Recipient in drugs
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Here is a comprehensive overview of receptors in pharmacology, drawn from authoritative textbooks:
Receptors in Drugs (Pharmacology)
What Is a Receptor?
A receptor is any biologic molecule to which a drug binds and produces a measurable response. Receptors are most commonly membrane-bound proteins, but enzymes, nucleic acids, and structural proteins can also serve as drug receptors.
"Most drugs exert effects, both beneficial and harmful, by interacting with specialized target macromolecules called receptors, which are present on or in the cell." — Lippincott Illustrated Reviews: Pharmacology
Signal Transduction
When a drug (agonist) binds to a receptor, it triggers signal transduction — a cascade of intracellular events that produce a biological response. The drug is the signal; the receptor is the detector.
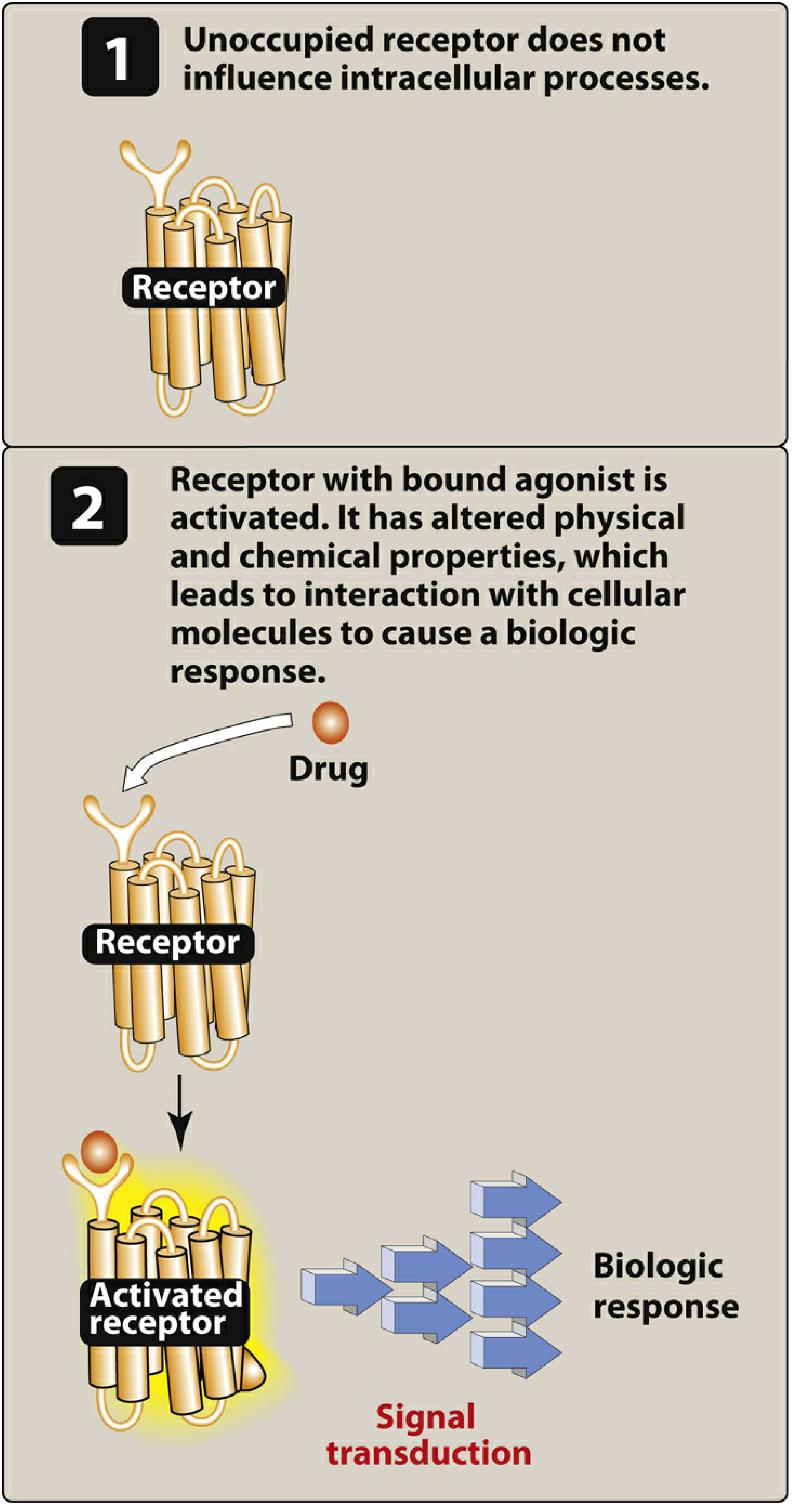
Receptor States
Receptors exist in at least two states:
- Inactive (R) — the resting state (favored in the absence of a drug)
- Active (R)* — the activated state
