All about copd , diagnosis, guidelines , treatment options and complications
Reading File
Finding Sources
Searching PubMed
"chronic obstructive pulmonary disease" AND "guidelines"
Searching the Web
GOLD 2025 COPD guidelines diagnosis treatment
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
I now have all the material needed for a comprehensive response. Here is the full synthesis:
Chronic Obstructive Pulmonary Disease (COPD)
1. Definition & Overview
COPD is a progressive, largely irreversible airflow limitation disorder caused by an abnormal inflammatory response of the lung to noxious particles or gases — principally cigarette smoke. It most commonly manifests as emphysema and/or chronic bronchitis, which frequently coexist. Unlike asthma, the obstruction is not fully reversible with treatment. COPD is now the third leading cause of death worldwide, affecting over 400 million people globally, with an estimated annual global cost projected to reach $50 trillion by 2030.
- Fishman's Pulmonary Diseases and Disorders, p. 696
- Katzung's Basic and Clinical Pharmacology, 16e, p. 565
2. Pathophysiology
Emphysema
- Enlargement of air spaces distal to terminal bronchioles caused by destruction of alveolar elastic support structures by proteases (mainly from neutrophils)
- Centriacinar (centrilobular): most common subtype; smoking-related; affects respiratory bronchioles first
- Panacinar: seen in α₁-antitrypsin (AAT) deficiency; destroys the entire acinus uniformly
- Results in: ↑ lung compliance, barrel chest, air trapping, static and dynamic hyperinflation
- Patients tend to maintain near-normal oxygenation at rest ("Pink Puffers")
- Robbins & Kumar Basic Pathology, p. 496
Chronic Bronchitis
- Clinically defined: persistent productive cough ≥3 months/year for ≥2 consecutive years
- Mucus overproduction from hyperplasia of mucous glands (Reid index > 0.5), goblet cell metaplasia, small airway inflammation (chronic bronchiolitis)
- Airway obstruction from small airway fibrosis, not mucus gland hypertrophy
- MUC5AC concentration ↑ 10-fold, MUC5B ↑ 3-fold in severe COPD
- Persistent infection with Haemophilus influenzae due to impaired mucociliary clearance
- Patients develop hypoxemia and hypercapnia ("Blue Bloaters")
- Fishman's Pulmonary Diseases, p. 143; Robbins, p. 496
Key Pathophysiologic Cascade
- Toxic particles → airway inflammation (neutrophil/macrophage-dominant) → protease-antiprotease imbalance → tissue destruction
- Dynamic hyperinflation during exercise → ↑ end-expiratory lung volume → reduced inspiratory reserve → neuromechanical uncoupling → dyspnea
- Ventilation-perfusion mismatch → hypoxemia → pulmonary vasoconstriction → pulmonary hypertension
3. Risk Factors
| Factor | Details |
|---|---|
| Cigarette smoking | Primary risk factor; only 15–30% of smokers develop COPD (though recent CT data suggest even more have subclinical disease) |
| Age + cumulative smoke exposure | Two most important risk factors in epidemiologic studies |
| α₁-antitrypsin deficiency | Panacinar emphysema, especially in never-smokers |
| Biomass smoke | Wood, coal, charcoal combustion — major cause in women/children in developing nations |
| Occupational dusts/chemicals | Coal mining, cotton dust, silica |
| Recurrent childhood infections | Impair lung growth; increase lifetime COPD risk |
| Preterm birth / low birth weight | Reduced maximal attained lung function |
4. Diagnosis
Clinical Presentation
- Symptoms: progressive exertional dyspnea, chronic cough (productive or dry), wheeze, sputum production
- Signs: barrel chest, hyperresonance to percussion, diminished breath sounds, prolonged expiration, use of accessory muscles, pursed-lip breathing
- COPD is underdiagnosed — >50% of patients with spirometric COPD lack a formal diagnosis; patients attribute symptoms to aging or smoking
Spirometry (Gold Standard)
Post-bronchodilator spirometry (400 µg albuterol) is required for a definitive diagnosis.
