Trans membrane potential
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
Reading File
Reading File
I now have comprehensive information to write a thorough answer on transmembrane potential. Let me compile it.
Transmembrane Potential
The transmembrane potential (also called the membrane potential) is the electrical potential difference across a cell membrane, expressed as the intracellular potential relative to the extracellular potential. By convention, a negative value means the inside is more negative than the outside.
1. Definition and Convention
The transmembrane potential is measured in millivolts (mV) and is defined as:
V_m = V_inside − V_outside
At rest ("resting membrane potential"), most excitable cells maintain a V_m of −70 to −90 mV — the inside is negative relative to the outside.
2. Ionic Basis — How It Is Generated
The potential arises from unequal distribution of ions across the membrane and differential membrane permeability to those ions.
Key ion concentrations (nerve fiber):
| Ion | Intracellular | Extracellular |
|---|---|---|
| Na⁺ | 14 mEq/L | 142 mEq/L |
| K⁺ | 140 mEq/L | 4 mEq/L |
| Cl⁻ | Low | High |
(Guyton and Hall Textbook of Medical Physiology)
Three contributing factors:
A. K⁺ diffusion potential
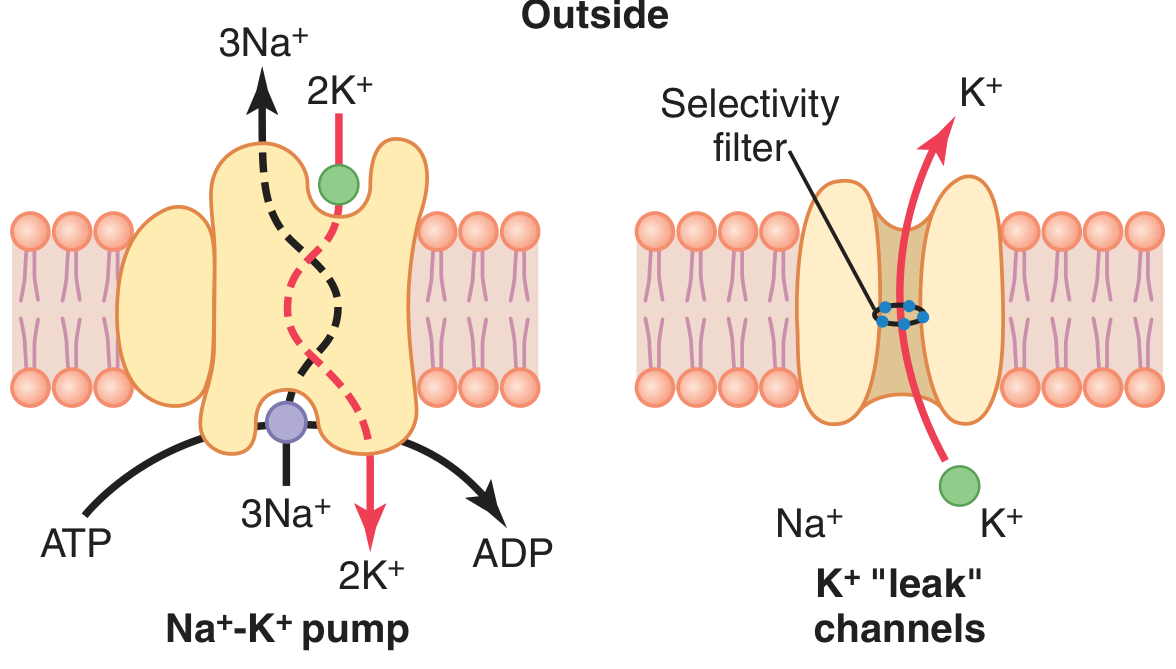
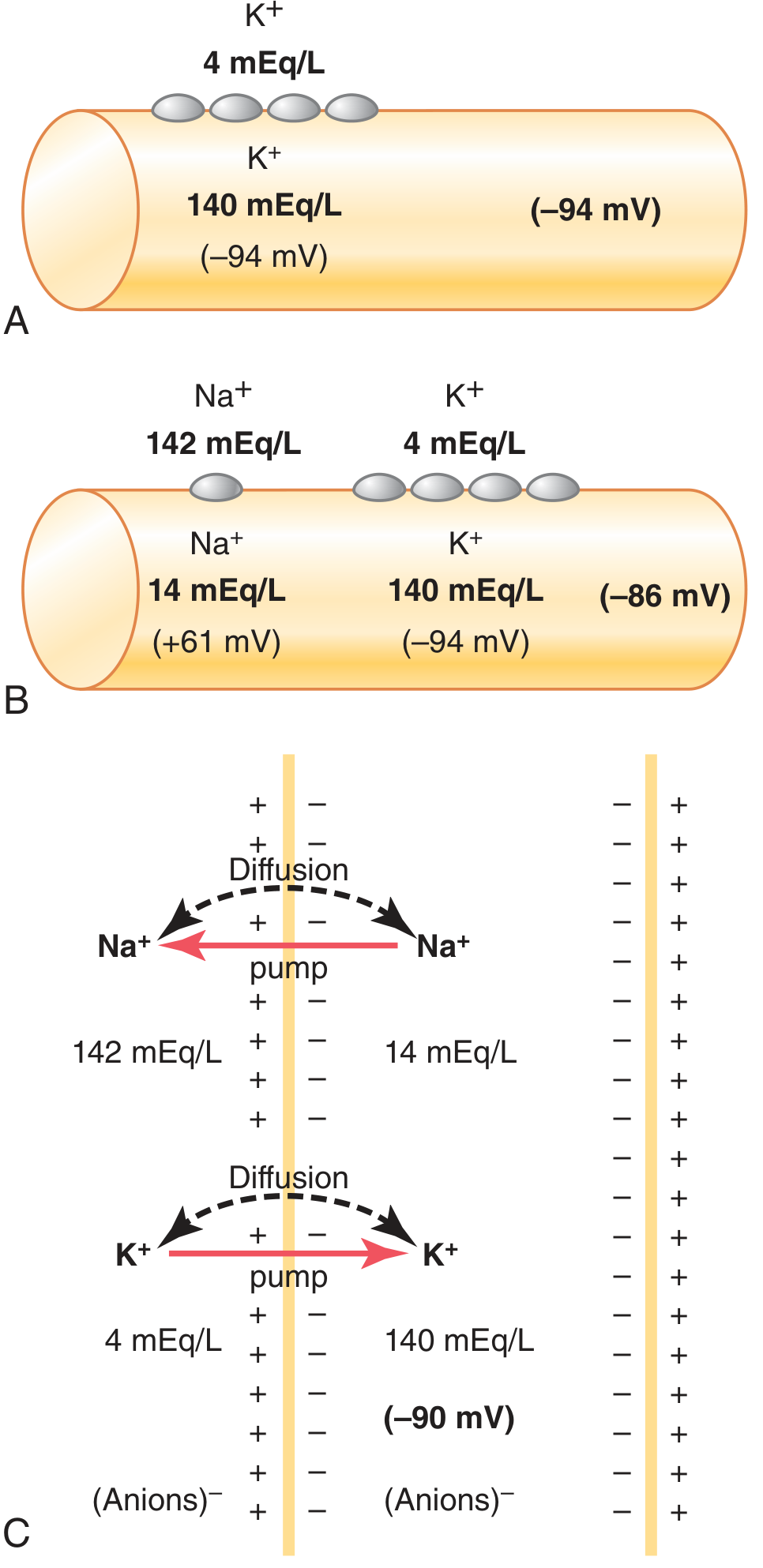
K⁺ is highly concentrated inside the cell (140 vs. 4 mEq/L, ratio ~35:1). At rest, the membrane is far more permeable to K⁺ than to Na⁺ (about 100× more). K⁺ leaks outward through K⁺ "leak" channels, leaving behind intracellular negative charges. This alone would produce a potential of approximately −94 mV (the K⁺ Nernst potential).