| Drug Type | Effect on Receptor |
|---|---|
| Agonist | Shifts R → R*, producing maximum effect |
| Antagonist | Binds receptor but does NOT activate R*; blocks agonists |
| Partial agonist | Shifts R → R* but less than full agonist |
The Four Major Receptor Families
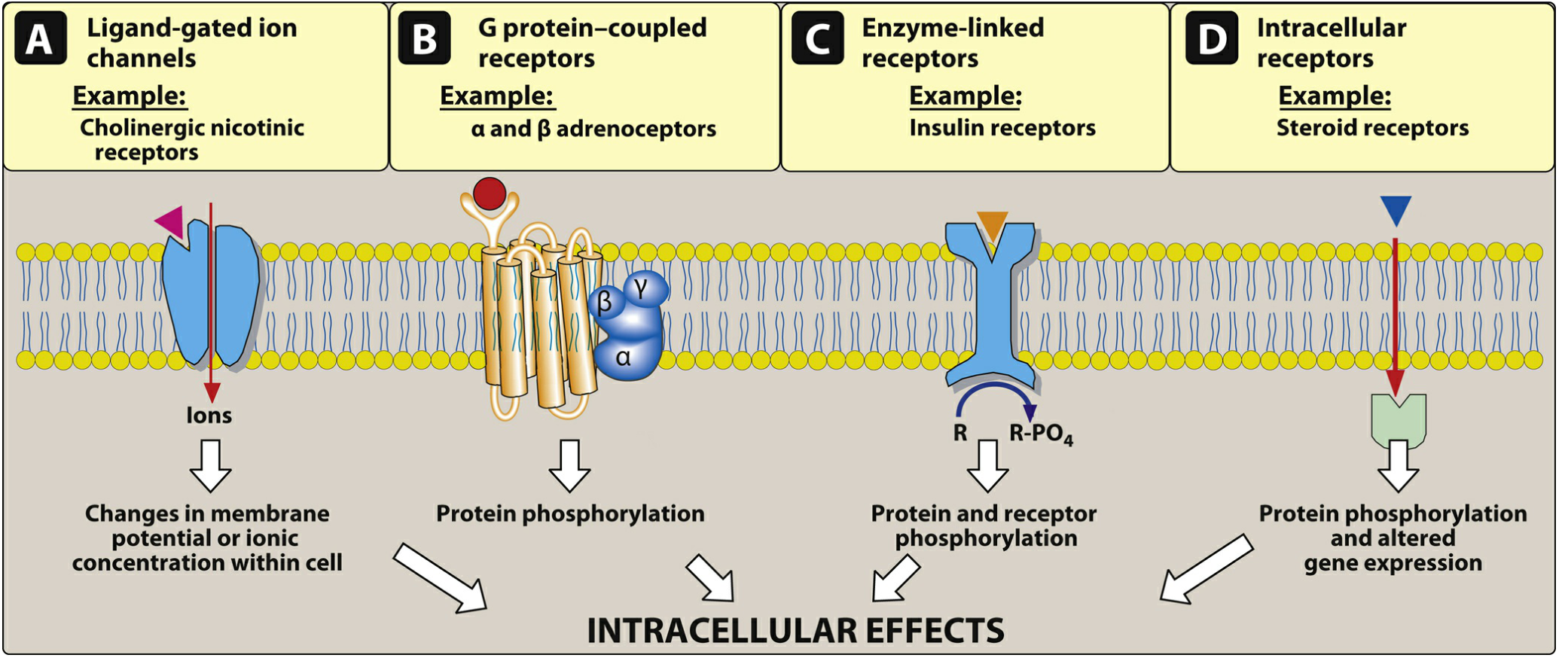
| Family | Location | Mechanism | Examples |
|---|---|---|---|
| 1. Ligand-gated ion channels | Cell surface | Drug binding opens ion channels (ms response) | Nicotinic ACh receptor (Na⁺/K⁺), GABA-A receptor (Cl⁻) |
| 2. G protein–coupled receptors (GPCRs) | Cell surface | Drug activates G protein → second messenger cascade (seconds–minutes) | β-adrenergic, muscarinic receptors |
| 3. Enzyme-linked receptors | Cell surface | Drug activates intracellular kinase | Insulin receptor (tyrosine kinase) |
| 4. Intracellular receptors | Inside cell | Lipid-soluble drug crosses membrane → alters gene transcription (hours–days) | Steroid hormones, thyroid hormone |
Hydrophilic ligands bind surface receptors; hydrophobic ligands cross the membrane to intracellular receptors.