Diagnostic criterion: FEV₁/FVC < 0.70 post-bronchodilator
- The fixed ratio (0.70) is the GOLD-recommended threshold — simple and universally applicable, though it may overdiagnose in elderly and underdiagnose younger patients
- ATS/ERS alternative: use the lower limit of normal (LLN) — the 5th percentile for age, avoiding age-related overdiagnosis
- Flow-volume loop: concave ("scooped-out") appearance; volume-time curve shows prolonged expiratory time
- Murray & Nadel's Textbook of Respiratory Medicine, p. 1471; Fishman's, p. 696
GOLD Spirometric Severity Grades (post-bronchodilator FEV₁ % predicted)
| GOLD Grade | Severity | FEV₁ % predicted |
|---|---|---|
| GOLD 1 | Mild | ≥ 80% |
| GOLD 2 | Moderate | 50–79% |
| GOLD 3 | Severe | 30–49% |
| GOLD 4 | Very severe | < 30% |
GOLD ABE Symptom/Risk Groups (updated from ABCD)
Current GOLD guidelines move beyond pure spirometry by combining symptom burden (mMRC dyspnea score or CAT score) and exacerbation history:
- Group A: 0–1 exacerbations (not hospitalised), low symptoms
- Group B: 0–1 exacerbations (not hospitalised), high symptoms
- Group E: ≥2 exacerbations OR ≥1 hospitalisation (high exacerbation risk)
Additional Investigations
- CXR: hyperinflation, flat diaphragms, increased AP diameter, bullae — the PA chest radiograph below shows a typical COPD pattern
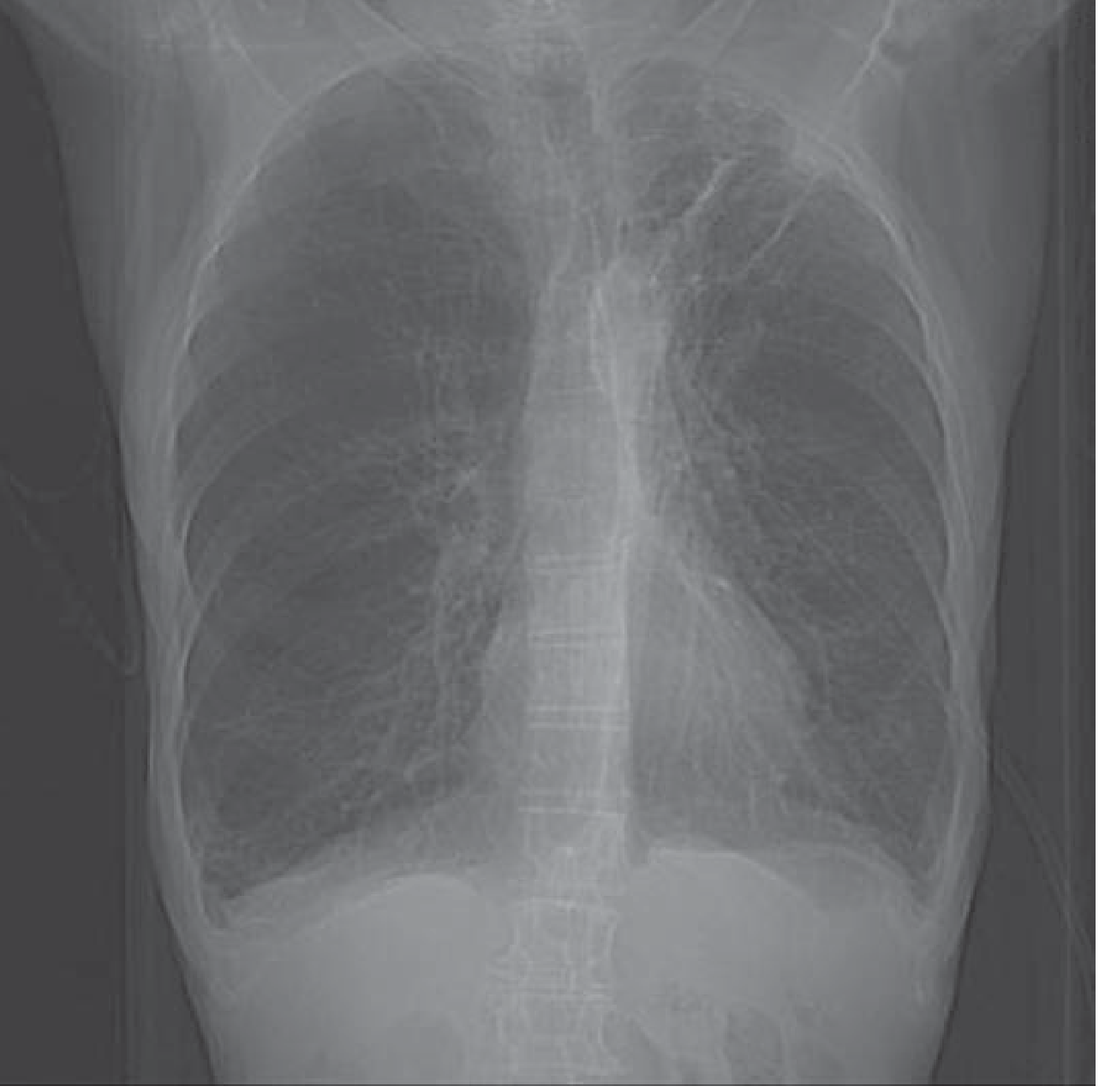
- CT chest: best for characterising emphysema subtype, airway wall thickening, bullae; small airway mucus occlusion correlates with degree of airflow obstruction
- ABG: hypoxemia, hypercapnia in advanced disease; type II respiratory failure
- BNP/NT-proBNP: helps distinguish COPD exacerbation from acute heart failure (BNP <100 pg/mL favours COPD; >500 favours HF)
- α₁-antitrypsin level: check in patients <45 years, non-smokers, or family history
- ECG: may show P pulmonale, right axis deviation, RV hypertrophy if cor pulmonale present
- BODE Index: multidimensional prognostic index (BMI, Obstruction, Dyspnea, Exercise) — BODE 7–10 carries 80% 52-month mortality
5. Treatment
5a. Stable COPD — Pharmacotherapy
Pharmacotherapy does not alter disease progression but reduces symptoms, controls exacerbations, improves quality of life, and exercise performance.
