What is apoptosis notes for concept plua
Reading File
Finding Sources
Searching the Web
apoptosis mechanism pathways notes medical
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
I now have comprehensive content from Robbins & Kumar Basic Pathology and Sleisenger & Fordtran's GI textbook. Here are complete concept notes on apoptosis:
Apoptosis — Concept Notes
Definition
Apoptosis is a pathway of programmed cell death in which cells activate enzymes (caspases) that degrade their own nuclear DNA and cytoplasmic/nuclear proteins. The plasma membrane remains intact throughout the process. Fragments (apoptotic bodies) are rapidly phagocytosed by macrophages — crucially, no inflammatory reaction is triggered, distinguishing it from necrosis.
"The plasma membrane of the apoptotic cell remains intact, but… the fragments, called apoptotic bodies, are recognized and rapidly phagocytosed by macrophages." — Robbins & Kumar Basic Pathology
Apoptosis vs. Necrosis
| Feature | Apoptosis | Necrosis |
|---|---|---|
| Cell membrane | Intact | Disrupted |
| Inflammation | No | Yes |
| Cell size | Shrinks | Swells |
| DNA fragmentation | Internucleosomal (ladder) | Random |
| Energy requirement | Active (ATP-dependent) | Passive |
| Trigger | Programmed / controlled | Uncontrolled injury |
Causes of Apoptosis
Physiologic
| Condition | Mechanism |
|---|---|
| Embryogenesis | Loss of growth factor signaling |
| Tissue turnover (intestinal epithelium, lymphocytes) | Loss of survival signals |
| Hormone-dependent involution (e.g., endometrium) | Decreased hormone levels |
| End of immune/inflammatory responses | Loss of leukocyte survival signals |
| Elimination of self-reactive lymphocytes | Strong self-antigen recognition activates both pathways |
Pathologic
| Condition | Mechanism |
|---|---|
| DNA damage (radiation, cytotoxic drugs) | Activation of proapoptotic BH3-only proteins |
| Misfolded protein accumulation (ER stress) | Activation of BH3-only proteins; possibly direct caspase activation |
| Viral infections | Viral proteins activate caspases; CTLs kill infected cells |
Mechanisms: Two Pathways
Both pathways converge on caspase activation — cysteine proteases that cleave after aspartate residues.
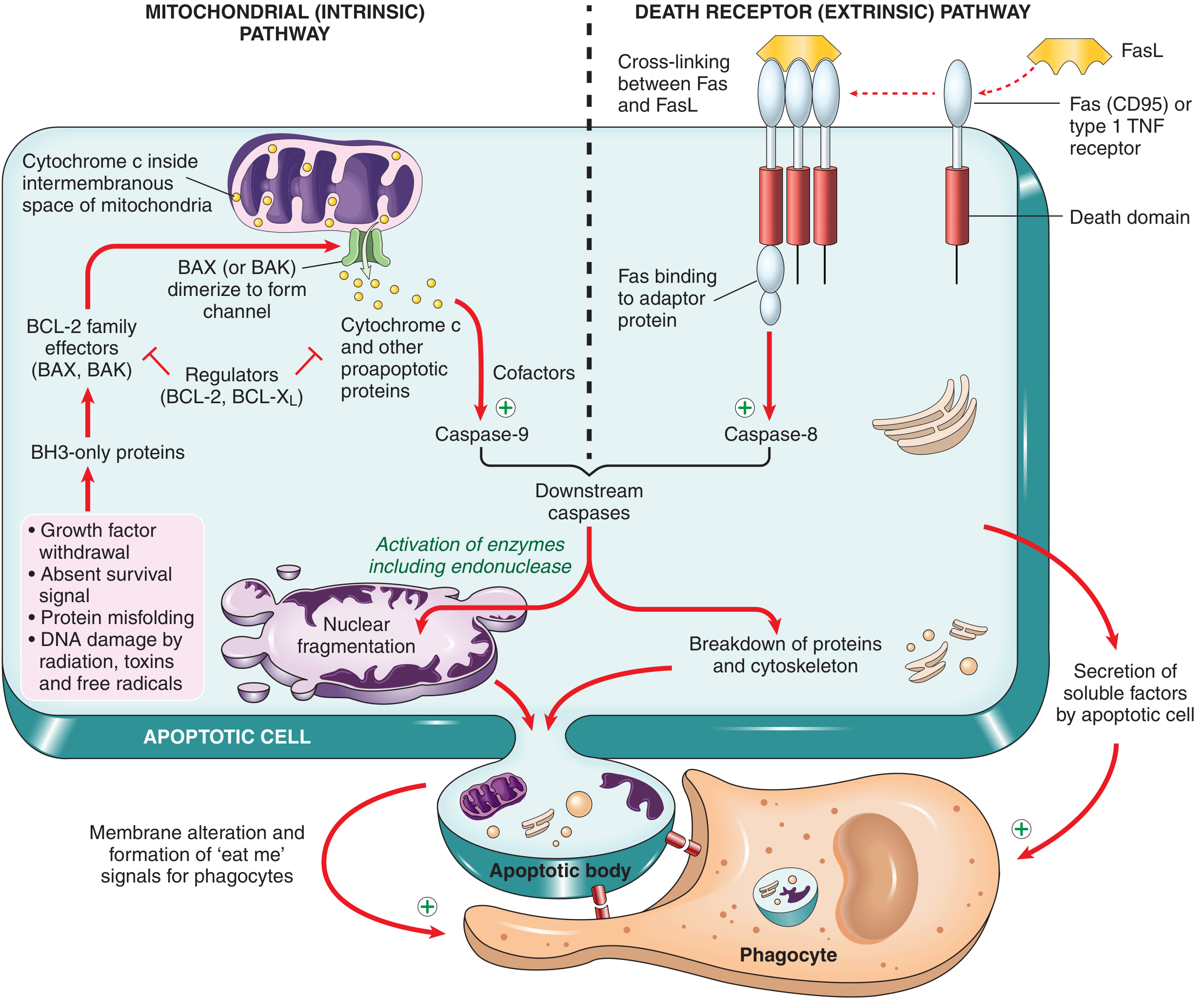
1. Mitochondrial (Intrinsic) Pathway
Responsible for most physiologic and pathologic apoptosis.
