Aortic dissection
Reading File
Finding Sources
Searching Images
aortic dissection classification CT imaging
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Now I have comprehensive information to provide a thorough answer. Let me compile this.
Aortic Dissection
Definition & Pathogenesis
Aortic dissection occurs when the laminar planes of the media split apart, forming a blood-filled false lumen within the aortic wall. The initiating event is either a primary intimal tear with secondary dissection into the media, or medial hemorrhage that disrupts the intima. Once the tear occurs, blood under systemic pressure dissects along elastic lamellar plates, typically between the middle and outer thirds of the media. — Robbins & Kumar Basic Pathology, p. 325; Harrison's Principles of Internal Medicine 22E, p. 2212
The most common sites of intimal tear:
- Ascending aorta, within 10 cm of the aortic valve (right lateral wall, where hydraulic shear stress is highest)
- Descending thoracic aorta, just distal to the ligamentum arteriosum
The dissection propagates distally (and sometimes retrograde toward the heart) into major branches, causing malperfusion syndrome — organ ischemia from compromised branch vessel flow.
Classification
Two systems are in common use:
Stanford Classification (most clinically practical):
| Type | Involvement | Management |
|---|---|---|
| A | Involves ascending aorta (with or without extension distally) | Surgical emergency |
| B | Does not involve ascending aorta (limited to arch/descending aorta) | Medical management (unless complicated) |
DeBakey Classification:
| Type | Description |
|---|---|
| I | Originates in ascending aorta, extends to arch and typically descending aorta and beyond |
| II | Confined to ascending aorta only |
| III | Originates in descending aorta, usually just distal to left subclavian artery, extends distally |
DeBakey I & II = Stanford A; DeBakey III = Stanford B
A newer SVS/STS classification combines Stanford A/B with endovascular aortic zone numbering to guide intervention planning, more precisely defining arch and zone involvement.