Bronchodilators (Cornerstone of Therapy)
Short-Acting (SABA / SAMA) — rescue
- SABA: albuterol (salbutamol), levalbuterol
- SAMA: ipratropium bromide (M3-antagonist)
- Combination: fenoterol/ipratropium, salbutamol/ipratropium
Long-Acting (LABA / LAMA) — maintenance
| Class | Examples |
|---|---|
| LABA | Salmeterol, formoterol, indacaterol, olodaterol, vilanterol |
| LAMA | Tiotropium, aclidinium, umeclidinium, glycopyrronium |
| LABA+LAMA | Indacaterol/glycopyrronium, umeclidinium/vilanterol, tiotropium/olodaterol |
Long-acting anticholinergics (LAMA) are preferred over short-acting for maintenance. Combining LABA+LAMA provides greater bronchodilation without a significant increase in side effects.
Inhaled Corticosteroids (ICS)
- Less central in COPD than asthma; associated with increased risk of bacterial pneumonia
- Indicated when: FEV₁ < 50% predicted, frequent exacerbations, overlap with asthma features
- Blood eosinophil count guides ICS use: high eosinophils (≥300 cells/µL) → likely to benefit; low eosinophils → benefit unlikely
- ICS combinations: formoterol/budesonide, salmeterol/fluticasone, formoterol/mometasone, vilanterol/fluticasone
Triple Therapy (ICS + LABA + LAMA)
- For high-risk patients (Group E + FEV₁ <50% + frequent exacerbations + high eosinophils)
Other Agents
| Drug | Mechanism | Use |
|---|---|---|
| Roflumilast | Selective PDE-4 inhibitor | Severe COPD (FEV₁ <50%) with chronic bronchitis + frequent exacerbations; ↓ exacerbation frequency |
| Azithromycin | Macrolide antibiotic + anti-inflammatory | Daily use reduces exacerbations in older patients/milder COPD |
| Theophylline | Non-selective PDE inhibitor | Limited role; recent large RCT showed no benefit on exacerbation frequency at low doses |
Katzung's, p. 565; Tintinalli's Emergency Medicine, p. 509
5b. Non-Pharmacological Treatment
| Intervention | Details |
|---|---|
| Smoking cessation | Only intervention that reduces both rate of FEV₁ decline AND respiratory mortality |
| Long-term oxygen therapy (LTOT) | Criteria: PaO₂ ≤55 mmHg, SaO₂ ≤88%, OR PaO₂ 56–59 mmHg with cor pulmonale/polycythemia; goal SaO₂ ≥90%, PaO₂ ≥60 mmHg; flow rates 1–3 L/min typically |
| Pulmonary rehabilitation | Improves exercise capacity and quality of life in moderate–severe COPD; evidence-based per ATS 2023 guidelines |
| Vaccination | Influenza annually; pneumococcal vaccination; COVID-19; pertussis |
| Secretion mobilization | Hydration, humidification; mucolytics (NAC) have marginal benefit; avoid antihistamines/antitussives |
| Surgical options | Lung volume reduction surgery (LVRS) in upper-lobe emphysema; bronchoscopic valve placement; lung transplantation in end-stage disease |
5c. Managing Acute Exacerbations of COPD (AECOPD)
Definition: Acute worsening of respiratory symptoms (dyspnea, cough, sputum) beyond normal day-to-day variation, requiring a change in therapy.
