | Emergent removal of circulating antibodies |
| IVIG | Fc receptor blockade |
| Retransplantation | Only salvage option; prognosis extremely poor |
2. Acute Cellular Rejection (ACR)
Pathophysiology
- CD4+ and CD8+ T-lymphocytes recognize donor MHC via:
- Direct pathway: recipient T-cells recognize intact donor MHC on donor APCs
- Indirect pathway: recipient T-cells recognize processed donor peptides on self-APCs
- Activated T-cells release IL-2, IFN-γ → lymphocytic infiltration of perivascular and peribronchial tissue
- CMV infection is the strongest trigger — upregulates MHC expression and co-stimulatory molecules (Harrison's, p. 8150)
- ACR is a major independent risk factor for CLAD
Clinical Features
- Fever, malaise, dyspnea, decreased exercise tolerance
- Decline in FEV1 (≥10–15% from personal best)
- Hypoxemia, new bilateral infiltrates on CXR/CT
- Often asymptomatic — detected only on surveillance bronchoscopy
ISHLT Histological Grading of ACR
A-Grade: Perivascular Rejection
| Grade | Histology |
|---|---|
| A0 | No rejection |
| A1 (Minimal) | Scattered mononuclear cells around 3+ vessels; no eosinophils or endothelialitis |
| A2 (Mild) | Frequent perivascular cuffing (2–3 cell layers); eosinophils may be present |
| A3 (Moderate) | Dense perivascular infiltrate + extension into alveolar septa and air spaces |
| A4 (Severe) | Diffuse perivascular, interstitial, and alveolar infiltrates with necrosis and hyaline membranes |
A2 and above requires treatment. A1 may be treated depending on clinical context.
B-Grade: Lymphocytic Bronchiolitis (Airway Rejection)
| Grade | Histology |
|---|---|
| B0 | No airway inflammation |
| B1R (Low-grade) | Scattered mononuclear cells in bronchiolar submucosa |
| B2R (High-grade) | Dense mononuclear infiltrate with epithelial damage/necrosis |
| BX | Ungradeable (inadequate tissue) |
Diagnosis of ACR
| Investigation | Findings |
|---|---|
| Transbronchial biopsy (TBBx) | Gold standard — perivascular lymphocytic infiltrate |
| BAL | Rule out infection first (always) |
| PFTs | FEV1 decline ≥10–15% from baseline |
| CT chest | Ground-glass opacities, septal thickening, consolidation |
| CXR | Bilateral infiltrates, pleural effusion |
Treatment of ACR
First-Line
- IV methylprednisolone 500–1000 mg/day × 3 days — response rate ~80–90%
- Followed by oral prednisone taper
After Pulse Steroids
- Optimize baseline immunosuppression:
- Check/increase tacrolimus trough levels (target 10–15 ng/mL early)
- Maximize mycophenolate mofetil (MMF) dose
Steroid-Refractory ACR
| Agent | Mechanism |
|---|---|
| Antithymocyte globulin (ATG) | Polyclonal antibody — T-cell depletion |
| Total lymphoid irradiation (TLI) | Depletes lymphocytes in lymph nodes/spleen |
| Photopheresis (ECP) | Extracorporeal photochemotherapy — induces T-cell apoptosis |
| Rituximab | Anti-CD20 — B-cell depletion (if AMR component) |
3. Antibody-Mediated Rejection (AMR)
Pathophysiology
- Donor-specific antibodies (DSA) against HLA Class I and II (detected by Luminex single-antigen bead assay)
- DSA bind endothelium → activate complement cascade → C4d deposition in alveolar capillaries → neutrophilic capillaritis → microvascular injury (Harrison's, p. 8150)
- Can be de novo (post-transplant sensitization) or from pre-formed antibodies
ISHLT 2016 Diagnostic Criteria for AMR (all required):
- Allograft dysfunction (PGD, declining FEV1, or new infiltrates)
- DSA positivity (circulating anti-HLA antibodies)
- Histology: neutrophilic capillaritis ± diffuse alveolar damage
- C4d deposition in alveolar capillaries (immunofluorescence or IHC)
Treatment of AMR — Multimodal Strategy
| Therapy | Mechanism | Role |
|---|---|---|
| Plasmapheresis (5–10 sessions) | Removes circulating DSA | First-line; rapid DSA reduction |
| IVIG (1–2 g/kg) | Fc receptor blockade; anti-idiotype neutralization | First-line; combined with plasmapheresis |
| Rituximab (375 mg/m²) | Anti-CD20; depletes B-cells | Prevents DSA re-synthesis |
| Pulse corticosteroids | Broad immunosuppression | Adjunct |
| Bortezomib | Proteasome inhibitor; depletes long-lived plasma cells | Refractory AMR |
| Carfilzomib | Next-gen proteasome inhibitor | Refractory AMR |
| Eculizumab | Anti-C5; blocks terminal complement | Investigational/severe refractory |
| Tocilizumab | Anti-IL-6; reduces DSA production | Emerging evidence |
4. Chronic Lung Allograft Dysfunction (CLAD)
CLAD is the umbrella term for chronic rejection. Defined as a persistent ≥20% decline in FEV1 from baseline (average of two best post-transplant values) on two occasions ≥3 weeks apart, with no reversible cause.