Steps:
- Stress signals (growth factor withdrawal, DNA damage, misfolded proteins, radiation, free radicals) activate BH3-only proteins (sensors).
- BH3-only proteins shift the balance away from antiapoptotic BCL-2/BCL-XL toward proapoptotic BAX/BAK.
- BAX and BAK dimerize, insert into the mitochondrial outer membrane, and form channels.
- Cytochrome c leaks from the intermembranous space into the cytosol.
- Cytochrome c + APAF-1 + cofactors → forms the apoptosome.
- The apoptosome activates caspase-9 (initiator caspase).
- Caspase-9 activates caspase-3 / caspase-7 (executioner caspases).
- Executioner caspases cleave downstream targets → cell death and fragmentation.
Key regulators — BCL-2 family:
| Type | Members | Action |
|---|---|---|
| Antiapoptotic | BCL-2, BCL-XL, MCL-1 | Inhibit BAX/BAK; maintain mitochondrial membrane integrity |
| Proapoptotic effectors | BAX, BAK | Form mitochondrial channels → cytochrome c release |
| Proapoptotic sensors | BH3-only proteins (BIM, BID, BAD, PUMA, NOXA) | Neutralize BCL-2/BCL-XL; activate BAX/BAK |
The ratio of antiapoptotic to proapoptotic BCL-2 family members determines whether a cell lives or dies.
2. Death Receptor (Extrinsic) Pathway
Triggered by external signals via cell-surface death receptors.
Key receptors: Fas (CD95) and TNF Receptor 1 (TNFR1) — members of the TNF receptor superfamily with cytoplasmic "death domains."
Steps:
- FasL (expressed on activated T lymphocytes) binds Fas on target cells → Fas cross-linking.
- Death domain recruits adaptor proteins → form the Death-Inducing Signaling Complex (DISC).
- DISC recruits and activates caspase-8 (initiator caspase).
- Caspase-8 activates caspase-3 / caspase-7 (executioner caspases).
- Final result: same as intrinsic pathway — fragmentation and death.
Roles: Elimination of self-reactive lymphocytes; cytotoxic T lymphocyte (CTL)-mediated killing.