Triggers (>75% have viral or bacterial infection):
- Viral URTIs (rhinovirus most common), bacterial (H. influenzae, Streptococcus pneumoniae, Moraxella catarrhalis)
- Air pollution, cold weather, β-blockers, opioids/sedatives, GERD
Pathophysiology: inflammatory mediators → bronchoconstriction + mucus hypersecretion + pulmonary vasoconstriction → ↑ work of breathing → hypercapnia → respiratory acidosis. Mechanism is V/Q mismatch (not expiratory flow limitation as in asthma).
Emergency Management:
| Treatment | Details |
|---|---|
| Controlled O₂ | Target SaO₂ 88–92% (avoid hypoxic drive suppression); Venturi mask preferred |
| SABA ± SAMA | Nebulized albuterol ± ipratropium; ↑ dose and frequency |
| Systemic corticosteroids | Prednisone 40 mg/day × 5 days; reduces treatment failure, length of stay |
| Antibiotics | Indicated when purulent sputum or severe exacerbation; β-lactams, doxycycline, azithromycin (active vs. H. influenzae) |
| Non-invasive ventilation (NIV/BIPAP) | First-line for hypercapnic respiratory failure (pH <7.35, PaCO₂ >45); reduces intubation rate and mortality |
| Invasive ventilation | If NIV fails or contraindicated |
| Heliox | Helium-oxygen mixture; reduces airway resistance in severe obstruction |
Clinical signs of severe exacerbation: use of accessory muscles, pulsus paradoxus, pursed-lip breathing, inability to speak in full sentences, altered consciousness.
6. Complications
| Complication | Details |
|---|---|
| Pulmonary hypertension | Chronic hypoxemia → pulmonary vasoconstriction → vascular remodeling → ↑ pulmonary arterial pressure |
| Cor pulmonale | Right ventricular hypertrophy/failure secondary to pulmonary hypertension; signs: JVD, peripheral edema, hepatomegaly |
| Chronic respiratory failure | Type II (hypercapnic) in advanced disease; requires LTOT or domiciliary NIV |
| Polycythemia | Secondary to chronic hypoxemia; ↑ erythropoietin → ↑ RBC mass |
| Recurrent pneumonia | Impaired mucociliary clearance and mucus plugging; risk ↑ with ICS use |
| Spontaneous pneumothorax | Rupture of bullae/blebs; life-threatening in severe COPD |
| Acute exacerbations | Major driver of morbidity, mortality, and healthcare costs; mortality exceeds asthma exacerbations due to older age and comorbidities |
| Muscle wasting / cachexia | Systemic inflammation, disuse atrophy; contributes to exercise intolerance |
| Cardiovascular disease | Leading cause of death in mild-moderate COPD; systemic inflammation, shared risk factors with atherogenesis |
| Depression and anxiety | Highly prevalent; worsens quality of life and increases exacerbation frequency |
| Sleep-disordered breathing | Overlap syndrome (COPD + OSA) → worse nocturnal hypoxemia, higher mortality |
| Lung cancer | ↑ risk independent of smoking history |
Swanson's Family Medicine Review; Barash Clinical Anesthesia, 9e, p. 361
7. Prognosis — BODE Index
| BODE Score | 52-Month Mortality |
|---|---|
| 0–2 | 19% |
| 3–4 | 32% |
| 5–6 | 40% |
| 7–10 | 80% |
BODE = BMI + airflow Obstruction (FEV₁%) + Dyspnea (mMRC) + Exercise capacity (6-min walk distance)
- Murray & Nadel's, p. 1471
Summary Algorithm
Symptoms (dyspnea, cough, sputum) + Risk Factors (smoking, exposure)
↓
Post-bronchodilator Spirometry → FEV₁/FVC < 0.70 → COPD confirmed
↓
GOLD Grade (1–4 by FEV₁%) + GOLD Group (A/B/E by symptoms + exacerbations)
↓
Stable Management:
Group A → SABA/SAMA prn
Group B → LAMA or LABA (or LABA+LAMA)
Group E → LABA+LAMA ± ICS (if eos ≥300 or FEV₁<50%)
↓
Non-pharmacologic: Smoking cessation · O₂ if hypoxic · Pulmonary rehab · Vaccination
↓
Exacerbation: O₂ (88–92%) → SABA+SAMA → Steroids → Antibiotics → NIV → Invasive MV
Key Sources:
- Fishman's Pulmonary Diseases and Disorders, 2-Volume Set
- Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume
- Katzung's Basic and Clinical Pharmacology, 16e
- Robbins & Kumar Basic Pathology (Robbins Pathology)
- Tintinalli's Emergency Medicine
- The Washington Manual of Medical Therapeutics
- GOLD 2024/2025 Guidelines (goldcopd.org)
This is a shared conversation. Sign in to Orris to start your own chat.