Temporal classification: hyperacute (<24 h), acute (24 h–14 days), subacute (15–90 days), chronic (>90 days). — Current Surgical Therapy 14e
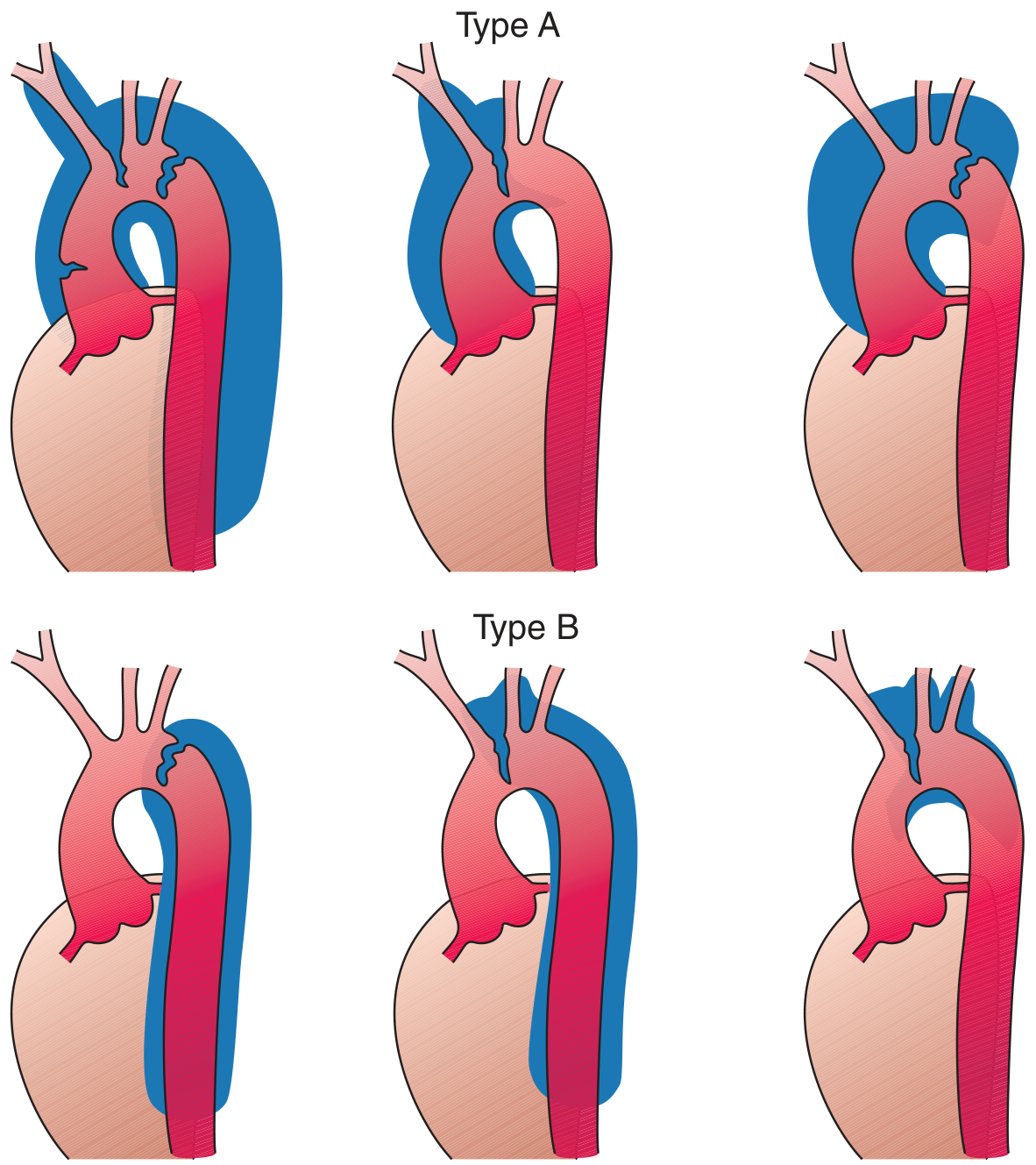
Stanford Type A (top) vs. Type B (bottom); DeBakey Types I, II, III. — Harrison's Principles of Internal Medicine 22E, p. 2213
Risk Factors
- Hypertension — present in >70% of cases; the dominant risk factor
- Connective tissue disorders: Marfan syndrome (major cause in young patients), Ehlers-Danlos syndrome type IV, Loeys-Dietz syndrome
- Bicuspid aortic valve, coarctation of the aorta
- Inflammatory aortitis: Takayasu's arteritis, giant cell arteritis
- Cocaine use — abrupt BP surge; implicated in 0.5–37% of cases
- Pregnancy (third trimester) — hormone-induced vascular remodeling
- Iatrogenic: arterial cannulation, cardiopulmonary bypass
- Atherosclerosis (interestingly, extensive atherosclerosis can arrest propagation by causing medial fibrosis)
Peak incidence: 6th–7th decade; men > women 2:1 (mean age 63 vs. 67 years in IRAD). — Braunwald's Heart Disease
Clinical Presentation
- Classic: sudden, severe, "tearing" or "ripping" chest pain (10/10), often radiating to the back between the scapulae — the most common initial symptom
- Atypical presentations: syncope, acute neurologic deficit (stroke, paraplegia), pulse or BP asymmetry between extremities, acute limb ischemia, abdominal pain, renal insufficiency, new diastolic murmur (aortic regurgitation from retrograde extension)
- Type A: may cause cardiac tamponade, acute aortic regurgitation, coronary ostium involvement → MI, CHF
- Older patients: chest pain less common; syncope, CVA, or heart failure may predominate; hypotension at presentation carries ominous prognosis
- "Sense of impending doom" is a recognizable feature
Key warning signs: differential blood pressures/pulse deficits in extremities, wide mediastinum on CXR, ECG without ischemic changes in a patient with tearing chest pain.
Diagnosis
CT Angiography (CTA) — gold standard; sensitivity and specificity 90–100%; should cover entire aorta to iliofemorals to evaluate for malperfusion. Most widely available.