B. Na⁺ diffusion potential (opposing effect)
Slight permeability to Na⁺ allows a small inward Na⁺ leak (Nernst potential = +61 mV). Combining both ions via the Goldman equation gives a net V_m of approximately −86 mV.
C. The Na⁺–K⁺ ATPase (electrogenic pump)
The pump exports 3 Na⁺ out for every 2 K⁺ in — a net outward movement of positive charge — adding approximately −4 mV more negativity. The total resting membrane potential is thus ~−90 mV in neurons.
3. The Nernst Equation
The equilibrium potential (Nernst potential, E_ion) for any single ion is the transmembrane potential at which there is no net movement of that ion:
E_ion = 61 × log(C_out / C_in) (at physiological temperature, monovalent cation)
The driving force for an ion is the difference between the actual transmembrane potential and the ion's equilibrium potential:
Driving force = V_m − E_ion
This is also called the electrochemical gradient. — Katzung's Basic and Clinical Pharmacology, 16th Ed.
4. The Goldman (Goldman-Hodgkin-Katz) Equation
When multiple ions are present, the transmembrane potential depends on relative permeabilities (P) of each ion:
V_m ≈ weighted sum of equilibrium potentials, weighted by conductance
The chord conductance equation from Costanzo Physiology states:
$$E_m = \frac{G_{K^+}}{G_T}E_{K^+} + \frac{G_{Na^+}}{G_T}E_{Na^+} + \frac{G_{Cl^-}}{G_T}E_{Cl^-} + \frac{G_{Ca^{2+}}}{G_T}E_{Ca^{2+}}$$
Ions with the highest conductance/permeability drive the membrane potential toward their equilibrium potential. At rest, K⁺ and Cl⁻ dominate (high permeability); Na⁺ and Ca²⁺ contribute little (low permeability). — Costanzo Physiology, 7th Ed.
5. Resting Values by Cell Type
| Cell type | Resting V_m |
|---|---|
| Neuron (large nerve fiber) | ~−70 mV |
| Skeletal muscle | ~−90 mV |
| Cardiac ventricular muscle | ~−85 to −90 mV |
| Cardiac SA node | ~−55 to −60 mV |
| Erythrocyte | ~−10 mV |
6. Voltage-Gated Channels and Action Potentials
Voltage-gated channels (Na⁺, Ca²⁺, K⁺) sense changes in the transmembrane potential. When V_m becomes sufficiently depolarized (threshold ~−55 mV), Na⁺ channels open → rapid Na⁺ influx → action potential. The transmembrane potential transiently reverses to approximately +30 mV before repolarization restores the resting potential.
In pharmacology, many drugs exploit the transmembrane potential:
- Local anesthetics block Na⁺ channel gates (voltage-sensor residues perpendicular to the membrane plane respond to transmembrane potential changes)
- Antiarrhythmics target voltage-gated Na⁺, K⁺, and Ca²⁺ channels
- Hyperkalemia reduces the transmembrane potential (less negative), destabilizing the membrane — treatment involves stabilizing the transmembrane potential (calcium gluconate), shifting K⁺ intracellularly (insulin, bicarbonate), and eliminating excess K⁺ — Current Surgical Therapy, 14th Ed.
Summary: The transmembrane potential results from ion concentration gradients (maintained by the Na⁺–K⁺ ATPase) and differential membrane permeability. K⁺ is the dominant determinant of the resting potential (~−90 mV in neurons). Dynamic changes in transmembrane potential underlie action potentials, synaptic transmission, muscle contraction, and cardiac rhythm — and are the target of a wide range of pharmacological agents.
This is a shared conversation. Sign in to Orris to start your own chat.