Drug–Receptor Bonds
Three types of chemical bonds form between drugs and receptors (Katzung):
| Bond Type | Strength | Reversibility | Example |
|---|---|---|---|
| Covalent | Very strong | Often irreversible | Aspirin → cyclooxygenase; DNA-alkylating agents |
| Electrostatic (ionic, H-bonds, van der Waals) | Moderate to weak | Reversible | Most drugs |
| Hydrophobic | Weak | Reversible | Lipid-soluble drugs in membrane "pockets" |
Drugs with weaker bonds are generally more selective, because they require a very precise fit to the receptor.
Drug Shape and Chirality
The drug's shape must be complementary to the receptor (like a key to a lock). More than half of useful drugs are chiral molecules (enantiomers). Typically, one enantiomer fits the receptor better and is far more potent (e.g., carvedilol: the S-isomer is a potent β-blocker, while the R-isomer is 100× weaker at β-receptors).
Quantitative Drug–Receptor Relationships
The law of mass action governs binding:
Drug + Receptor ⇌ Drug–Receptor Complex → Biologic Effect
- Kd (dissociation constant): Drug concentration at which 50% of receptors are occupied. Lower Kd = higher affinity.
- Efficacy: The maximum response a drug can produce (Emax). More clinically important than potency.
- Potency: The dose/concentration needed to produce a given effect (EC₅₀). A drug can be highly potent but have low efficacy.
Example: Morphine has greater efficacy than naproxen (can relieve severe cancer pain); naproxen and ibuprofen have equal efficacy for headache, though different potencies. — Lippincott Illustrated Reviews: Pharmacology
Key Summary
- Receptors are the molecular targets of most drugs
- Agonists activate receptors; antagonists block them
- Four receptor families differ in location, mechanism, and response speed
- Bond type determines selectivity; Kd determines affinity; Emax determines efficacy
Sources: Lippincott Illustrated Reviews: Pharmacology; Katzung's Basic and Clinical Pharmacology, 16th Ed.
Excipient
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Here is a comprehensive overview of excipients in pharmaceuticals, synthesized from medical textbooks:
Excipients in Drugs
Definition
An excipient is any ingredient in a pharmaceutical formulation that is not the active drug (the pharmacologically active ingredient). They are sometimes called "inactive ingredients" — but this is a misnomer.
"There is really no such thing as an inactive ingredient (or excipient). Ingredients can modulate penetration, enhance the activity of other ingredients, or inactivate them depending on the order in which they are used." — Fitzpatrick's Dermatology, 5e
Excipients are added to drug products to serve specific functional roles in the formulation.
Why Excipients Are Used
| Purpose | Role |
|---|---|
| Bulk / Volume | Make a tablet or capsule large enough to handle |
| Stability | Protect the active drug from heat, light, pH, oxidation |
| Drug delivery | Control how and where the drug is released |
| Manufacturing | Aid in tablet compression, mixing, and filling |
| Palatability | Improve taste, smell, or appearance |
| Preservation | Prevent microbial contamination |
Categories of Excipients
1. Fillers / Diluents
Add bulk to a formulation. Examples: lactose, microcrystalline cellulose, starch, mannitol.