CTA showing intimal flap separating true and false lumens in ascending (green arrow) and descending (red arrow) aorta — Stanford Type A.
Transesophageal Echocardiography (TEE): ~98% sensitivity, ~90% specificity for ascending and descending aorta (not arch); also evaluates tamponade, AR, and wall motion. Preferred in hemodynamically unstable patients going directly to OR.
Transthoracic Echo (TTE): sensitivity 60–85%; >80% for proximal ascending; limited for arch/descending.
MRI: >90% sensitivity/specificity; less practical acutely.
Chest X-ray: widened mediastinum (classic finding), left pleural effusion (serosanguineous), descending aorta wider than ascending.
ECG: absence of ischemic changes helps distinguish from ACS — though right coronary ostium involvement can cause inferior ST elevation.
Related Entities (Acute Aortic Syndromes)
- Intramural hematoma (IMH) — hemorrhage within the media without an identifiable intimal tear (10–20% of cases); appears as crescentic high-attenuation aortic wall thickening on non-contrast CT; managed similarly to dissection. Can result from vasa vasorum rupture or penetrating ulcer.
- Penetrating atherosclerotic ulcer — plaque erosion into the media; found mainly in the mid-to-distal descending thoracic aorta; can progress to IMH, dissection, or rupture.
Management
Immediate (All Types)
- IV beta-blocker first to reduce dP/dt (rate of pressure change with each cardiac cycle): esmolol (500 mcg/kg bolus, then 0.05–0.2 mcg/kg/min) or metoprolol/propranolol
- Target HR <60 bpm, then add vasodilator (sodium nitroprusside, nicardipine, clevidipine) to achieve SBP <120 mmHg within 30 minutes
- Critical rule: Start beta-blocker before vasodilator — vasodilators alone cause reflex tachycardia, worsening dP/dt
- Labetalol (α + β block) is an alternative but has a long half-life (~4–6 h), limiting titratability
- Isolated hydralazine is contraindicated (increases shear stress and heart rate)
- ICU admission for hemodynamic monitoring
Type A — Surgical Emergency
- Immediate operative repair is the standard of care
- Untreated Type A: mortality >50% within 48 hours
- Surgery involves excision of the intimal flap, obliteration of the false lumen, and replacement of the ascending aorta (with or without arch/root replacement)
- In octogenarians with uncomplicated Type A: surgical management still favored (63% 1-year survival vs. 38–55% with medical management alone)
- Type A with neurologic deficit, mesenteric ischemia, or need for CPR: medical management may be preferable given extreme operative risk
Type B — Medical ± Endovascular
- Uncomplicated Type B: medical management; similar short-term mortality to surgery
- Complicated Type B (rupture, refractory pain, progression, branch vessel ischemia, Marfan syndrome): endovascular thoracic aortic repair (TEVAR) preferred over open surgery
- Endovascular options: stent graft (seals entry tear, promotes false lumen thrombosis), fenestration of the intimal flap (for static obstruction)
- Ongoing false lumen flow risks aneurysmal dilation and rupture in 20–50% within 1–5 years
- Long-term surveillance with CT or MRI for all patients
Prognosis
- Type A untreated: mortality ~1–2% per hour in early hours
- Type A with surgery: operative mortality ~15–25% overall; higher in elderly and complicated cases
- Type B uncomplicated: 30-day mortality ~10% with medical management; lower with TEVAR in selected cases
- Chronic phase survivors require lifelong antihypertensive therapy and imaging surveillance
Sources: Harrison's Principles of Internal Medicine 22E; Braunwald's Heart Disease; Robbins & Kumar Basic Pathology; Current Surgical Therapy 14e; Rosen's Emergency Medicine; Grainger & Allison's Diagnostic Radiology
Cardiovascular disease according to Harrison
Reading File
Finding Sources
Finding Sources
Finding Sources
Asking for Preferences
This is a shared conversation. Sign in to Orris to start your own chat.