Two Major Phenotypes
| Feature | BOS (Bronchiolitis Obliterans Syndrome) | RAS (Restrictive Allograft Syndrome) |
|---|---|---|
| Frequency | ~70% of CLAD | ~30% of CLAD |
| Physiology | Obstructive (FEV1↓, FVC preserved) | Restrictive (TLC↓, FVC↓) |
| Histology | Obliterative bronchiolitis (fibrous luminal occlusion) | Pleuroparenchymal fibroelastosis (PPFE) |
| CT pattern | Air trapping, mosaic attenuation, bronchiectasis | Upper lobe fibrosis, pleural thickening |
| Prognosis | Poor | Worse than BOS |
BOS Staging (ISHLT)
| Stage | FEV1 (% of best post-transplant baseline) |
|---|---|
| BOS 0 | >90% |
| BOS 0-p (at risk) | 81–90% AND/OR FEF25–75 decline >25% |
| BOS 1 | 66–80% |
| BOS 2 | 51–65% |
| BOS 3 | ≤50% |
CT Findings in BOS / CLAD
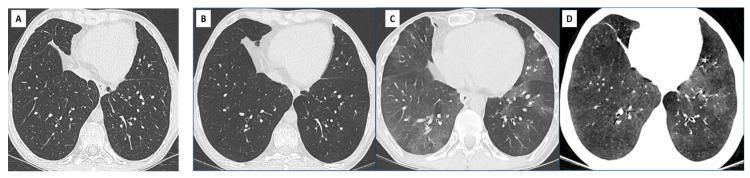
Axial CT in BOS: (A) Baseline inspiratory — normal parenchyma; (B) 2 years later inspiratory — global hypoperfusion, decreased parenchymal density, attenuated vasculature; (C) Expiratory CT — mosaic attenuation and heterogeneous air trapping (hallmark of small airway disease); (D) MinIP reconstruction — enhances geographic mosaic perfusion. Key teaching point: expiratory CT or MinIP is essential as inspiratory scans can appear deceptively normal in early BOS.
Pathogenesis of CLAD
Alloimmune injury (ACR, AMR)
↓
Epithelial and endothelial damage
↓
Aberrant repair → TGF-β↑, fibroblast activation
↓
Submucosal fibrosis → luminal obliteration (BOS)
OR
Pleuroparenchymal fibrosis (RAS)
Non-immunological triggers amplifying CLAD:
- GERD with microaspiration (very important)
- CMV, Pseudomonas, Aspergillus infections
- Primary graft dysfunction (PGD)
- Air pollution exposure
Treatment of CLAD/BOS
| Treatment | Details |
|---|---|
| Azithromycin 250 mg 3×/week | Macrolide with immunomodulatory + anti-neutrophilic properties; stabilizes/improves ~30% of BOS cases; first-line |
| Augmented immunosuppression | Pulse steroids, optimize tacrolimus + MMF |
| Photopheresis (ECP) | Most evidence-based treatment for progressive BOS; stabilizes decline |
| mTOR inhibitors (Sirolimus/Everolimus) | Switch from CNI; some anti-fibrotic and anti-proliferative benefit |
| Montelukast | Leukotriene receptor antagonist; modest adjunct benefit |
| Pirfenidone | Anti-fibrotic; investigated particularly for RAS phenotype |
| GERD treatment | Fundoplication if microaspiration confirmed — can slow CLAD progression |
| Antimicrobial optimization | Treat CMV, Pseudomonas, Aspergillus aggressively |
| Retransplantation | Only definitive treatment; controversial due to resource allocation and inferior outcomes vs. primary transplant |
Maintenance Immunosuppression — Triple Therapy
All lung transplant recipients receive triple immunosuppression (Bailey & Love's, p. 1666):
| Pillar | Drug | Mechanism |
|---|---|---|
| Calcineurin inhibitor | Tacrolimus (preferred) or Cyclosporine | Blocks calcineurin → ↓IL-2 transcription → T-cell activation blockade |
| Antiproliferative | MMF (preferred) or Azathioprine | Inhibits inosine monophosphate dehydrogenase → ↓purine synthesis → blocks lymphocyte proliferation |
| Corticosteroid | Prednisolone (low-dose maintenance) | Broad anti-inflammatory; inhibits cytokine transcription |
Induction therapy (peri-operative):