2. Binders
Hold tablet ingredients together. Examples: povidone (PVP), hydroxypropyl cellulose.
3. Disintegrants
Help tablets break apart in the GI tract for drug release. Examples: croscarmellose sodium, sodium starch glycolate.
4. Lubricants & Glidants
Prevent ingredients from sticking to manufacturing equipment and improve powder flow. Example: magnesium stearate, talc.
5. Stabilizers
Protect against degradation during manufacturing, transport, and storage. Used extensively in vaccines:
"Stabilizers used in vaccines include sugars (eg, lactose or sucrose), amino acids (eg, glycine), or proteins (eg, gelatin)." — Red Book 2021 (AAP)
6. Preservatives
Prevent microbial growth, especially in multi-dose vials. Examples:
- Thimerosal (ethyl mercury compound, used in some influenza vaccines)
- Formaldehyde (to inactivate viral toxins)
- Phenol derivatives
"Preservatives are included in multidose vials of vaccines as a safety measure to prevent the growth of microorganisms." — Red Book 2021
7. Adjuvants (in vaccines)
Enhance the immune response to the antigen. Not technically excipients in the narrow sense, but non-antigen components.
- Aluminum salts — most widely used; in hepatitis B, diphtheria-tetanus vaccines
- Oil-in-water emulsions — used in adult vaccines
8. Diluents (in vaccines/injections)
Liquids used to reconstitute lyophilized (freeze-dried) vaccine powders. Must be product-specific — only the supplied diluent should be used.
9. Solubilizers / Carriers
Help poorly soluble drugs dissolve. Example: hydroxypropyl-β-cyclodextrin (HPCD) — used in itraconazole oral solution and IV voriconazole.
⚠ HPCD accumulates in renal failure — IV voriconazole and itraconazole oral solution are contraindicated if GFR <50 mL/min. — Harriet Lane Handbook, 23e
Clinical Importance of Excipients
Excipients are not always inert and can cause significant clinical problems:
| Issue | Example |
|---|---|
| Hypersensitivity/allergy | Vaccine reactions often due to excipients (gelatin, antibiotics, latex), not the antigen — Red Book 2021 |
| Organ toxicity | Cyclodextrin excipient accumulates in renal failure |
| Teratogenicity | NMP (N-methyl-2-pyrrolidone), excipient in Sublocade (buprenorphine), associated with miscarriage and developmental delays in animals — Creasy & Resnik's Maternal-Fetal Medicine |
| Pulmonary disease | Talc (a lubricant excipient) can cause granulomatous lung disease ("excipient lung") in IV drug abusers — Goldman-Cecil Medicine |
| Drug interactions | Excipients can modulate drug absorption or penetration |
Key Takeaway
Excipients are pharmacologically functional, not merely passive carriers. Clinicians should be aware of them when:
- A patient has an allergy to a vaccine or drug product
- A patient has renal or hepatic impairment that affects excipient clearance
- A patient is pregnant (some excipients carry fetal risk)
- Generic vs. brand differences in excipients may affect bioavailability or tolerability
Sources: Fitzpatrick's Dermatology 5e; Red Book 2021 (AAP); Harriet Lane Handbook 23e; Goldman-Cecil Medicine; Creasy & Resnik's Maternal-Fetal Medicine
Why amoebic liver abscess is more common in alcoholic males
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Now I have comprehensive information. Here is a detailed, evidence-based answer:
Why Amoebic Liver Abscess (ALA) Is More Common in Alcoholic Males
ALA is 10 times more common in men than women, and alcohol is a well-established risk factor. This male-and-alcohol predominance is multifactorial.
1. Male Sex Predominance — The Hormonal Factor
"Although there is no difference in gender distribution of patients with intestinal amebiasis, there is a large male predominance in patients with amebic abscess." — Yamada's Textbook of Gastroenterology, 7e
Intestinal E. histolytica infection rates are equal in men and women, yet liver abscess is vastly more common in men. This means the male sex predisposes specifically to hepatic invasion, not just intestinal infection.