Pathway Diagram (from Sleisenger & Fordtran's):
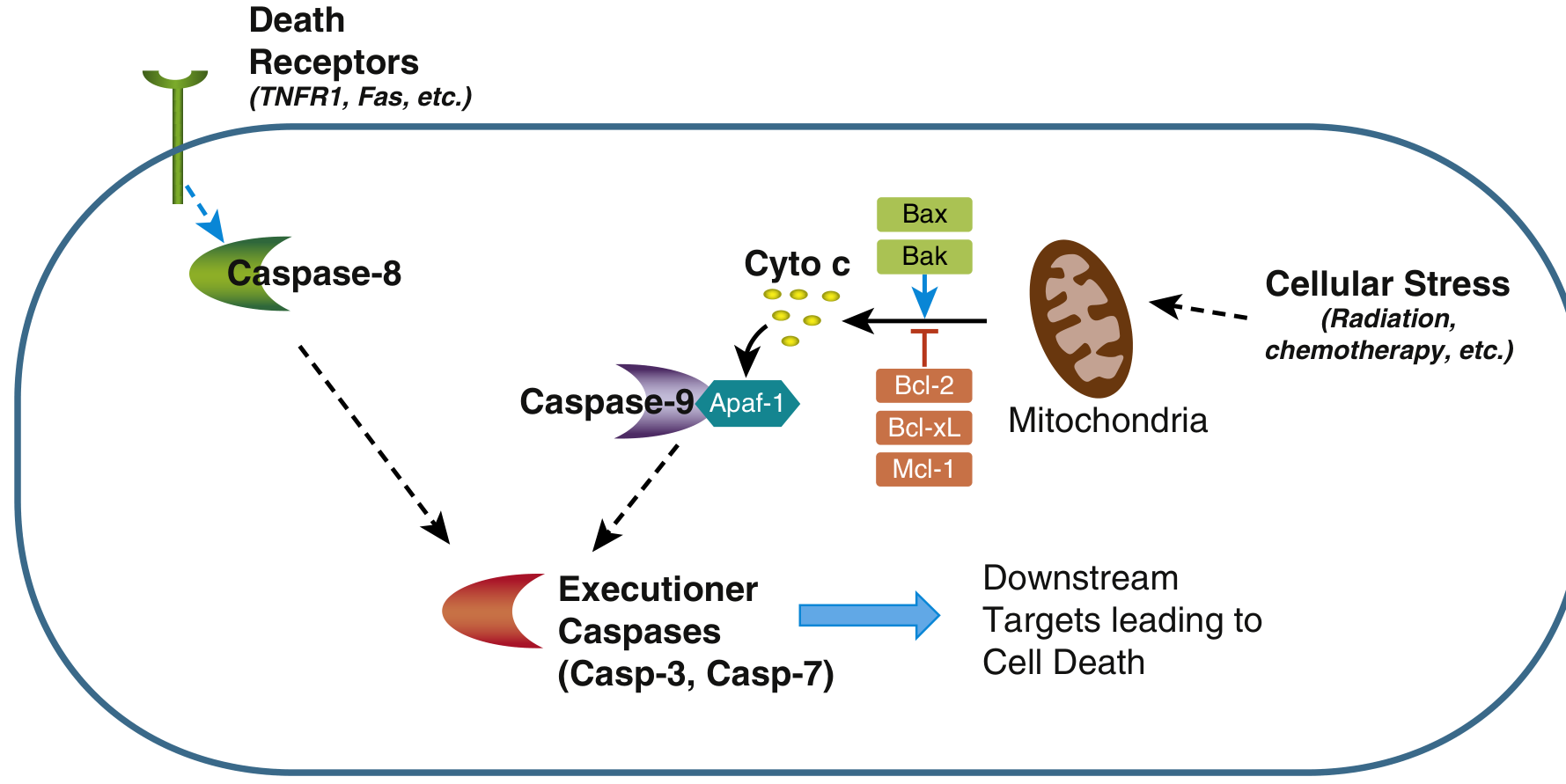
Terminal (Execution) Phase
Both pathways converge here:
- Activated caspase-8 or caspase-9 → activate more caspases
- Executioner caspases (3 & 7) activate:
- Endonucleases → internucleosomal DNA fragmentation (DNA ladder on gel)
- Cytoskeletal and nuclear protein degradation
Morphology of Apoptotic Cells
| Feature | Description |
|---|---|
| Nuclear changes | Chromatin condensation → karyorrhexis (nuclear fragmentation) |
| Cell size | Shrinkage |
| Membrane | Cell buds → apoptotic bodies (membrane-bound fragments of cytosol + organelles) |
| DNA | Fragmented into nucleosome-sized pieces |
| Inflammation | None — cells cleared before contents leak |
Apoptosis can be histologically undetectable even when extensive, because fragments are so rapidly phagocytosed.
Clearance of Apoptotic Cells
- Phosphatidylserine normally on the inner leaflet of plasma membrane flips to the outer leaflet in apoptotic cells → recognized as "eat-me" signal by tissue macrophages.
- Apoptotic cells also secrete soluble chemotactic factors that recruit phagocytes.
- Phagocytosis is efficient, rapid, and non-inflammatory.
Other Cell Death Pathways (for comparison)
| Pathway | Key Feature |
|---|---|
| Necroptosis | TNF-induced; morphology between necrosis and apoptosis |
| Pyroptosis | Inflammasome activation → IL-1 release → inflammation and fever |
| Ferroptosis | Dependent on cellular iron levels |
Clinical Relevance
- Cancer: Escape from apoptosis (e.g., overexpression of BCL-2 in follicular lymphoma t(14;18)) → tumor survival
- Autoimmunity: Defective Fas/FasL pathway → failure to eliminate self-reactive lymphocytes → autoimmune lymphoproliferative syndrome (ALPS)
- Neurodegeneration: Excessive apoptosis of neurons (e.g., in Alzheimer's, Parkinson's)
- HIV: Depletion of CD4+ T cells partly via apoptosis
- Therapy: BCL-2 inhibitors (e.g., venetoclax) exploit apoptosis to treat CLL and AML
Sources: Robbins & Kumar Basic Pathology; Sleisenger and Fordtran's Gastrointestinal and Liver Disease
This is a shared conversation. Sign in to Orris to start your own chat.