- Basiliximab (anti-IL-2Rα/CD25) — most common; IL-2 receptor blockade
- ATG (antithymocyte globulin) — T-cell depletion; used in higher-risk patients
- Not universally applied; significant infection/malignancy risk limits use despite survival benefit (Bailey & Love's, p. 1666)
mTOR inhibitors (Sirolimus, Everolimus):
- Block mTOR → ↓T-cell and B-cell proliferation
- Used as CNI-sparing agents (nephroprotective)
- Some evidence for reducing CLAD progression
- Contraindicated early post-operatively — impairs bronchial anastomotic healing
Surveillance Protocol Post-Transplant
| Tool | Purpose | Timing |
|---|---|---|
| PFTs (spirometry) | FEV1 monitoring for BOS staging | Weekly early, then monthly |
| Surveillance bronchoscopy + TBBx | Detect subclinical ACR | 1, 3, 6, 12 months (program-dependent) |
| BAL | Infection exclusion at each bronchoscopy | With every bronchoscopy |
| DSA monitoring (Luminex) | Early AMR detection | Every 3–6 months |
| CMV PCR | CMV-triggered rejection prevention | Per protocol |
| CT chest | CLAD phenotyping, infection exclusion | As clinically indicated |
High-Yield Comparison Table
| Feature | ACR | AMR | CLAD/BOS |
|---|---|---|---|
| Timing | Days–months (peak: year 1) | Variable | Months–years |
| Mechanism | CD4/CD8 T-cells | DSA + complement | Mixed fibroproliferative |
| Histology | Perivascular lymphocytic infiltrate | Neutrophilic capillaritis + C4d | Obliterative bronchiolitis |
| Key marker | A-grade on TBBx | C4d + DSA | FEV1 <80% baseline |
| Gold standard Dx | Transbronchial biopsy | TBBx + DSA + C4d | PFTs + CT (exclusion of reversible cause) |
| First-line Rx | IV methylprednisolone pulse | Plasmapheresis + IVIG | Azithromycin + augmented IS |
| Refractory Rx | ATG, photopheresis | Rituximab, bortezomib | ECP, retransplantation |
| Key risk factor | CMV infection | Prior sensitization/HLA mismatch | Prior ACR, GERD, CMV, PGD |
Key Exam Points to Remember
- Always exclude infection before treating rejection — BAL at bronchoscopy is mandatory; infection and rejection can coexist and mimic each other
- ACR grade A2 and above always requires treatment; grade A1 may be observed or treated based on clinical context
- BOS is the most common chronic rejection form (~70% of CLAD); diagnosed by FEV1 <80% of best post-transplant value on two occasions ≥3 weeks apart
- Azithromycin is the cornerstone of BOS treatment due to its immunomodulatory (not just antimicrobial) properties
- Lungs require more aggressive immunosuppression than other solid organs, increasing risks of infection, nephrotoxicity, diabetes, hypertension, and malignancy
- CMV infection is the single most important trigger for ACR
- GERD/microaspiration is a key modifiable non-immunological risk factor for CLAD — fundoplication may slow progression
- mTOR inhibitors must be avoided immediately post-transplant (impair anastomotic healing) but have a role later as CNI-sparing/anti-fibrotic agents
- RAS phenotype carries a worse prognosis than BOS and does not respond as well to standard BOS therapies
- Retransplantation is the only definitive treatment for end-stage CLAD but outcomes are inferior to primary transplant
This is a shared conversation. Sign in to Orris to start your own chat.