The proposed mechanisms include:
- Testosterone promotes hepatic invasion: Testosterone appears to suppress the local hepatic immune response, facilitating abscess formation. Animal models show that male sex hormones increase susceptibility to hepatic amebic disease.
- Estrogen is protective: Estrogen has been shown to enhance immune-mediated clearance of E. histolytica at the hepatic level, which is why premenopausal women are relatively protected.
- This also explains why the disease is unusual in children (pre-pubertal, low androgen levels) and rare in women of any age.
2. Alcohol — Immunosuppression and Barrier Breakdown
Alcohol creates multiple vulnerabilities:
a) Impaired Innate Hepatic Immunity
- The liver's Kupffer cells (resident macrophages) are the first line of defense against E. histolytica trophozoites arriving via the portal vein.
- Chronic alcohol use suppresses Kupffer cell function and reduces macrophage amebicidal activity.
- Macrophages acquire amebicidal activity through stimulation by IFN-γ and TNF-α — both of which are dampened by alcohol.
b) Compromised Gut Barrier
- Alcohol damages the intestinal mucosal barrier, increasing gut permeability ("leaky gut").
- This allows trophozoites to more easily invade the intestinal wall and enter the portal circulation.
c) Malnutrition
- Chronic alcoholics are often malnourished. Malnourished persons are explicitly listed as a high-risk group for severe amebiasis.
"Several groups are at increased risk for severe amebiasis, including the very young or old, malnourished persons, pregnant women, and patients treated with corticosteroids." — Goldman-Cecil Medicine
d) Impaired Cell-Mediated Immunity
- Effective clearance of E. histolytica requires a robust IFN-γ response (Th1) and mucosal IgA against the Gal/GalNAc lectin.
- Alcohol suppresses both arms — Th1 cytokines and mucosal immunity — leaving the liver unprotected from invading trophozoites.
e) Altered Gut Microbiome
- The gut microbiome composition influences symptomatic amebiasis by regulating the inflammatory immune response.
- Alcohol disrupts the gut microbiome (dysbiosis), which may further promote amebic invasion and hepatic spread.
3. The Pathogenesis Pathway
The sequence from intestinal infection to liver abscess:
Ingestion of E. histolytica cysts (fecal-oral route)
↓
Excystation in the colon → trophozoites
↓
Gal/GalNAc lectin binds colonic epithelium → invasion
↓
Entry into portal venous circulation
↓
Arrival at liver → normally cleared by Kupffer cells
↓ (in alcoholic males — impaired clearance)
Trophozoites survive → hepatic tissue destruction → ABSCESS
"Hepatic infection occurs via invasion of the portal venous circulation. Population studies suggest that the rates of amebic liver abscess are less than 1% of the rates of symptomatic enteric amebiasis." — Yamada's Textbook of Gastroenterology
The portal venous route means every intestinal infection exposes the liver, but only those with impaired hepatic defenses (males, alcoholics) progress to abscess.
4. Epidemiological Profile
"The typical patient with an amebic liver abscess in the United States is a 20- to 40-year-old Hispanic male immigrant, with fever, right upper quadrant pain, leukocytosis, and abnormal liver enzymes." — Goldman-Cecil Medicine
Key risk profile:
| Factor | Why It Matters |
|---|---|
| Male sex | Testosterone suppresses hepatic immune defense; estrogen is protective |
| Alcohol use | Impairs Kupffer cells, gut barrier, Th1 immunity, mucosal IgA |
| Age 20–40 | Peak testosterone + highest alcohol consumption demographic |
| Endemic region origin | Exposure to E. histolytica via contaminated food/water |
| Malnutrition | Impairs innate and adaptive immunity |
Summary
ALA is more common in alcoholic males because:
- Testosterone suppresses hepatic immunity, favoring progression from intestinal to hepatic disease
- Alcohol impairs multiple immune layers — Kupffer cell function, Th1/IFN-γ responses, mucosal IgA, and gut barrier integrity
- Together these factors allow E. histolytica trophozoites that reach the liver via the portal vein to escape clearance and establish an abscess
Sources: Goldman-Cecil Medicine; Yamada's Textbook of Gastroenterology 7e; Harrison's Principles of Internal Medicine 22e
This is a shared conversation. Sign in to Orris to start your own chat.